Abstract
Main conclusions
Eukaryotic microalgae hold incredible metabolic potential for the sustainable production of heterologous isoprenoid products. Recent advances in algal engineering have enabled the demonstration of prominent examples of heterologous isoprenoid production.
Isoprenoids, also known as terpenes or terpenoids, are the largest class of natural chemicals, with a vast diversity of structures and biological roles. Some have high-value in human-use applications, although may be found in their native contexts in low abundance or be difficult to extract and purify. Heterologous production of isoprenoid compounds in heterotrophic microbial hosts such as bacteria or yeasts has been an active area of research for some time and is now a mature technology. Eukaryotic microalgae represent sustainable alternatives to these hosts for biotechnological production processes as their cultivation can be driven by light and freely available CO2 as a carbon source. Their photosynthetic lifestyles require metabolic architectures structured towards the generation of associated isoprenoids (carotenoids, phytol) which participate in photon capture, energy dissipation, and electron transfer. Eukaryotic microalgae should, therefore, contain inherently high capacities for the generation of heterologous isoprenoid products. Although engineering strategies in eukaryotic microalgae have lagged behind the more genetically tractable bacteria and yeasts, recent advances in algal engineering concepts have demonstrated prominent examples of light-driven heterologous isoprenoid production from these photosynthetic hosts. This work seeks to provide practical insights into the choice of eukaryotic microalgae as biotechnological chassis. Recent reports of advances in algal engineering for heterologous isoprenoid production are highlighted as encouraging examples that promote their expanded use as sustainable green-cell factories. Current state of the art, limitations, and future challenges are also discussed.
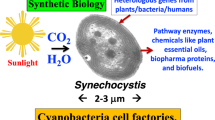
Graphical abstract






Picture courtesy of Dr. Olga Blifernez-Klassen, reproduced with permission







Similar content being viewed by others
Abbreviations
- IPP:
-
Isopentenyl pyrophosphate
- DMAPP:
-
Dimethylallyl pyrophosphate
- GPP(s):
-
Geranyl pyrophosphate (synthase)
- FPP(s):
-
Farnesyl pyrophosphate (synthase)
- GGPP(s):
-
Geranylgeranyl pyrophosphate (synthase)
- YFP:
-
Yellow fluorescent protein
- CFP:
-
Cyan fluorescent protein
- RFP:
-
Red fluorescent protein
- TPS:
-
Terpene synthase
- sTPS:
-
Sesquiterpenoid synthase
- diTPS:
-
Diterpene synthase
- tTPS:
-
Triterpenoid synthase
- CYP:
-
Cytochrome P450 monooxygenase
References
Ajjawi I, Verruto J, Aqui M et al (2017) Lipid production in Nannochloropsis gaditana is doubled by decreasing expression of a single transcriptional regulator. Nat Biotechnol 35:647–652. https://doi.org/10.1038/nbt.3865
Albertsen L, Chen Y, Bach LS et al (2011) Diversion of flux toward sesquiterpene production in Saccharomyces cerevisiae by fusion of host and heterologous enzymes. Appl Environ Microbiol 77:1033–1040. https://doi.org/10.1128/AEM.01361-10
Andersen-Ranberg J, Kongstad KT, Nielsen MT et al (2016) Expanding the landscape of diterpene structural diversity through stereochemically controlled combinatorial biosynthesis. Angew Chemie 128:2182–2186. https://doi.org/10.1002/ange.201510650
Archibald JM (2012) the evolution of algae by secondary and tertiary endosymbiosis. In: Piganeau G (ed) Advances in botanical research. Elsevier, New York, pp 87–118
Baier T, Wichmann J, Kruse O, Lauersen KJ (2018) Intron-containing algal transgenes mediate efficient recombinant gene expression in the green microalga Chlamydomonas reinhardtii. Nucleic Acids Res 46:6909–6919. https://doi.org/10.1093/nar/gky532
