Abstract
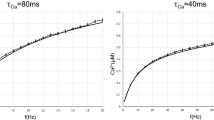
Short-term synaptic plasticity acts as a time- and firing rate-dependent filter that mediates the transmission of information across synapses. In the avian auditory brainstem, specific forms of plasticity are expressed at different terminals of the same auditory nerve fibers and contribute to the divergence of acoustic timing and intensity information. To identify key differences in the plasticity properties, we made patch-clamp recordings from neurons in the cochlear nucleus responsible for intensity coding, nucleus angularis, and measured the time course of the recovery of excitatory postsynaptic currents following short-term synaptic depression. These synaptic responses showed a very rapid recovery, following a bi-exponential time course with a fast time constant of ~40 ms and a dependence on the presynaptic activity levels, resulting in a crossing over of the recovery trajectories following high-rate versus low-rate stimulation trains. We also show that the recorded recovery in the intensity pathway differs from similar recordings in the timing pathway, specifically the cochlear nucleus magnocellularis, in two ways: (1) a fast recovery that was not due to recovery from postsynaptic receptor desensitization and (2) a recovery trajectory that was characterized by a non-monotonic bump that may be due in part to facilitation mechanisms more prevalent in the intensity pathway. We tested whether a previously proposed model of synaptic transmission based on vesicle depletion and sequential steps of vesicle replenishment could account for the recovery responses, and found it was insufficient, suggesting an activity-dependent feedback mechanism is present. We propose that the rapid recovery following depression allows improved coding of natural auditory signals that often consist of sound bursts separated by short gaps.
Similar content being viewed by others
References
Abbott LF, Regehr WG (2004) Synaptic computation. Nature 431: 796–803
Blitz DM, Foster KA, Regehr WG (2004) Short-term synaptic plasticity: a comparison of two synapses. Nat Rev Neurosci 5: 630–640
Boudreau CE, Ferster D (2005) Short-term depression in thalamocortical synapses of cat primary visual cortex. J Neurosci 25: 7179–7190
Brenowitz S, Trussell LO (2001a) Maturation of synaptic transmission at end-bulb synapses of the cochlear nucleus. J Neurosci 21: 9487–9498
Brenowitz S, Trussell LO (2001b) Minimizing synaptic depression by control of release probability. J Neurosci 21: 1857–1867
Brenowitz EA, Margoliash D, Nordeen KW (1997) An introduction to birdsong and the avian song system. J Neurobiol 33: 495–500
Cao XJ, McGinley MJ, Oertel D (2008) Connections and synaptic function in the posteroventral cochlear nucleus of deaf jerker mice. J Comp Neurol 510: 297–308
Carandini M, Heeger DJ, Senn W (2002) A synaptic explanation of suppression in visual cortex. J Neurosci 22: 10053–10065
Carr CE, Boudreau RE (1991) Central projections of auditory nerve fibers in the barn owl. J Comp Neurol 314: 306–318
Carver S, Roth E, Cowan NJ, Fortune ES (2008) Synaptic plasticity can produce and enhance direction selectivity. PLoS Comput Biol 4: e32
Chance FS, Nelson SB, Abbott LF (1998) Synaptic depression and the temporal response characteristics of V1 cells. J Neurosci 18: 4785–4799
Chang WP, Sudhof TC (2009) SV2 renders primed synaptic vesicles competent for Ca 2+-induced exocytosis. J Neurosci 29: 883–897
Chung S, Li X, Nelson SB (2002) Short-term depression at thalamocortical synapses contribute to rapid adaptation of cortical sensory responses in vivo. Neuron 34: 437–446
Cook DL, Schwindt PC, Grande LA, Spain WJ (2003) Synaptic depression in the localization of sound. Nature 421: 66–70
