Abstract
Purpose
The purpose of this study was to compare the feasibility and outcomes of two-stage hepatectomy in patients with or without accompanying digestive surgery.
Methods
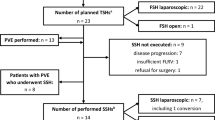
We analyzed prospectively data from 56 patients with colorectal liver metastases undergoing two-stage hepatectomy between 1995 and 2009. Patients undergoing associated digestive resection (group I, n = 32) were compared with patients without associated digestive surgery (group II, n = 17).
Results
The feasibility rate was 87.5 % (49 patients). Neither the type and extent of hepatectomy nor the type of chemotherapy administered differed between the two groups. The median interval between hepatectomies was 1.79 and 2.07 months for groups I and II, respectively (not significant). One patient (group I) died of liver failure after the second hepatectomy. Postoperative morbidity rates were comparable: 37.5 % (group I) vs. 35.5 % (group II) after the first hepatectomy and 46.9 % (group I) vs. 52.9 % (group II) after the second hepatectomy. The median hospital stay after the first hepatectomy was longer in group I (13.5 days) than in group II (10 days) (P < 0.01). Median follow-up was 54 months. The median overall survival (OS) was 45.8 months, and 3- and 5-year OS were 58 and 31 %, respectively. Median OS was longer for group II (58 months) than for group I (34 months) (P = 0.048).
Conclusions
Digestive tract resection associated with two-stage hepatectomy does not increase postoperative mortality or morbidity nor does it lead to delay in chemotherapy or a reduction in cycles administered. The need for digestive tract surgery should not affect the surgical management of two-stage hepatectomy patients.

Similar content being viewed by others
References
Wicherts DA, Miller R, de Haas RJ et al (2008) Long-term results of two-stage hepatectomy for irresectable colorectal cancer liver metastases. Ann Surg 248:994–1005
Simmonds PC, Primrose JN, Colquitt JL, Garden OJ, Poston GJ, Rees M (2006) Surgical resection of hepatic metastases from colorectal cancer: a systematic review of published studies. Br J Cancer 94:982–999
Chun YS, Vauthey JN, Ribero D et al (2007) Systemic chemotherapy and two-stage hepatectomy for extensive bilateral colorectal liver metastases: perioperative safety and survival. J Gastrointest Surg 11:1498–1504, discussion 504-5
Adam R, Laurent A, Azoulay D, Castaing D, Bismuth H (2000) Two-stage hepatectomy: a planned strategy to treat irresectable liver tumors. Ann Surg 232:777–785
Jaeck D, Oussoultzoglou E, Rosso E, Greget M, Weber JC, Bachellier P (2004) A two-stage hepatectomy procedure combined with portal vein embolization to achieve curative resection for initially unresectable multiple and bilobar colorectal liver metastases. Ann Surg 240:1037–1049, discussion 49-51
Hillingso JG, Wille-Jorgensen P (2009) Staged or simultaneous resection of synchronous liver metastases from colorectal cancer—a systematic review. Color Dis 11:3–10
Capussotti L, Ferrero A, Vigano L, Ribero D, Lo Tesoriere R, Polastri R (2007) Major liver resections synchronous with colorectal surgery. Ann Surg Oncol 14:195–201
Karoui M, Vigano L, Goyer P et al (2010) Combined first-stage hepatectomy and colorectal resection in a two-stage hepatectomy strategy for bilobar synchronous liver metastases. Br J Surg 97:1354–1362
Pang YY (2000) The Brisbane 2000 terminology of liver anatomy and resections. HPB 2:333–339, HPB (Oxford) 2002; 4:99
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Aydin HN, Remzi FH, Tekkis PP, Fazio VW (2005) Hartmann's reversal is associated with high postoperative adverse events. Dis Colon Rectum 48:2117–2126
Kaiser AM, Israelit S, Klaristenfeld D, Selvindoss P, Vukasin P, Ault G, Beart RW (2008) Morbidity of ostomy takedown. J Gastrointest Surg 12:437–441
Akiyoshi T, Fujimoto Y, Konishi T, Kuroyanagi H, Ueno M, Oya M, Yamaguchi T (2010) Complications of loop ileostomy closure in patients with rectal tumor. World J Surg 34:1937–1942
Tsim N, Healey AJ, Frampton AE et al (2011) Two-stage resection for bilobar colorectal liver metastases: R0 resection is the key. Ann Surg Oncol 18:1939–1946
Brouquet A, Abdalla EK, Kopetz S et al (2011) High survival rate after two-stage resection of advanced colorectal liver metastases: response-based selection and complete resection define outcome. J Clin Oncol 29:1083–1090
Martin RC 2nd, Augenstein V, Reuter NP, Scoggins CR, McMasters KM (2009) Simultaneous versus staged resection for synchronous colorectal cancer liver metastases. J Am Coll Surg 208:842–850, discussion 50-2
Martin R, Paty P, Fong Y et al (2003) Simultaneous liver and colorectal resections are safe for synchronous colorectal liver metastasis. J Am Coll Surg 197:233–241, discussion 41-2
Breukink S, Pierie J, Wiggers T (2006) Laparoscopic versus open total mesorectal excision for rectal cancer. Cochrane Database Syst Rev 4:CD005200
Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH (1999) Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg 230:309–318, discussion 18-21
Acknowledgments
We thank Thérèse Gargi for data management and Rob Stepney (medical writer, Charlbury, UK) for editing the manuscript.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stella, M., Dupre, A., Chabaud, S. et al. A comparative study of patients with and without associated digestive surgery in a two-stage hepatectomy setting. Langenbecks Arch Surg 397, 1289–1296 (2012). https://doi.org/10.1007/s00423-012-1002-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-012-1002-0