Barahimipour R, Neupert J, Bock R (2016) Efficient expression of nuclear transgenes in the green alga Chlamydomonas: synthesis of an HIV antigen and development of a new selectable marker. Plant Mol Biol. https://doi.org/10.1007/s11103-015-0425-8
Benemann J (2013) Microalgae for biofuels and animal feeds. Energies 6:5869–5886. https://doi.org/10.3390/en6115869
Black JB, Perez-Pinera P, Gersbach CA (2017) Mammalian synthetic biology: engineering biological systems. Annu Rev Biomed Eng 19:249–277. https://doi.org/10.1146/annurev-bioeng-071516-044649
Bogen C, Al-Dilaimi A, Albersmeier A et al (2013) Reconstruction of the lipid metabolism for the microalga Monoraphidium neglectum from its genome sequence reveals characteristics suitable for biofuel production. BMC Genomics 14:926. https://doi.org/10.1186/1471-2164-14-926
Bohlmann J, Keeling CI (2008) Terpenoid biomaterials. Plant J 54:656–669. https://doi.org/10.1111/j.1365-313X.2008.03449.x
Bohlmann J, Crock J, Jetter R, Croteau R (1998) Terpenoid-based defenses in conifers: cDNA cloning, characterization, and functional expression of wound-inducible (E)-alpha-bisabolene synthase from grand fir (Abies grandis). Proc Natl Acad Sci USA 95:6756–6761. https://doi.org/10.1073/pnas.95.12.6756
Bowler C, Allen AE, Badger JH et al (2008) The Phaeodactylum genome reveals the evolutionary history of diatom genomes. Nature 456:239–244. https://doi.org/10.1038/nature07410
Bruggeman AJ, Kuehler D, Weeks DP (2014) Evaluation of three herbicide resistance genes for use in genetic transformations and for potential crop protection in algae production. Plant Biotechnol J 12:894–902. https://doi.org/10.1111/pbi.12192
Buckingham J, Macdonald FM, Bradley HM et al (1994) Dictionary of natural products, 1st edn. Chapman and Hall, London
Cabanelas ITD, Marques SSI, de Souza CO et al (2015) Botryococcus, what to do with it? Effect of nutrient concentration on biorefinery potential. Algal Res 11:43–49. https://doi.org/10.1016/j.algal.2015.05.009
Carrera Pacheco SE, Hankamer B, Oey M (2018) Optimising light conditions increases recombinant protein production in Chlamydomonas reinhardtii chloroplasts. Algal Res 32:329–340. https://doi.org/10.1016/j.algal.2018.04.011
Chandran SS, Kealey JT, Reeves CD (2011) Microbial production of isoprenoids. Process Biochem 46:1703–1710. https://doi.org/10.1016/j.procbio.2011.05.012
Chaves JE, Melis A (2018) Engineering isoprene synthesis in cyanobacteria. FEBS Lett. https://doi.org/10.1002/1873-3468.13052
Corteggiani Carpinelli E, Telatin A, Vitulo N et al (2014) Chromosome scale genome assembly and transcriptome profiling of nannochloropsis gaditana in nitrogen depletion. Mol Plant 7:323–335. https://doi.org/10.1093/mp/sst120
Crozet P, Navarro FJ, Willmund F et al (2018) Birth of a photosynthetic chassis: a MoClo toolkit enabling synthetic biology in the microalga Chlamydomonas reinhardtii. ACS Synth Biol. https://doi.org/10.1021/acssynbio.8b00251
D’Adamo S, Schiano di Visconte G, Lowe G et al (2018) Engineering The Unicellular Alga Phaeodactylum tricornutum For high-value plant triterpenoid production. Plant Biotechnol J. https://doi.org/10.1111/pbi.12948
Danielewicz M, Anderson LA, Franz AK (2011) Triacylglycerol profiling of marine microalgae by mass spectrometry. J Lipid Res 52:2101–2108. https://doi.org/10.1194/jlr.D018408
Davies FK, Jinkerson RE, Posewitz MC (2015) Toward a photosynthetic microbial platform for terpenoid engineering. Photosynth Res 123:265–284. https://doi.org/10.1007/s11120-014-9979-6
Deguerry F, Pastore L, Wu S et al (2006) The diverse sesquiterpene profile of patchouli, Pogostemon cablin, is correlated with a limited number of sesquiterpene synthases. Arch Biochem Biophys 454:123–136. https://doi.org/10.1016/j.abb.2006.08.006
