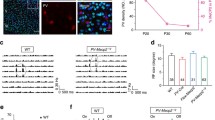
Abstract
We previously demonstrated that the deletion of phospholipase C-related catalytically inactive protein-1/2 (PRIP-1/2) enhances the desensitization of GABAA receptors (GABAARs), while it facilitates their resensitization at the offset of GABA puff, causing a hump-like tail current (tail-I) in layer 3 (L3) pyramidal cells (PCs) of the barrel cortex. In the present study, we investigated whether inhibitory synaptic transmission in L3 PCs in the barrel cortex is altered in the PRIP-1/2 double-knockout (PRIP-DKO) mice, and if so, how the interaction between excitation and inhibition is subsequently modified. PRIP-1/2 deletion resulted in the prolongation of the decay phase of inhibitory postsynaptic currents/potentials (IPSCs/IPSPs) in L3 PCs evoked by stimulation of L3, leaving the overall features of miniature IPSCs unchanged. An optical imaging revealed that the spatiotemporal profile of a horizontal excitation spread across columns in L2/3 caused by L4 stimulation in the barrel cortex was more restricted in PRIP-DKO mice compared to the wild type, while those obtained in the presence of bicuculline were almost identical between the two genotypes. These findings suggest that PRIP-1/2 deletion enhances the lateral inhibition by prolonging inhibitory synaptic actions to limit the intercolumnar integration in the barrel cortex. Considering the present findings together with our previous study including a mathematical simulation, the prolongation of inhibitory synaptic actions is likely to result from an enhancement of desensitization followed by an enhanced resensitization in GABAARs.






Similar content being viewed by others
References
Anstrom KK, Schallert T, Woodlee MT, Shattuck A, Roberts DC (2007) Repetitive vibrissae-elicited forelimb placing before and immediately after unilateral 6-hydroxydopamine improves outcome in a model of Parkinson’s disease. Behav Brain Res 179:183–191. doi:10.1016/j.bbr.2007.01.028, PMID: 17374405
Armstrong-James M, Fox K, Das-Gupta A (1992) Flow of excitation within rat barrel cortex on striking a single vibrissa. J Neurophysiol 68:1345–1358, PMID: 1432088
Dykes RW, Landry P, Metherate R, Hicks TP (1984) Functional role of GABA in cat primary somatosensory cortex: shaping receptive fields of cortical neurons. J Neurophysiol 52:1066–1093, PMID: 6151590
Foeller E, Celikel T, Feldman DE (2005) Inhibitory sharpening of receptive fields contributes to whisker map plasticity in rat somatosensory cortex. J Neurophysiol 94:4387–4400. doi:10.1152/jn.00553.2005, PMID: 16162832
Fox K, Wright N, Wallace H, Glazewski S (2003) The origin of cortical surround receptive fields studied in the barrel cortex. J Neurosci 23:8380–8391, PMID: 12968000
Grinvald A, Anglister L, Freeman JA, Hildesheim R, Manker A (1984) Real-time optical imaging of naturally evoked electrical activity in intact frog brain. Nature 308:848–850, PMID: 6717577
Kanematsu T, Jang IS, Yamaguchi T, Nagahama H, Yoshimura K, Hidaka K, Matsuda M, Takeuchi H, Misumi Y, Nakayama K, Yamamoto T, Akaike N, Hirata M, Nakayama K (2002) Role of the PLC-related, catalytically inactive protein p130 in GABAA receptor function. EMBO J 21:1004–1011. doi:10.1093/emboj/21.5.1004, PMID: 11867528
Keros S, Hablitz JJ (2005) Subtype-specific GABA transporter antagonists synergistically modulate phasic and tonic GABAA conductances in rat neocortex. J Neurophysiol 94:2073–2085, PMID: 15987761
Kitayama T, Morita K, Sultana R, Kikushige N, Mgita K, Ueno S, Hirata M, Kanematsu T (2013) Phospholipase C-related but catalytically inactive protein modulates pain behavior in a neuropathic pain model in mice. Mol Pain 9:23. doi:10.1186/1744-8069-9-23, PMID: 23639135
