Abstract
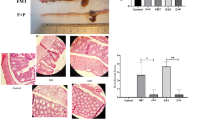
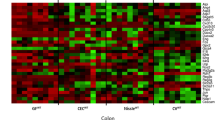
Glutathione transferases (GSTs) play a central role in the cellular defense against harmful endogenous compounds and xenobiotics in mouse and man. The gastrointestinal channel is constantly exposed to bacteria, bacterial products, and xenobiotics. In the present study the distribution of alpha, mu, and pi class GSTs was examined immunohistologically in the colon of conventional and germ-free (GF) mice subjected to experimental colitis. The tissues samples were from conventional mice with and without colitis induced by dextran sulfate sodium (DSS); GF mice treated with DSS or carrageenan; and GF mice inoculated with normal mouse bacterial flora as well as with Lactobacillus GG. In conventional as well as in GF mice the mu and pi class GSTs showed reduced intestinal expression when colitis was induced. In contrast, the level of GSTs reacting with antibodies directed against the alpha class, in particular mGST A4-4, was elevated after induction of inflammation. Of special interest is mGST A4-4 because of its high catalytic activity with toxic products of lipid peroxidation. In the colon of conventionalized GF mice that were given mouse intestinal flora, the mGST A4-4 expression was increased with time for several weeks, but then showed a decrease to a normal level. Additionally, the inoculation of GF mice with Lactobacillus GG induced all the intestinal GSTs studied.


Similar content being viewed by others
References
Alder V, Yin Z, Fuchs SY, Benezra M, Rosario L, Tew KD, Pincus MR, Sardana M, Henderson CJ, Wolf CR, Davis RJ, Ronai Z (1999) Regulation of JNK signaling by GSTp. EMBO J 18:1321–1334
Axelsson L-G (1996) Experimental animal models for the study of inflammatory bowel diseases and gastrointestinal anti-inflammatory drugs. Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology 215, Uppsala, Sweden, pp 62 (ISBN 91-554-3781-4)
Axelsson L-G (1999) Xenobiotics, intestinal microflora and innate gastrointestinal immunity: what can clinical observations and experimental animal models tell us about environmentally related diseases. Microecol Ther 28:327–335
Axelsson L-G, Midtvedt T (1997) Sulfation and Mw-optimized carrageenan induces colon inflammation in germfree mice. Gastroenterology 112:A925
Axelsson L-G, Landström E, Goldschmidt TJ, Gronberg A, Bylund-Fellenius AC (1996a) Dextran sulfate sodium (DSS) induced experimental colitis in immunodeficient mice. Effects in CD4+-cell depleted, athymic and NK-cell depleted SCID mice. Inflamm Res 45:181–191
Axelsson L-G, Midtvedt T, Bylund-Fellenius A-C (1996b) The role of intestinal bacteria, bacterial translocation and endotoxin in dextran sodium sulfate-induced colitis in the mouse. Microb Ecol Health Dis 9:225–237
Axelsson L-G, Landström E, Bylund-Fellenius A-C (1998) Experimental colitis induced by dextran sulfate sodium in mice: beneficial effects of sulfasalazine and olsalazine. Aliment Pharmacol Ther 12:925–934
Berhane K, Widersten M, Engström A, Kozarich JW, Mannervik B (1994) Detoxication of base propenals and other alpha, beta-unsaturated aldehyde products of radical reactions and lipid peroxidation by human glutathione transferases. Proc Natl Acad Sci U S A 91:1480–1484
Board PG, Coggan M, Chelvanayagam G, Easteal S, Jermiin LS, Schulte GK, Danley DE, Hoth LR, Griffor MC, Kamath AV, Rosner MH, Chrunyk BA, Perregaux DE, Gabel CA, Geoghegan KF, Pandit J (2000) Identification, characterization, and crystal structure of the Omega class glutathione transferases. J Biol Chem 275:24798–24806
Bruin WCC de, Wagenmans MJM, Board PG, Peters WHM (1999) Expression of glutathione S-transferase theta class isoenzymes in human colorectal and gastric cancers. Carcinogenesis 20:1453–1457
Bruin WCC de, Wagenmans MJM, Peters WHM (2000) Expression of glutathione S-transferase alpha, P1-1 and T1-1 in the human gastrointestinal tract. Jpn J Cancer Res 9:310–316
Bylund-Fellenius A-C, Landström E, Axelsson L-G, et al (1994) Experimental colitis induced by dextran sulfate in normal and germfree mice. Microb Ecol Health Dis 7:207–215
Cheng JZ, Singhal SS, Saini M, Singhal J, Piper JT, Van Kuijk FJ, Zimniak P, Awasthi YC, Awasthi S (1999) Effects of mGST A4 transfection on 4-hydroxynonenal-mediated apoptosis and differentiation of K562 human erythroleukemia cells. Arch Biochem Biophys 372:29–36
