Abstract
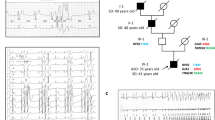
Atrial fibrillation (AF) is the most common arrhythmia. Gain-of-function mutations in KCNQ1, the pore-forming α-subunit of the slow delayed rectifier K current (I Ks) channel, have been associated with AF. The purpose of this study was functional assessment of a mutation in KCNQ1 identified in a family with persistent AF and sinus bradycardia. We investigated whether this KCNQ1 missense mutation could form the genetic basis for AF and bradycardia simultaneously in this family. Sanger sequencing in a family with hereditary persistent AF identified a novel KCNQ1 variant (V241F) in a highly conserved region of S4 domain. The proband and her son developed bradycardia and persistent AF in an age-dependent fashion. The other son was a mutation carrier but he showed sinus bradycardia and not AF. Whole-cell patch clamp electrophysiology showed that V241F mutation in KCNQ1 shifted the activation curve to the left and dramatically slowed deactivation, leading to a constitutively open-like phenotype. Computer modeling showed that V241F would slow pacemaker activity. Also, simulations of atrial excitation predicted that V241F results in extreme shortening of action potential duration, possibly resulting in AF. Our study indicates that V241F might cause sinus bradycardia by increasing I Ks. Additionally, V241F likely shortens atrial refractoriness to promote a substrate for reentry. KCNQ1 mutations have previously been described in AF, yet this is the first time a mutation in KCNQ1 is associated with age-dependent bradycardia and persistent AF. This finding further supports the hypothesis that sinus node dysfunction contributes to the development of AF.





Similar content being viewed by others
References
Andalib A, Brugada R, Nattel S (2008) Atrial fibrillation: evidence for genetically determined disease. Curr Opin Cardiol 23(3):176–183. doi:10.1097/HCO.0b013e3282fa7142
Barhanin J, Lesage F, Guillemare E, Fink M, Lazdunski M, Romey G (1996) K(V)LQT1 and lsK (minK) proteins associate to form the I(Ks) cardiac potassium current. Nature 384(6604):78–80. doi:10.1038/384078a0
Bartos DC, Anderson JB, Bastiaenen R, Johnson JN, Gollob MH, Tester DJ, Burgess DE, Homfray T, Behr ER, Ackerman MJ, Guicheney P, Delisle BP (2013) A KCNQ1 mutation causes a high penetrance for familial atrial fibrillation. J Cardiovasc Electrophysiol 24 (5):562–569. doi:10.1111/jce.12068 [doi]
Bartos DC, Duchatelet S, Burgess DE, Klug D, Denjoy I, Peat R, Lupoglazoff JM, Fressart V, Berthet M, Ackerman MJ, January CT, Guicheney P, Delisle BP (2011) R231C mutation in KCNQ1 causes long QT syndrome type 1 and familial atrial fibrillation. Heart Rhythm 8(1):48–55. doi:10.1016/j.hrthm.2010.09.010
Chan PJ, Osteen JD, Xiong D, Bohnen MS, Doshi D, Sampson KJ, Marx SO, Karlin A, Kass RS (2012) Characterization of KCNQ1 atrial fibrillation mutations reveals distinct dependence on KCNE1. J Gen Physiol 139 (2):135–144. doi:jgp.201110672 [pii] 10.1085/jgp.201110672 [doi]
Chen YH, Xu SJ, Bendahhou S, Wang XL, Wang Y, Xu WY, Jin HW, Sun H, Su XY, Zhuang QN, Yang YQ, Li YB, Liu Y, Xu HJ, Li XF, Ma N, Mou CP, Chen Z, Barhanin J, Huang W (2003) KCNQ1 gain-of-function mutation in familial atrial fibrillation. Science 299(5604):251–254. doi:10.1126/science.1077771
Das S, Makino S, Melman YF, Shea MA, Goyal SB, Rosenzweig A, Macrae CA, Ellinor PT (2009) Mutation in the S3 segment of KCNQ1 results in familial lone atrial fibrillation. Heart Rhythm 6(8):1146–1153. doi:10.1016/j.hrthm.2009.04.015