Kyriazi HT, Carvell GE, Brumberg JC, Simons DJ (1996) Quantitative effects of GABA and bicuculline methiodide on receptive field properties of neurons in real and simulated whisker barrels. J Neurophysiol 75:547–560, PMID: 8714634
Laaris N, Carlson GC, Keller A (2000) Thalamic-evoked synaptic interactions in barrel cortex revealed by optical imaging. J Neurosci 20:1529–1537, PMID: 10662842
Laaris N, Keller A (2002) Functional independence of layer IV barrels. J Neurophysiol 87:1028–1034, PMID: 11826066
Mizokami A, Kanematsu T, Ishibashi H, Yamaguchi T, Tanida I, Takenaka K, Nakayama KI, Fukami K, Takenawa T, Kominami E, Moss SJ, Yamamoto T, Nabekura J, Hirata M (2007) Phospholipase C-related inactive protein is involved in trafficking of γ2 subunit-containing GABAA receptors to the cell surface. J Neurosci 27:1692–1701. doi:10.1523/JNEUROSCI.3155-06.2007, PMID: 17301177
Mizokami A, Tanaka H, Ishibashi H, Umebayashi H, Fukami K, Takenawa T, Nakayama KI, Yokoyama T, Nabekura J, Kanematsu T, Hirata M (2010) GABAA receptor subunit alteration-dependent diazepam insensitivity in the cerebellum of phospholipase C-related inactive protein knockout mice. J Neurochem 114:302–310. doi:10.1111/j.1471-4159.2010.06754.x, PMID: 20412381
Nusser Z, Cull-Candy S, Farrant M (1997) Differences in synaptic GABAA receptor number underlie variation in GABA mini amplitude. Neuron 19:697–709. doi:10.1016/S0896-6273(00)80382-7, PMID: 9331359
Overstreet LS, Westbrook GL (2003) Synapse density regulates independence at unitary inhibitory synapses. J Neurosci 23:2618–2626, PMID: 12684447
Petersen CC, Sakmann B (2001) Functionally independent columns of rat somatosensory barrel cortex revealed with voltage-sensitive dye imaging. J Neurosci 21:8435–8446, PMID: 11606632
Sato H, Shimanuki Y, Saito M, Toyoda H, Nokubi T, Maeda Y, Yamamoto T, Kang Y (2008) Differential columnar processing in local circuits of barrel and insular cortices. J Neurosci 28:3076–3089. doi:10.1523/JNEUROSCI.0172-08.2008, PMID: 18354011
Sigworth FJ (1980) The variance of sodium current fluctuations at the node of Ranvier. J Physiol 307:97–129, PMID: 6259340
Thompson SM, Capogna M, Scanziani M (1993) Presynaptic inhibition in the hippocampus. Trends Neurosci 16:222–227, PMID: 7688163
Toyoda H, Saito M, Sato H, Tanaka T, Ogawa T, Yatani H, Kanematsu T, Hirata M, Kang Y (2014) Deletion of phospholipase C-related inactive protein-1/2 enhances desensitization and resensitization of GABAA receptors in pyramidal cells of the barrel cortex. Pflugers Arch
Traynelis SF, Silver RA, Cull-Candy SG (1993) Estimated conductance of glutamate receptor channels activated during EPSCs at the cerebellar mossy fiber-granule cell synapse. Neuron 11:279–289. doi:10.1016/0896-6273(93)90184-S, PMID: 7688973
Wu X, Wu Z, Ning G, Guo Y, Ali R, Macdonald RL, De Blas AL, Luscher B, Chen G (2012) γ-Aminobutyric acid type A (GABAA) receptor α subunits play a direct role in synaptic versus extrasynaptic targeting. J Biol Chem 287:27417–27430. doi:10.1074/jbc.M112.360461, PMID: 22711532
Zarnowska ED, Keist R, Rudolph U, Pearce RA (2009) GABAA receptor α5 subunits contribute to GABAA, slow synaptic inhibition in mouse hippocampus. J Neurophysiol 101:1179–1191. doi:10.1152/jn.91203.2008, PMID: 19073796
Acknowledgments
This study was supported by a Grant-in-Aid for Scientific Research to Y.K. (B; 26290006) and by the Japan Ministry of Education, Culture, Sports, Science and Technology to M.H. (S; 24229009).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 419 kb)
Rights and permissions
About this article
Cite this article
Toyoda, H., Saito, M., Sato, H. et al. Enhanced lateral inhibition in the barrel cortex by deletion of phospholipase C-related catalytically inactive protein-1/2 in mice. Pflugers Arch - Eur J Physiol 467, 1445–1456 (2015). https://doi.org/10.1007/s00424-014-1592-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-014-1592-1