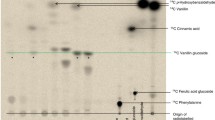
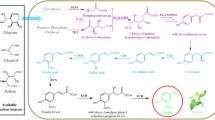
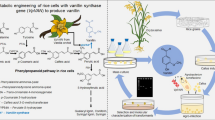
Abstract
Main conclusion
The present review compiles the up-to-date knowledge on vanillin biosynthesis in plant systems to focus principally on the enzymatic reactions of in planta vanillin biosynthetic pathway and to find out its impact and prospect in future research in this field.
Vanillin, a very popular flavouring compound, is widely used throughout the world. The principal natural resource of vanillin is the cured vanilla pods. Due to the high demand of vanillin as a flavouring agent, it is necessary to explore its biosynthetic enzymes and genes, so that improvement in its commercial production can be achieved through metabolic engineering. In spite of significant advancement in elucidating vanillin biosynthetic pathway in the last two decades, no conclusive demonstration had been reported yet for plant system. Several biosynthetic enzymes have been worked upon but divergences in published reports, particularly in characterizing the crucial biochemical steps of vanillin biosynthesis, such as side-chain shortening, methylation, and glucoside formation and have created a space for discussion. Recently, published reviews on vanillin biosynthesis have focused mainly on the biotechnological approaches and bioconversion in microbial systems. This review, however, aims to compile in brief the overall vanillin biosynthetic route and present a comparative as well as comprehensive description of enzymes involved in the pathway in Vanilla planifolia and other plants. Special emphasis has been given on the key enzymatic biochemical reactions that have been investigated extensively. Finally, the present standpoint and future prospects have been highlighted.




Similar content being viewed by others
Abbreviations
- HBS:
-
Hydroxybenzaldehyde synthase
- HCHL:
-
4-Hydroxycinnamoyl-CoA hydratase/lyase
- COMT:
-
Caffeic acid O-methyltransferase
References
Achterholt S, Priefert H, Steinbüchel A (2000) Identification of Amycolatopsis sp. strain HR167 genes, involved in the bioconversionof ferulic acid to vanillin. Appl Microbiol Biotechnol 54:799–807
Ahmed MA, El-Mawla A, Beerhues L (2002) Benzoic acid biosynthesisin cell cultures of Hypericum androsaemum. Planta 214:727–733
Anwar MH (1963) Paper chromatography of monohydroxyphenols in vanilla extract. Anal Chem 35:1974–1976
Dignum MJW, Kerler J, Verpoorte R (2001) Vanilla production: technological, chemical, and biosynthetic aspects. Food Rev Int 17:199–219
French CJ, Vance CP, Towers GHN (1976) Conversion of p-coumaric acid to p-hydroxybenzoic acid by cell free extracts of potato tubers and Polyporushispidus. Phytochemistry 15:564–566
Fritz RR, Hodgins DS, Abell CW (1976) Phenylalanine ammonia-lyase. Induction and purification from yeast and clearance in mammals. J Biol Chem 251:4646–4650
Funk C, Brodelius PE (1990a) Phenylpropanoid metabolism in suspension cultures of Vanilla planifolia Andr. II Effects of precursor feeding and metabolic inhibitors. Plant Physiol 94:95–101
Funk C, Brodelius PE (1990b) Phenylpropanoid metabolism in suspension cultures of Vanilla planifoliaAndr. III Conversion of 4-methoxycinnamic acids into 4-hydroxybenzoic acids. Plant Physiol 94:102–108
Gallage NJ, Møller BL (2015) Vanillin–bioconversion and bioengineering of the most popular plant flavor and its de novo biosynthesis in the vanilla orchid. Mol Plant 8:40–57