Dong B, Hu HH, Li ZF et al (2017) A novel bicistronic expression system composed of the intraflagellar transport protein gene ift25 and FMDV 2A sequence directs robust nuclear gene expression in Chlamydomonas reinhardtii. Appl Microbiol Biotechnol 101:4227–4245. https://doi.org/10.1007/s00253-017-8177-9
Duncker BP, Davies PL, Walker VK (1997) Introns boost transgene expression in Drosophila melanogaster. Mol Gen Genet 254:291–296. https://doi.org/10.1007/s004380050418
Dyo YM, Purton S (2018) The algal chloroplast as a synthetic biology platform for production of therapeutic proteins. Microbiology. https://doi.org/10.1099/mic.0.000599
Eichler-Stahlberg A, Weisheit W, Ruecker O, Heitzer M (2009) Strategies to facilitate transgene expression in Chlamydomonas reinhardtii. Planta 229:873–883. https://doi.org/10.1007/s00425-008-0879-x
Eilers U, Bikoulis A, Breitenbach J et al (2016) Limitations in the biosynthesis of fucoxanthin as targets for genetic engineering in Phaeodactylum tricornutum. J Appl Phycol 28:123–129. https://doi.org/10.1007/s10811-015-0583-8
Engler C, Kandzia R, Marillonnet S (2008) A one pot, one step, precision cloning method with high throughput capability. PLoS One. https://doi.org/10.1371/journal.pone.0003647
Eroglu E, Melis A (2010) Extracellular terpenoid hydrocarbon extraction and quantitation from the green microalgae Botryococcus braunii var. Showa. Bioresour Technol 101:2359–2366. https://doi.org/10.1016/j.biortech.2009.11.043
Fabregas J, Abalde J, Herrero C et al (1984) Growth of the marine microalga Tetraselmis suecica in batch cultures with different salinities and nutrient concentrations. Aquaculture 42:207–215. https://doi.org/10.1016/0044-8486(84)90101-7
Fairley P (2011) Introduction: next generation biofuels. Nature 474:S2–S5. https://doi.org/10.1038/474S02a
Fernández E, Schnell R, Ranum LP et al (1989) Isolation and characterization of the nitrate reductase structural gene of Chlamydomonasreinhardtii. Proc Natl Acad Sci 86:6449–6453
Fresewinkel M, Rosello R, Wilhelm C et al (2014) Integration in microalgal bioprocess development: design of efficient, sustainable, and economic processes. Eng Life Sci. https://doi.org/10.1002/elsc.201300153
Fuhrmann M, Oertel W, Hegemann P (1999) A synthetic gene coding for the green fluorescent protein (GFP) is a versatile reporter in Chlamydomonas reinhardtii. Plant J 19:353–361. https://doi.org/10.1046/j.1365-313X.1999.00526.x
Gallegos JE, Rose AB (2015) The enduring mystery of intron-mediated enhancement. Plant Sci 237:8–15. https://doi.org/10.1016/j.plantsci.2015.04.017
Goldschmidt-Clermont M (1991) Transgenic expression of aminoglycoside adenine transferase in the chloroplast: a selectable marker of site-directed transformation of Chlamydomonas. Nucleic Acids Res 19:4083–4089
Gorman DS, Levine RP (1965) Cytochrome f and plastocyanin: their sequence in the photosynthetic electron transport chain of Chlamydomonas reinhardi. Proc Natl Acad Sci 54:1665–1669. https://doi.org/10.1073/pnas.54.6.1665
Gruchattka E, Kayser O (2015) In vivo validation of in silico predicted metabolic engineering strategies in yeast: disruption of α-ketoglutarate dehydrogenase and expression of ATP-citrate lyase for terpenoid production. PLoS One 10:e0144981. https://doi.org/10.1371/journal.pone.0144981
Gruchattka E, Hädicke O, Klamt S et al (2013) In silico profiling of Escherichia coli and Saccharomyces cerevisiae as terpenoid factories. Microb Cell Fact 12:84. https://doi.org/10.1186/1475-2859-12-84
Gumpel NJ, Rochaix JD, Purton S (1994) Studies on homologous recombination in the green alga Chlamydomonas reinhardtii. Curr Genet 26:438–442
Hallmann A (2007) Algal transgenics and biotechnology. Transgenic Plant J 1:81–98