Demolombe S, Lande G, Charpentier F, van Roon MA, van den Hoff MJ, Toumaniantz G, Baro I, Guihard G, Le Berre N, Corbier A, de Bakker J, Opthof T, Wilde A, Moorman AF, Escande D (2001) Transgenic mice overexpressing human KvLQT1 dominant-negative isoform. Part I: Phenotypic characterisation. Cardiovasc Res 50 (2):314–327. doi:S0008636301002310
Duhme N, Schweizer PA, Thomas D, Becker R, Schroter J, Barends TR, Schlichting I, Draguhn A, Bruehl C, Katus HA, Koenen M (2012) Altered HCN4 channel C-linker interaction is associated with familial tachycardia-bradycardia syndrome and atrial fibrillation. Eur Heart J. doi:10.1093/eurheartj/ehs391
Fedorov VV, Chang R, Glukhov AV, Kostecki G, Janks D, Schuessler RB, Efimov IR (2010) Complex interactions between the sinoatrial node and atrium during reentrant arrhythmias in the canine heart. Circulation 122(8):782–789. doi:10.1161/CIRCULATIONAHA.109.935288
Henrion U, Zumhagen S, Steinke K, Strutz-Seebohm N, Stallmeyer B, Lang F, Schulze-Bahr E, Seebohm G (2012) Overlapping cardiac phenotype associated with a familial mutation in the voltage sensor of the KCNQ1 channel. Cell Physiol Biochem 29(5–6):809–818. doi:10.1159/000178470
Hong K, Piper DR, Diaz-Valdecantos A, Brugada J, Oliva A, Burashnikov E, Santos-de-Soto J, Grueso-Montero J, Diaz-Enfante E, Brugada P, Sachse F, Sanguinetti MC, Brugada R (2005) De novo KCNQ1 mutation responsible for atrial fibrillation and short QT syndrome in utero. Cardiovasc Res 68(3):433–440. doi:10.1016/j.cardiores.2005.06.023
Joung B, Shinohara T, Zhang H, Kim D, Choi EK, On YK, Piccirillo G, Chen PS, Lin SF (2010) Tachybradycardia in the isolated canine right atrium induced by chronic sympathetic stimulation and pacemaker current inhibition. Am J Physiol Heart Circ Physiol 299 (3):H634-642. doi: 10.1152/ajpheart.00347.2010
Kharche S, Adeniran I, Stott J, Law P, Boyett MR, Hancox JC, Zhang H (2012) Pro-arrhythmogenic effects of the S140G KCNQ1 mutation in human atrial fibrillation—insights from modelling. J Physiol 590 (Pt 18):4501–4514. doi: 10.1113/jphysiol.2012.229146
Kurata Y, Hisatome I, Imanishi S, Shibamoto T (2002) Dynamical description of sinoatrial node pacemaking: improved mathematical model for primary pacemaker cell. Am J Physiol Heart Circ Physiol 283(5):H2074–H2101. doi:10.1152/ajpheart.00900.2001
Lee SY, Choi HK, Kim ST, Chung S, Park MK, Cho JH, Ho WK, Cho H (2010) Cholesterol inhibits M-type K+ channels via protein kinase C-dependent phosphorylation in sympathetic neurons. J Biol Chem 285(14):10939–10950. doi:10.1074/jbc.M109.048868
Lundby A, Ravn LS, Svendsen JH, Olesen SP, Schmitt N (2007) KCNQ1 mutation Q147R is associated with atrial fibrillation and prolonged QT interval. Heart Rhythm 4(12):1532–1541. doi:10.1016/j.hrthm.2007.07.022
Mahida S, Lubitz SA, Rienstra M, Milan DJ, Ellinor PT (2011) Monogenic atrial fibrillation as pathophysiological paradigms. Cardiovasc Res 89(4):692–700. doi:10.1093/cvr/cvq381
Newton-Cheh C, Eijgelsheim M, Rice KM, de Bakker PI, Yin X, Estrada K, Bis JC, Marciante K, Rivadeneira F, Noseworthy PA, Sotoodehnia N, Smith NL, Rotter JI, Kors JA, Witteman JC, Hofman A, Heckbert SR, O'Donnell CJ, Uitterlinden AG, Psaty BM, Lumley T, Larson MG, Stricker BH (2009) Common variants at ten loci influence QT interval duration in the QTGEN Study. Nat Gen 41 (4):399–406. doi: 10.1038/ng.364 [doi]