Gallage NJ, Hansen EH, Kannangara R, Olsen CE, Motawia MS, Jørgensen K, Holme I, Hebelstrup K, Grisoni M, Møller BL (2014) Vanillin formation from ferulic acid in Vanilla planifolia is catalysed by a single enzyme. Nat Commun 5:4037. doi:10.1038/ncomms5037
Gasson MJ, Kitamura Y, McLauchlan WR, Narbad A, Parr AJ, Parsons ELH, Payne J, Rhodes MJC, Walton NJ (1998) Metabolism of ferulic acid to vanillin: a bacterial gene of the enoyl-SCoA hydratase/isomerase superfamily encodes an enzyme for the hydration and cleavage of a hydroxycinnamic acid SCoA thioester. J Biol Chem 273:4163–4170
Havkin-Frenkel D, Podstolski A, Dixon R (2003) Vanillin biosynthetic pathway enzyme from Vanilla planifolia. United States patent application publication. https://docs.google.com/viewer?url=patentimages.storage.googleapis.com/pdfs/US20030070188.pdf
Ibrahim RK, Bruneau A, Bantignies B (1998) Plant O-methyltransferases: molecular analysis, common signature and classification. Plant Mol Biol 36:1–10
Jarvis AP, Schaaf O, Oldham NJ (2000) 3-Hydroxy-3-phenylpropanoic acid is an intermediate in the biosynthesis of benzoic acidand salicylic acid but benzaldehyde is not. Planta 212:119–126
Kanisawa T, Tokoro K, Kawahara S (1994) In: Kurihara K, Suzuki N, Ogawa H (eds) Olfaction taste XI (Proceeding of the International Symposium). Springer, Tokyo, p 268
Kaur B, Chakraborty D (2013) Biotechnological and molecular approaches for vanillin production: a review. Appl Biochem Biotechnol 169(4):1353–1372
Kundu A, Jawali N, Mitra A (2012) Shikimate pathway modulates the elicitor-stimulated accumulation of fragrant 2-hydroxy-4-methoxybenzaldehyde in Hemidesmusindicusroots. Plant PhysiolBiochem 56:104–108
Lam KC, Ibrahim RK, Behdad B, Dayanandan S (2007) Structure, function, and evolution of plant O-methyltransferases. Genome 50:1001–1013
Li L, Popko JL, Umezawa T, Chiang VL (2000) 5-Hydroxyconiferylaldehyde modulates enzymatic methylation for syringyl monolignol formation, a new view of monolignol biosynthesis in angiosperms. J Biol Chem 275:6537–6545
Löscher R, Heide L (1994) Biosynthesis of p-hydroxybenzoate fromp-coumarate and p-coumaroyl-coenzyme A in cell-free extracts of Lithospermum erythrorhizon cell cultures. Plant Physiol 106:271–279
Makkar HPS, Beeker K (1994) Isolation of tannins from leaves of some trees and shrubs and their properties. J Agric Food Chem 42:731–734
Mitra A, Kitamura Y, Gasson MJ, Narbad A, Parr AJ, Payne J, Rhodes MJC, Sewter C, Walton NJ (1999) 4-Hydroxycinnamoyl-CoA hydratase/lyase (HCHL)-an enzyme of phenylpropanoid chain cleavage from Pseudomonas. Arch Biochem Biophys 365:10–16
Nair RB, Bastress KL, Ruegger MO, Denault JW, Chapple C (2004) The Arabidopsis thaliana REDUCED EPIDERMAL FLUORESCENCE1 gene encodes an aldehyde dehydrogenase involved in ferulic acid and sinapic acid biosynthesis. Plant Cell 16:544–554
Narbad A, Gasson MJ (1998) Metabolism of ferulic acid via vanillinusing a novel CoA-dependent pathway in a newly-isolated strain of Pseudomonas fluorescens. Microbiology 144:1397–1404
Negishi O, Sugiura K, Negishi Y (2009) Biosynthesis of vanillin via ferulic acid in Vanilla planifolia. J Agric Food Chem 57:9956–9961
Ni J, Tao F, Du H, Xu P (2015) Mimicking a natural pathwayfor de novo biosynthesis: natural vanillin production from accessible carbon sources. Sci Rep. doi:10.1038/srep13670