Hallmann A (2016) Algae biotechnology—green cell-factories on the rise. Curr Biotechnol 4:389–415. https://doi.org/10.2174/2211550105666151107001338
Hallmann A, Rappel A (1999) Genetic engineering of the multicellular green alga Volvox: a modified and multiplied bacterial antibiotic resistance gene as a dominant selectable marker. Plant J 17:99–109. https://doi.org/10.1046/j.1365-313X.1999.00342.x
Henke N, Wichmann J, Baier T et al (2018) Patchoulol production with metabolically engineered Corynebacterium glutamicum. Genes (Basel) 9:219. https://doi.org/10.3390/genes9040219
Hu Q, Sommerfeld M, Jarvis E et al (2008) Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J 54:621–639. https://doi.org/10.1111/j.1365-313X.2008.03492.x
Ignea C, Trikka FA, Nikolaidis AK et al (2015) Efficient diterpene production in yeast by engineering Erg20p into a geranylgeranyl diphosphate synthase. Metab Eng 27:65–75. https://doi.org/10.1016/j.ymben.2014.10.008
Jaeger D, Hübner W, Huser T et al (2017) Nuclear transformation and functional gene expression in the oleaginous microalga Monoraphidium neglectum. J Biotechnol 249:10–15. https://doi.org/10.1016/j.jbiotec.2017.03.011
Jakob G, Wolf J, Bui TVL et al (2013) Surveying a diverse pool of microalgae as a bioresource for future biotechnological applications. J Pet Environ Biotechnol 4:1–8. https://doi.org/10.4172/2157-7463.1000153
Jiang L, Luo S, Fan X et al (2011) Biomass and lipid production of marine microalgae using municipal wastewater and high concentration of CO2. Appl Energy 88:3336–3341. https://doi.org/10.1016/j.apenergy.2011.03.043
Kajikawa M, Kinohira S, Ando A et al (2015) Accumulation of squalene in a microalga Chlamydomonas reinhardtii by genetic modification of squalene synthase and squalene epoxidase genes. PLoS One 10:e0120446. https://doi.org/10.1371/journal.pone.0120446
Karas BJ, Diner RE, Lefebvre SC et al (2015) Designer diatom episomes delivered by bacterial conjugation. Nat Commun 6:6925. https://doi.org/10.1038/ncomms7925
Keeling PJ (2010) The endosymbiotic origin, diversification and fate of plastids. Philos Trans R Soc Lond B Biol Sci 365:729–748. https://doi.org/10.1098/rstb.2009.0103
Kilian O, Benemann CSE, Niyogi KK, Vick B (2011) High-efficiency homologous recombination in the oil-producing alga Nannochloropsis sp. Proc Natl Acad Sci USA 108:21265–21269. https://doi.org/10.1073/pnas.1105861108
Kindle KL (1990) High-frequency nuclear transformation of Chlamydomonas reinhardtii. Proc Natl Acad Sci USA 87:1228–1232
Kindle KL, Schnell RA, Fernández E, Lefebvre PA (1989) Stable nuclear transformation of Chlamydomonas using the Chlamydomonas gene for nitrate reductase. J Cell Biol 109:2589–2601. https://doi.org/10.1083/jcb.109.6.2589
Kirby J, Keasling JD (2009) Biosynthesis of plant isoprenoids: perspectives for microbial engineering. Annu Rev Plant Biol 60:335–355. https://doi.org/10.1146/annurev.arplant.043008.091955
Kruse O, Rupprecht J, Mussgnug JH et al (2005) Photosynthesis: a blueprint for solar energy capture and biohydrogen production technologies. Photochem Photobiol Sci 4:957–970. https://doi.org/10.1039/b506923h
Kumar A, Falcao VR, Sayre RT (2013) Evaluating nuclear transgene expression systems in Chlamydomonas reinhardtii. Algal Res 2:321–332. https://doi.org/10.1016/j.algal.2013.09.002
Lange BM, Rujan T, Martin W, Croteau R (2000) Isoprenoid biosynthesis: the evolution of two ancient and distinct pathways across genomes. Proc Natl Acad Sci 97:13172–13177. https://doi.org/10.1073/pnas.240454797
Larkum AWD, Ross IL, Kruse O, Hankamer B (2012) Selection, breeding and engineering of microalgae for bioenergy and biofuel production. Trends Biotechnol 30:198–205. https://doi.org/10.1016/j.tibtech.2011.11.003