Cho SG, Lee YH, Park HS, Ryoo K, Kang KW, Park J, Eom SJ, Kim MJ, Cang TS, Choi SY, Shim J, Kim Y, Dong MS, Lee MJ, Kim SG, Ichijo H, Choi EJ (2001) Glutathione S-transferase mu modulates the stress-activated signals by suppressing apoptosis signal-regulating kinase 1. J Biol Chem 26:12749–12755
Clapper ML, Adrian RH, Pfeiffer GR, Kido K, Everley L, Cooper HS, Murthy S (1999) Depletion of colonic detoxication enzyme activity in mice with dextran sulfate sodium-induced colitis. Aliment Pharmacol Ther 13:389–396
Cooper HS, Murthy SNS, Shah RS, Sedergran DJ (1993) Clinicopathologic study of dextran sulfate sodium experimental murine colitis. Lab Invest 69:238–249
Cooper HS, Murthy S, Kido K, Yoshitake H, Flanigan A (2000) Dysplasia and cancer in the dextran sulfate sodium mouse colitis model. Relevance to colitis-associated neoplasia in the human: a study of the histopathology, B-catenin and p53 expression and the role of inflammation. Carcinogenesis 21:757–768
Desmots F, Rissel M, Gilot D, Lagadic-Gossmann D, Morel F, Guguen-Guillouzo C, Guillouzo A, Loyer P (2002) Pro-inflammatory cytokines tumor necrosis factor alpha and interleukin-6 and survival factor epidermal growth factor positively regulate the murine GSTA4 enzyme in hepatocytes. J Biol Chem 277:17892–17900
Edalat M, Pettersson S, Persson MA, Mannervik B (2002) Probing biomolecular interactions of glutathione transferase M2-2 by using peptide phage display. Chembiochem 3:823–828
Elson CO, Sartor RB, Tennyson GS, Riddell RH (1995) Experimental models of inflammatory bowel disease. Gastroenterology 109:1344–1367
French N, Pettersson S (2000) Microbe-host interactions in the alimentary tract: the gateway to understanding inflammatory bowel disease. Gut 47:162–163
Grisham MB (1993) Animal models of inflammatory bowel disease. Curr Opin Gastroenterol 9:524–533
Gustafsson BE (1959) Lightweight stainless steel systems for rearing germfree animals. NY Acad Sci 78:17–28
Hao XY, Castro VM, Bergh J, Sundström B, Mannervik B (1994) Isoenzyme-specific quantitative immunoassays for cytosolic glutathione transferases and measurement of the enzymes in blood plasma from cancer patients and in tumor cell lines. Biochim Biophys Acta 1225:223–230
Hayes JD, McLellan LI (1999) Glutathione and glutathione-dependent enzymes represent a co-ordinately regulated defence against oxidative stress. Free Radic Res 1:273–300
Hirono I, Sumi Y, Kuhara K, Miyakawa M (1981) Effect of degraded carrageenan on the intestine in germfree rats. Toxicol Lett 8:207–212
Hooper LV, Gordon JI (2001) Commensal host-bacterial relationships in the gut. Science 292:1115–1118
Hubatsch I, Ridderström M, Mannervik B (1998) Human glutathione transferase A4-4: an alpha class enzyme with high catalytic efficiency in the conjugation of 4-hydroxynonenal and other genotoxic products of lipid peroxidation. Biochem J 330:175–179
Ishioka T, Kuwabara N, Oohasi Y, Wakabayashi K (1987) Induction of colorectal tumors in rats by sulfated polysaccharides. CRC Crit Rev Toxicol 17:215–244
Kimura J, Hayakari M, Kumano T, Nakano H, Satoh K, Tsuchida S (1998) Altered glutathione transferase levels in rat skin inflamed due to contact hypersensitivity: induction of the alpha-class subunit 1. Biochem J 335:605–610
Malin M, Suomalainen H, Saxelin M, Isolauri E (1996) Promotion of IgA immune response in patients with Crohn’s disease by oral bacteriotherapy with Lactobacillus GG. Ann Nutr Metab 40:137–145
Mannervik B (1985) The isoenzymes of glutathione transferase. Adv Enzymol Relat Areas Mol Biol 57:357–417
Mannervik B, Danielson UH (1988) Glutathione transferases: structure and catalytic activity. CRC Crit Rev Biochem 23:263–337
Mannervik B, Ålin P, Guthenberg C, Jensson H, Tahir MK, Warholm M, Jörnvall H (1985) Identification of three classes of cytosolic glutathione transferase common to several mammalian species: correlation between structural data and enzymatic properties. Proc Natl Acad Sci U S A 82:7202–7206
Marteau PR, de Vrese M, Cellier CJ, Schrezenmeir J (2001) Protection from gastrointestinal diseases with the use of probiotics. Am J Clin Nutr 73:430S–436S
Mattey DL, Hassell AB, Plant M, Dawes PT, Ollier WR, Jones PW, Fryer AA, Alldersea JE, Strange RC (1999) Association of polymorphism in glutathione S-transferase loci with susceptibility and outcome in rheumatoid arthritis: comparison with the shared epitope. Ann Rheum Dis 58:164–168