Osakabe K, Tsao CC, Li L, Popko JL, Umezawa T, Carraway DT (1999) Coniferyl aldehyde 5-hydroxylation and methylation direct syringyl lignin biosynthesis in angiosperms. Proc Natl Acad Sci USA 96:8955–8960
Overhage J, Priefert H, Steinbüchel A (1999) Biochemical andgenetic analyses of ferulic acid catabolism in Pseudomonas sp. Strain HR199. Appl Environ Microbiol 65:4837–4847
Pak FE, Gropper S, Dai WD, Havkin-Frenkel D, Belanger FC (2004) Characterization of a multifunctional methyltransferases from the orchid Vanilla planifolia. Plant Cell Rep 22:959–966
Parvathi K, Chen F, Guo D, Blount JW, Dixon RA (2001) Substrate preferences of O-methyltransferases in alfalfa suggest new pathways for 3-O-methylation of monolignols. Plant J 25:193–202
Podstolski A, Havkin-Frenkel D, Malinowski J, Blount JW, Kourteva G, Dixon RA (2002) Unusual 4-hydroxybenzaldehyde synthase activity from tissue cultures of the vanilla orchid Vanilla planifolia. Phytochemistry 61:611–620
Priefert H, Rabenhorst J, Steinbüchel A (1997) Molecular characterization of genes of Pseudomonas sp. strain HR199 involved inbioconversion of vanillin to protocatechuate. J Bacteriol 179:2595–2607
Ro DK, Mah N, Ellis BE, Douglas CJ (2001) Functional characterization and subcellular localization of poplar (Populustrichocarpa × Populusdeltoides) cinnamate 4-hydroxylase. Plant Physiol 126:317–329
Schnitzler J-P, Madlung J, Rose A, Seitz HU (1992) Biosynthesis of p-hydroxybenzoic acid in elicitor-treated carrot cell cultures. Planta 188:594–600
Schoch G, Goepfert S, Morant M, Hehn A, Meyer D, Ullmann P, Werck-Reichhart D (2001) CYP98A3 from Arabidopsis thaliana is a 30-hydroxylase of phenolic esters, a missing link in the phenylpropanoid pathway. J Biol Chem 276:36566–36574
Sinha AK, Sharma UK, Sharma N (2008) A comprehensive review on vanilla flavor: extraction, isolation and quantification of vanillin and others constituents. Int J Food Sci Nutr 59(4):299–326
Sircar D, Mitra A (2008) Evidence for p-hydroxybenzoate formation involving phenylpropanoid chain-cleavage in hairy roots of Daucus carota. J Plant Physiol 165:407–414
Tokoro K, Kawahara S, Amano A, Kanisawa T, Indo M (1990). In: Bessiere Y, Thomas AF (eds) Flavour science and technology, vol 73. Wiley, Chichester, p 73
Venturi V, Zennaro F, Degrassi G, Okeke BC, Bruschi CV (1998) Genetics of ferulic acid bioconversion to protocatechuic acidin plant-growth-promoting Pseudomonas putida WCS358. Microbiology 144:965–973
Walton NJ, Mayer MJ, Narbad A (2003) Vanillin. Phytochemistry 63:505–515
Yazaki K, Heide L, Tabata M (1991) Formation of p-hydroxybenzoic acid from p-coumaric acid by cell free extract of Lithospermum erythrorhizon cell cultures. Phytochemistry 30:2233–2236
Zamzuri NA, Abd-Aziz S (2012) Biovanillin from agro wastes as an alternative food flavour. J Sci Food Agric 93:429–438
Zenk MH (1965) Biosynthese von vanillin in Vanilla planifolia. Andr Z Pflanzenphysiol 53:404
Acknowledgements
The author acknowledges the Indian Institute of Technology Kharagpur for permitting to access the digital library and for providing ‘Institutional Assistantship’ during preparing the manuscript. Author also acknowledges Ms. Shruti Mishra, M.Sc. for helping in linguistic editing of the article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kundu, A. Vanillin biosynthetic pathways in plants. Planta 245, 1069–1078 (2017). https://doi.org/10.1007/s00425-017-2684-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-017-2684-x