Lauersen KJ, Berger H, Mussgnug JH, Kruse O (2013a) Efficient recombinant protein production and secretion from nuclear transgenes in Chlamydomonas reinhardtii. J Biotechnol 167:101–110. https://doi.org/10.1016/j.jbiotec.2012.10.010
Lauersen KJ, Vanderveer TL, Berger H et al (2013b) Ice recrystallization inhibition mediated by a nuclear-expressed and -secreted recombinant ice-binding protein in the microalga Chlamydomonas reinhardtii. Appl Microbiol Biotechnol 97:9763–9772. https://doi.org/10.1007/s00253-013-5226-x
Lauersen KJ, Huber I, Wichmann J et al (2015a) Investigating the dynamics of recombinant protein secretion from a microalgal host. J Biotechnol 215:62–71. https://doi.org/10.1016/j.jbiotec.2015.05.001
Lauersen KJ, Kruse O, Mussgnug JH (2015b) Targeted expression of nuclear transgenes in Chlamydomonas reinhardtii with a versatile, modular vector toolkit. Appl Microbiol Biotechnol 99:3491–3503. https://doi.org/10.1007/s00253-014-6354-7
Lauersen KJ, Baier T, Wichmann J et al (2016) Efficient phototrophic production of a high-value sesquiterpenoid from the eukaryotic microalga Chlamydomonas reinhardtii. Metab Eng 38:331–343. https://doi.org/10.1016/j.ymben.2016.07.013
Lauersen KJ, Wichmann J, Baier T et al (2018) Phototrophic production of heterologous diterpenoids and a hydroxy-functionalized derivative from Chlamydomonas reinhardtii. Metab Eng 49:116–127. https://doi.org/10.1016/j.ymben.2018.07.005
Leavell MD, McPhee DJ, Paddon CJ (2016) Developing fermentative terpenoid production for commercial usage. Curr Opin Biotechnol 37:114–119. https://doi.org/10.1016/j.copbio.2015.10.007
Lehr F, Posten C (2009) Closed photo-bioreactors as tools for biofuel production. Curr Opin Biotechnol 20:280–285. https://doi.org/10.1016/j.copbio.2009.04.004
Lichtenthaler HK (1999) The 1-deoxy-D-xylulose-5-phosphate pathway of isoprenoid biosynthesis in plants. Annu Rev Plant Physiol Plant Mol Biol 50:47–65. https://doi.org/10.1146/annurev.arplant.50.1.47
Lichtenthaler HK, Schwender J, Disch A, Rohmer M (1997) Biosynthesis of isoprenoids in higher plants chloroplasts proceeds via a mevalonate pathway. FEBS Lett 400:271–274
Lin X, Hezari M, Koepp AE et al (1996) Mechanism of taxadiene synthase, a diterpene cyclase that catalyzes the first step of taxol biosynthesis in Pacific yew. Biochemistry 35:2968–2977. https://doi.org/10.1021/bi9526239
Lohr M, Schwender J, Polle JEW (2012) Isoprenoid biosynthesis in eukaryotic phototrophs: a spotlight on algae. Plant Sci 185–186:9–22
Lorenz RT, Cysewski GR (2000) Commercial potential for Haematococcus microalgae as a natural source of astaxanthin. Trends Biotechnol 18:160–167
Lumbreras V, Stevens RD, Purton S et al (1998) Efficient foreign gene expression in Chlamydomonas reinhardtii mediated by an endogenous intron. Plant J 14:441–447. https://doi.org/10.1046/j.1365-313X.1998.00145.x
Luo D, Callari R, Hamberger B et al (2016) Oxidation and cyclization of casbene in the biosynthesis of Euphorbia factors from mature seeds of Euphorbia lathyris L. Proc Natl Acad Sci 113:E5082–E5089. https://doi.org/10.1073/pnas.1607504113
McFadden GI (2001) Primary and secondary endosymbiosis and the origin of plastids. J Phycol 37:951–959. https://doi.org/10.1046/j.1529-8817.2001.01126.x
McFadden GI, Van Dooren GG (2004) Evolution: red algal genome affirms a common origin of all plastids. Curr Biol 14:514–516. https://doi.org/10.1016/j.cub.2004.06.041
Melis A (2012) Photosynthesis-to-fuels: from sunlight to hydrogen, isoprene, and botryococcene production. Energy Environ Sci 5:5531–5539. https://doi.org/10.1039/C1EE02514G
Melis A (2013) Carbon partitioning in photosynthesis. Curr Opin Chem Biol 17:453–456. https://doi.org/10.1016/j.cbpa.2013.03.010