Nygren A, Fiset C, Firek L, Clark JW, Lindblad DS, Clark RB, Giles WR (1998) Mathematical model of an adult human atrial cell: the role of K+ currents in repolarization. Circ Res 82(1):63–81
Olesen MS, Bentzen BH, Nielsen JB, Steffensen AB, David JP, Jabbari J, Jensen HK, Haunso S, Svendsen JH, Schmitt N (2012) Mutations in the potassium channel subunit KCNE1 are associated with early-onset familial atrial fibrillation. BMC Med Genet 13:24. doi: 10.1186/1471-2350-13-24 [doi]
Olesen MS, Jespersen T, Nielsen JB, Liang B, Moller DV, Hedley P, Christiansen M, Varro A, Olesen SP, Haunso S, Schmitt N, Svendsen JH (2011) Mutations in sodium channel beta-subunit SCN3B are associated with early-onset lone atrial fibrillation. Cardiovasc Res 89(4):786–793. doi:10.1093/cvr/cvq348
Panaghie G, Abbott GW (2007) The role of S4 charges in voltage-dependent and voltage-independent KCNQ1 potassium channel complexes. J Gen Physiol 129(2):121–133. doi:10.1085/jgp.200609612
Peroz D, Rodriguez N, Choveau F, Baro I, Merot J, Loussouarn G (2008) Kv7.1 (KCNQ1) properties and channelopathies. J Physiol 586(7):1785–1789. doi:10.1113/jphysiol.2007.148254
Restier L, Cheng L, Sanguinetti MC (2008) Mechanisms by which atrial fibrillation-associated mutations in the S1 domain of KCNQ1 slow deactivation of IKs channels. J Physiol 586(Pt 17):4179–4191. doi:10.1113/jphysiol.2008.157511
Sanguinetti MC, Curran ME, Zou A, Shen J, Spector PS, Atkinson DL, Keating MT (1996) Coassembly of K(V)LQT1 and minK (IsK) proteins to form cardiac I(Ks) potassium channel. Nature 384(6604):80–83. doi:10.1038/384080a0
Tsai CT, Lai LP, Hwang JJ, Lin JL, Chiang FT (2008) Molecular genetics of atrial fibrillation. J Am Coll Cardiol 52(4):241–250. doi:10.1016/j.jacc.2008.02.072
Verkerk AO, van-Borren MM, Peters RJ, Broekhuis E, Lam KY, Coronel R, de-Bakker JM, Tan HL, Wilders R (2007) Single cells isolated from human sinoatrial node: action potentials and numerical reconstruction of pacemaker current. Conf Proc IEEE Eng Med Biol Soc 2007:904–907. doi:10.1109/IEMBS.2007.4352437
Verkerk AO, Wilders R, van-Borren MM, Peters RJ, Broekhuis E, Lam K, Coronel R, de-Bakker JM, Tan HL (2007) Pacemaker current (I(f)) in the human sinoatrial node. Eur Heart J 28(20):2472–2478. doi:10.1093/eurheartj/ehm339
Wijffels MC, Kirchhof CJ, Dorland R, Allessie MA (1995) Atrial fibrillation begets atrial fibrillation. A study in awake chronically instrumented goats. Circulation 92(7):1954–1968
Youm JB, Jo SH, Leem CH, Ho WK, Earm YE (2004) Role of stretch-activated channels in stretch-induced changes of electrical activity in rat atrial myocytes. Korean J Physiol Pharmacol 8(1):33–41
Acknowledgments
This work was supported by the Samsung Biomedical Research Institute grant (SBRI #C-B0-206-3) and by the National Research Foundation of Korea (grant nos.: NRF-2012046878 and NRF-2013025108), funded by the Ministry of Science and Technology, Republic of Korea.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Chang-Seok Ki and Chae Lim Jung contributed equally to this work.
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Ki, CS., Jung, C.L., Kim, Hj. et al. A KCNQ1 mutation causes age-dependant bradycardia and persistent atrial fibrillation. Pflugers Arch - Eur J Physiol 466, 529–540 (2014). https://doi.org/10.1007/s00424-013-1337-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-013-1337-6