Dittman JS, Regehr WG (1998) Calcium dependence and recovery kinetics of presynaptic depression at the climbing fiber to Purkinje cell synapse. J Neurosci 18: 6147–6162
Dittman JS, Kreitzer AC, Regehr WG (2000) Interplay between facilitation, depression, and residual calcium at three presynaptic terminals. J Neurosci 20: 1374–1385
Doupe AJ, Kuhl PK (1999) Birdsong and human speech: common themes and mechanisms. Annu Rev Neurosci 22: 567–631
Fortune ES, Rose GJ (2000) Short-term synaptic plasticity contributes to the temporal filtering of electrosensory information. J Neurosci 20: 7122–7130
Fortune ES, Rose GJ (2002) Roles for short-term synaptic plasticity in behavior. J Physiol Paris 96: 539–545
Fuhrmann G, Segev I, Markram H, Tsodyks M (2002) Coding of temporal information by activity-dependent synapses. J Neurophysiol 87: 140–148
Griesinger CB, Richards CD, Ashmore JF (2005) Fast vesicle replenishment allows indefatigable signalling at the first auditory synapse. Nature 435: 212–215
Hosoi N, Sakaba T, Neher E (2007) Quantitative analysis of calcium-dependent vesicle recruitment and its functional role at the calyx of Held synapse. J Neurosci 27: 14286–14298
Kuba H, Koyano K, Ohmori H (2002) Synaptic depression improves coincidence detection in the nucleus laminaris in brainstem slices of the chick embryo. Eur J Neurosci 15: 984–990
Levin MD, Kubke MF, Schneider M, Wenthold R, Carr CE (1997) Localization of AMPA-selective glutamate receptors in the auditory brainstem of the barn owl. J Comp Neurol 378: 239–253
Lorteije JA, Rusu SI, Kushmerick C, Borst JG (2009) Reliability and precision of the mouse calyx of Held synapse. J Neurosci 29: 13770–13784
MacLeod KM, Carr CE (2005) Synaptic physiology in the cochlear nucleus angularis of the chick. J Neurophysiol 93: 2520–2529
MacLeod KM, Carr CE (2007) Beyond timing in the auditory brainstem: intensity coding in the avian cochlear nucleus angularis. Prog Brain Res 165: 123–133
MacLeod KM, Horiuchi TK, Carr CE (2007) A role for short-term synaptic facilitation and depression in the processing of intensity information in the auditory brain stem. J Neurophysiol 97: 2863–2874
Markram H, Gupta A, Uziel A, Wang Y, Tsodyks M (1998) Information processing with frequency-dependent synaptic connections. Neurobiol Learn Mem 70: 101–112
O’Donovan MJ, Rinzel J (1997) Synaptic depression: a dynamic regulator of synaptic communication with varied functional roles. Trends Neurosci 20: 431–433
Oertel D (1999) The role of timing in the brain stem auditory nuclei of vertebrates. Annu Rev Physiol 61: 497–519
Otis TS, Wu YC, Trussell LO (1996) Delayed clearance of transmitter and the role of glutamate transporters at synapses with multiple release sites. J Neurosci 16: 1634–1644
Raman IM, Zhang S, Trussell LO (1994) Pathway-specific variants of AMPA receptors and their contribution to neuronal signaling. J Neurosci 14: 4998–5010
Renden R, Taschenberger H, Puente N, Rusakov DA, Duvoisin R, Wang LY, Lehre KP, von Gersdorff H (2005) Glutamate transporter studies reveal the pruning of metabotropic glutamate receptors and absence of AMPA receptor desensitization at mature calyx of Held synapses. J Neurosci 25: 8482–8497
Rizzoli SO, Betz WJ (2004) The structural organization of the readily releasable pool of synaptic vesicles. Science 303: 2037–2039
Rizzoli SO, Betz WJ (2005) Synaptic vesicle pools. Nat Rev Neurosci 6: 57–69
Sakaba T, Neher E (2001) Calmodulin mediates rapid recruitment of fast-releasing synaptic vesicles at a calyx-type synapse. Neuron 32: 1119–1131
Saviane C, Silver RA (2006) Fast vesicle reloading and a large pool sustain high bandwidth transmission at a central synapse. Nature 439: 983–987
Schneggenburger R, Sakaba T, Neher E (2002) Vesicle pools and short-term synaptic depression: lessons from a large synapse. Trends Neurosci 25: 206–212