Merchant SS, Prochnik SE, Vallon O et al (2007) The Chlamydomonas genome reveals the evolution of key animal and plant functions. Science 318:245–250. https://doi.org/10.1126/science.1143609
Morweiser M, Kruse O, Hankamer B, Posten C (2010) Developments and perspectives of photobioreactors for biofuel production. Appl Microbiol Biotechnol 87:1291–1301. https://doi.org/10.1007/s00253-010-2697-x
Mussgnug JH (2015) Genetic tools and techniques for Chlamydomonas reinhardtii. Appl Microbiol Biotechnol. https://doi.org/10.1007/s00253-015-6698-7
Neupert J, Karcher D, Bock R (2009) Generation of Chlamydomonas strains that efficiently express nuclear transgenes. Plant J 57:1140–1150. https://doi.org/10.1111/j.1365-313X.2008.03746.x
Palmiter RD, Sandgren EP, Avarbock MR et al (1991) Heterologous introns can enhance expression of transgenes in mice. Proc Natl Acad Sci 88:478–482. https://doi.org/10.1073/pnas.88.2.478
Pateraki I, Andersen-Ranberg J, Hamberger B et al (2014) Manoyl oxide (13R), the biosynthetic precursor of forskolin, is synthesized in specialized root cork cells in Coleus forskohlii. Plant Physiol 164:1222–1236. https://doi.org/10.1104/pp.113.228429
Pateraki I, Heskes AM, Hamberger B (2015) Cytochromes P450 for Terpene Functionalisation and Metabolic Engineering. In: Advances in biochemical engineering/biotechnology. pp 107–139
Pateraki I, Andersen-Ranberg J, Jensen NB et al (2017) Total biosynthesis of the cyclic AMP booster forskolin from Coleus forskohlii. Elife 6:1–28. https://doi.org/10.7554/eLife.23001
Peralta-Yahya PP, Ouellet M, Chan R et al (2011) Identification and microbial production of a terpene-based advanced biofuel. Nat Commun 2:483. https://doi.org/10.1038/ncomms1494
Poliner E, Farré EM, Benning C (2018a) Advanced genetic tools enable synthetic biology in the oleaginous microalgae Nannochloropsis sp. Plant Cell Rep. https://doi.org/10.1007/s00299-018-2270-0
Poliner E, Pulman JA, Zienkiewicz K et al (2018b) A toolkit for Nannochloropsis oceanica CCMP1779 enables gene stacking and genetic engineering of the eicosapentaenoic acid pathway for enhanced long-chain polyunsaturated fatty acid production. Plant Biotechnol J 16:298–309. https://doi.org/10.1111/pbi.12772
Poliner E, Takeuchi T, Du ZY et al (2018c) Nontransgenic marker-free gene disruption by an episomal CRISPR system in the Oleaginous Microalga, Nannochloropsis oceanica CCMP1779. ACS Synth Biol 7:962–968. https://doi.org/10.1021/acssynbio.7b00362
Posten C (2009) Design principles of photo-bioreactors for cultivation of microalgae. Eng Life Sci 9:165–177. https://doi.org/10.1002/elsc.200900003
Potvin G, Zhang Z (2010) Strategies for high-level recombinant protein expression in transgenic microalgae: a review. Biotechnol Adv 28:910–918. https://doi.org/10.1016/j.biotechadv.2010.08.006
Purton S, Szaub JB, Wannathong T et al (2013) Genetic engineering of algal chloroplasts: progress and prospects. Russ J Plant Physiol 60:491–499. https://doi.org/10.1134/S1021443713040146
Radakovits R, Jinkerson RE, Fuerstenberg SI et al (2012) Draft genome sequence and genetic transformation of the oleaginous alga Nannochloropis gaditana. Nat Commun 3:686. https://doi.org/10.1038/ncomms1688
Raja R, Hemaiswarya S, Rengasamy R (2007) Exploitation of Dunaliella for beta-carotene production. Appl Microbiol Biotechnol 74:517–523. https://doi.org/10.1007/s00253-006-0777-8
Rasala BA, Lee PA, Shen Z et al (2012) Robust expression and secretion of Xylanase1 in Chlamydomonas reinhardtii by fusion to a selection gene and processing with the FMDV 2A peptide. PLoS One 7:e43349
Rasala BA, Barrera DJ, Ng J et al (2013) Expanding the spectral palette of fluorescent proteins for the green microalga Chlamydomonas reinhardtii. Plant J 74:545–556. https://doi.org/10.1111/tpj.12165