Morgenstern R, DePierre JW, Jörnvall H (1985) Microsomal glutathione transferase: primary structure. J Biol Chem 260:13976–13983
Nieto N, Torres MI, Fernández MI, Giron MD, Rios A, Suarez MD, Gil A (2000) Experimental ulcerative colitis impairs antioxidant defense system in rat intestine. Dig Dis Sci 45:1820–1827
Oestreicher P, Nielsen ST, Rainsford KD (1992) Sulphasalazine fails to prevent development of mucosal ulceration and 5-lipoxygenase activity in guinea-pigs with chronic inflammatory bowel disease induced by combined bacterial immunization and oral carrageenin. J Pharm Pharmacol 44:528–531
Okayasu I, Hatakeyama S, Yamada M, Ohkusa T, Inagaki Y, Nakaya R (1990) A novel method in the induction of reliable experimental acute and chronic ulcerative colitis in mice. Gastroenterology 98:694–702
Ranganathan S, Tew KD (1991) Immunohistochemical localization of glutathione S-transferases alpha, mu, and pi in normal tissue and carcinomas from human colon. Carcinogenesis 12:2383–2387
Rollinson S, Levene AP, Mensah FK, Roddam PL, Allan JM, Diss TC, Roman E, Jack A, MacLennan K, Dixon MF, Morgan GJ (2003) Gastric marginal zone lymphoma is associated with polymorphisms in genes involved in inflammatory response and antioxidative capacity. Blood 102:1007–1011
Schotte L, Steidler L, Vandekerckhove J, Remaut E (2000) Secretion of biologically active murine interleukin-10 by Lactococcus lactis. Enzyme Microb Technol 27:761–765
Spiik A-K, Ridderstad A, Axelsson L-G, Midtvedt T, Björk L, Pettersson S (2002) P65 antisense oligonucleotides treatment inhibits lymphocyte infiltration and CD14 expression in dextran sulfate induced colitis in the mouse. Int J Colorectal Dis 17:223–232
Steidler L, Hans W, Schotte L, Neirynck S, Obermeier F, Falk W, Fiers W, Remaut E (2000) Treatment of murine colitis by Lactococcus lactis secreting interleukin-10. Science 289:1352–1355
Uribe A, Alam M, Johansson O, Midtvedt T, Theodorsson E (1994) Microflora modulates endocrine cells in the gastrointestinal mucosa of the rat. Gastroenterology 107:1259–1269
Vuilleumier S, Pagni M (2002) The elusive roles of bacterial glutathione S-transferases: new lessons from genomes. Appl Microb Biotech 58:138–146
Weiner ML (1991) Toxicological properties of carrageenan. Agents Actions 32:46–51
WHO Technical Reports Series (1984) Toxicological evaluation of certain food additives and food contaminants. Twenty-eighth report on the joint FAO/WHO expert committee on food additives, Geneva, pp 21–33
Willis CM, Britton LE, Reiche L, Wilkinson JD (2001) Reduced levels of glutathione S-transferases in patch test reactions to dithranol and sodium lauryl sulphate as demonstrated by quantitative immunocytochemistry: evidence for oxidative stress in acute irritant contact dermatitis. Eur J Dermatol 11:99–104
Yamada M, Ohkusa T, Okayasu I (1992) Occurrence of dysplasia and adenocarcinoma after experimental chronic ulcerative colitis in hamster induced by dextran sulfate sodium. Gut 33:1521–1527
Yamada T, Sartor RB, Marshall S, Specian RD, Grisham MB (1993) Mucosal injury and inflammation in a model of chronic granulomatous colitis in rats. Gastroenterology 104:759–771
Yin ZL, Dahlstrom JE, Le Couteur DG, Board PG (2001) Immunohistochemistry of omega class glutathione S-transferase in human tissues. J Histochem Cytochem 49:983–987
Zimniak P, Singhal SS, Srivastava SK, Awasthi S, Sharma R, Hayden JB, Awasthi YC (1994) Estimation of genomic complexity, heterologous expression, and enzymatic characterization of mouse glutathione S-transferase mGST A4-4 (GST 5.7). J Biol Chem 269:992–1000
Acknowledgements
We thank Dr. Yogesh C. Awasthi, University of Texas Medical Branch, Galveston, TX, USA, for the antibody against mGST A4-4. We are also grateful to Ros-Mari Johansson, Sandra Stenberg, Eva Österlund, and Anna-Karin Persson for practical assistance in the experimental work. This investigation was supported by the Swedish Cancer Society.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Edalat, M., Mannervik, B. & Axelsson, LG. Selective expression of detoxifying glutathione transferases in mouse colon: effect of experimental colitis and the presence of bacteria. Histochem Cell Biol 122, 151–159 (2004). https://doi.org/10.1007/s00418-004-0688-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-004-0688-7