Stevens CF, Wesseling JF (1998) Activity-dependent modulation of the rate at which synaptic vesicles become available to undergo exocytosis. Neuron 21: 415–424
Stuart GJ, Doft HU, Sakmann B (1993) Patch-clamp recordings from the soma and dendrites of neurons in brain slices using infrared video microscopy. Pflugers Arch 423: 511–518
Sudhof TC (2004) The synaptic vesicle cycle. Annu Rev Neurosci 27: 509–547
Sugden SG, Zirpel L, Dietrich CJ, Parks TN (2002) Development of the specialized AMPA receptors of auditory neurons. J Neurobiol 52: 189–202
Trussell LO (1999) Synaptic mechanisms for coding timing in auditory neurons. Annu Rev Physiol 61: 477–496
Trussell LO (2002) Modulation of transmitter release at giant synapses of the auditory system. Curr Opin Neurobiol 12: 400–404
Usrey WM, Alonso JM, Reid RC (2000) Synaptic interactions between thalamic inputs to simple cells in cat visual cortex. J Neurosci 20: 5461–5467
von Gersdorff H, Borst JG (2002) Short-term plasticity at the calyx of held. Nat Rev Neurosci 3: 53–64
Wadel K, Neher E, Sakaba T (2007) The coupling between synaptic vesicles and Ca 2+ channels determines fast neurotransmitter release. Neuron 53: 563–575
Wang L-Y, Kaczmarek LK (1998) High-frequency firing helps replenish the readily releasable pool of synaptic vesicles. Nature 394: 384–388
Wang Y, Manis PB (2008) Short-term synaptic depression and recovery at the mature mammalian endbulb of Held synapse in mice. J Neurophysiol 100: 1255–1264
Wong AY, Graham BP, Billups B, Forsythe ID (2003) Distinguishing between presynaptic and postsynaptic mechanisms of short-term depression during action potential trains. J Neurosci 23: 4868–4877
Wu LG, Ryan TA, Lagnado L (2007) Modes of vesicle retrieval at ribbon synapses, calyx-type synapses, and small central synapses. J Neurosci 27: 11793–11802
Wu XS, McNeil BD, Xu J, Fan J, Xue L, Melicoff E, Adachi R, Bai L, Wu LG (2009) Ca(2+) and calmodulin initiate all forms of endocytosis during depolarization at a nerve terminal. Nat Neurosci 12: 1003–1010
Xu-Friedman MA, Regehr WG (2000) Probing fundamental aspects of synaptic transmission with strontium. J Neurosci 20: 4414–4422
Yang H, Xu-Friedman MA (2008) Relative roles of different mechanisms of depression at the mouse endbulb of Held. J Neurophysiol 99: 2510–2521
Young ED, Sachs MB (2008) Auditory nerve inputs to cochlear nucleus neurons studied with cross-correlation. Neuroscience 154: 127–138
Zucker RS (1999) Calcium- and activity-dependent synaptic plasticity. Curr Opin Neurobiol 9: 305–313
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
The Below is the Electronic Supplementary Material.
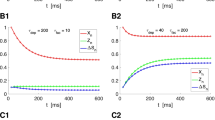
Fig. S1 EGTA slows the recovery but fails to eliminate cross over a, Recovery following 50 Hz trains under control and two EGTA incubation concentrations. Steady state levels are plotted using corresponding markers on the far left. b, Recovery following 200 Hz trains under control and two EGTA concentrations. c, Comparison of recovery following 50 Hz and 200 Hz trains under 0.1 mM EGTA showing the cross over. d, e Recovery curves from a and b replotted as fractional recovery to normalize for differences in steady state depression. Note the similarity of the 0.02 mM EGTA curve with control, and the flatness of the 50 Hz 0.1mM EGTA curve, suggesting that EGTA affected the recovery rate. Data are mean ± s.e.m. Control, n = 5; 0.02 mM EGTA-AM, n = 3; 0.1mM EGTA-AM, n = 6.
Rights and permissions
About this article
Cite this article
MacLeod, K.M., Horiuchi, T.K. A rapid form of activity-dependent recovery from short-term synaptic depression in the intensity pathway of the auditory brainstem. Biol Cybern 104, 209–223 (2011). https://doi.org/10.1007/s00422-011-0428-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00422-011-0428-8