Reed J, Stephenson MJ, Miettinen K et al (2017) A translational synthetic biology platform for rapid access to gram-scale quantities of novel drug-like molecules. Metab Eng 42:185–193. https://doi.org/10.1016/j.ymben.2017.06.012
Remacle C, Cardol P, Coosemans N et al (2006) High-efficiency biolistic transformation of Chlamydomonas mitochondria can be used to insert mutations in complex I genes. Proc Natl Acad Sci USA 103:4771–4776. https://doi.org/10.1073/pnas.0509501103
Rohmer M (1999) The discovery of a mevalonate-independent pathway for isoprenoid biosynthesis in bacteria, algae and higher plants. Nat Prod Rep 16:565–574. https://doi.org/10.1039/a709175c
Rohmer M, Knani M, Simonin P et al (1993) Isoprenoid biosynthesis in bacteria: a novel pathway for the early steps leading to isopentenyl diphosphate. Biochem J 295(Pt 2):517–524. https://doi.org/10.1042/bj2950517
Rosenberg JN, Oyler GA, Wilkinson L, Betenbaugh MJ (2008) A green light for engineered algae: redirecting metabolism to fuel a biotechnology revolution. Curr Opin Biotechnol 19:430–436. https://doi.org/10.1016/j.copbio.2008.07.008
Scaife MA, Smith AG (2016) Towards developing algal synthetic biology. Biochem Soc Trans 44:716–722. https://doi.org/10.1042/BST20160061
Scala S, Carels N, Falciatore A et al (2002) Genome properties of the diatom Phaeodactylum tricornutum. Plant Physiol 129:993–1002. https://doi.org/10.1104/pp.010713
Schenk PM, Thomas-Hall SR, Stephens E et al (2008) Second generation biofuels: high-efficiency microalgae for biodiesel production. BioEnergy Res 1:20–43. https://doi.org/10.1007/s12155-008-9008-8
Schierenbeck L, Ries D, Rogge K et al (2015) Fast forward genetics to identify mutations causing a high light tolerant phenotype in Chlamydomonas reinhardtii by whole-genome-sequencing. BMC Genomics 16:1–15. https://doi.org/10.1186/s12864-015-1232-y
Schnell RA, Lefebvre PA (1993) Isolation of the chlamydomonas regulatory gene NIT2 by transposon tagging. Genetics 134:737–747
Schroda M, Blöcker D, Beck CF (2000) The HSP70A promoter as a tool for the improved expression of transgenes in Chlamydomonas. Plant J 21:121–131. https://doi.org/10.1046/j.1365-313x.2000.00652.x
Schwender J, Seemann M, Lichtenthaler HK, Rohmer M (1996) Biosynthesis of isoprenoids (carotenoids, sterols, prenyl side-chains of chlorophylls and plastoquinone) via a novel pyruvate/glyceraldehyde 3-phosphate non-mevalonate pathway in the green alga Scenedesmus obliquus. Biochem J 316:73–80. https://doi.org/10.1042/bj3160073
Scranton MA, Ostrand JT, Georgianna DR et al (2016) Synthetic promoters capable of driving robust nuclear gene expression in the green alga Chlamydomonas reinhardtii. Algal Res 15:135–142. https://doi.org/10.1016/j.algal.2016.02.011
Shao N, Bock R (2008) A codon-optimized luciferase from Gaussia princeps facilitates the in vivo monitoring of gene expression in the model alga Chlamydomonas reinhardtii. Curr Genet 53:381–388. https://doi.org/10.1007/s00294-008-0189-7
Siaut M, Heijde M, Mangogna M et al (2007) Molecular toolbox for studying diatom biology in Phaeodactylum tricornutum. Gene 406:23–35. https://doi.org/10.1016/j.gene.2007.05.022
Siddique HR, Saleem M (2011) Beneficial health effects of lupeol triterpene: a review of preclinical studies. Life Sci 88:302–306. https://doi.org/10.1016/j.lfs.2010.11.020
Singh RN, Sharma S (2012) Development of suitable photobioreactor for algae production—a review. Renew Sustain Energy Rev 16:2347–2353. https://doi.org/10.1016/j.rser.2012.01.026
Specht E, Miyake-Stoner S, Mayfield S (2010) Micro-algae come of age as a platform for recombinant protein production. Biotechnol Lett 32:1373–1383. https://doi.org/10.1007/s10529-010-0326-5
Specht EA, Karunanithi PS, Gimpel JA et al (2016) Host organisms: algae. Industrial biotechnology. Wiley, Weinheim, Germany, pp 605–641
Streatfield SJ (2007) Approaches to achieve high-level heterologous protein production in plants. Plant Biotechnol J 5:2–15. https://doi.org/10.1111/j.1467-7652.2006.00216.x
Tikhonov MV, Maksimenko OG, Georgiev PG, Korobko IV (2017) Optimal artificial mini-introns for transgenic expression in the cells of mice and hamsters. Mol Biol 51:592–595. https://doi.org/10.1134/S0026893317040173
Trikka FA, Nikolaidis A, Ignea C et al (2015) Combined metabolome and transcriptome profiling provides new insights into diterpene biosynthesis in S. pomifera glandular trichomes. BMC Genomics 16:1–19. https://doi.org/10.1186/s12864-015-2147-3
Ugwu CU, Aoyagi H, Uchiyama H (2008) Photobioreactors for mass cultivation of algae. Bioresour Technol 99:4021–4028. https://doi.org/10.1016/j.biortech.2007.01.046
Verruto J, Francis K, Wang Y et al (2018) Unrestrained markerless trait stacking in Nannochloropsis gaditana through combined genome editing and marker recycling technologies. Proc Natl Acad Sci 115:E7015–E7022. https://doi.org/10.1073/pnas.1718193115
Vickers CE, Williams TC, Peng B, Cherry J (2017) Recent advances in synthetic biology for engineering isoprenoid production in yeast. Curr Opin Chem Biol 40:47–56. https://doi.org/10.1016/j.cbpa.2017.05.017
Vieler A, Wu G, Tsai C-H et al (2012) Genome, functional gene annotation, and nuclear transformation of the heterokont oleaginous alga Nannochloropsis oceanica CCMP1779. PLoS Genet 8:e1003064. https://doi.org/10.1371/journal.pgen.1003064
Weber E, Engler C, Gruetzner R et al (2011) A modular cloning system for standardized assembly of multigene constructs. PLoS One. https://doi.org/10.1371/journal.pone.0016765
Weiner I, Atar S, Schweitzer S et al (2018) Enhancing heterologous expression in Chlamydomonas reinhardtii by transcript sequence optimization. Plant J 94:22–31. https://doi.org/10.1111/tpj.13836
Wichmann J, Baier T, Wentnagel E et al (2018) Tailored carbon partitioning for phototrophic production of (E)-α-bisabolene from the green microalga Chlamydomonas reinhardtii. Metab Eng 45:211–222. https://doi.org/10.1016/j.ymben.2017.12.010
Wijffels RH, Kruse O, Hellingwerf KJ (2013) Potential of industrial biotechnology with cyanobacteria and eukaryotic microalgae. Curr Opin Biotechnol 24:405–413. https://doi.org/10.1016/j.copbio.2013.04.004
Wolf J, Stephens E, Steinbusch S et al (2016) Multifactorial comparison of photobioreactor geometries in parallel microalgae cultivations. Algal Res 15:187–201. https://doi.org/10.1016/j.algal.2016.02.018
Yang B, Liu J, Jiang Y, Chen F (2016) Chlorella species as hosts for genetic engineering and expression of heterologous proteins: progress, challenge and perspective. Biotechnol J 11:1244–1261. https://doi.org/10.1002/biot.201500617
Zedler JAZ, Gangl D, Hamberger B et al (2014) Stable expression of a bifunctional diterpene synthase in the chloroplast of Chlamydomonas reinhardtii. J Appl Phycol. https://doi.org/10.1007/s10811-014-0504-2
Zerbe P, Hamberger B, Yuen MMS et al (2013) Gene discovery of modular diterpene metabolism in nonmodel systems. Plant Physiol 162:1073–1091. https://doi.org/10.1104/pp.113.218347
Acknowledgements
This work has been supported by the technology platform and infrastructure at the Center for Biotechnology (CeBiTec) of Bielefeld University. Sincere thanks to Dr. Thomas Baier for critical reading of this manuscript and those mentioned in the text who provided pictures.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no conflict of interest.
Rights and permissions
About this article
Cite this article
Lauersen, K.J. Eukaryotic microalgae as hosts for light-driven heterologous isoprenoid production. Planta 249, 155–180 (2019). https://doi.org/10.1007/s00425-018-3048-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-018-3048-x