Abstract
Background
To compare the efficacy and safety of timolol 0.5% versus brinzolamide 1.0% when added to travoprost monotherapy in patients with primary open-angle glaucoma or ocular hypertension.
Methods
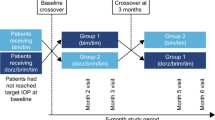
Patients meeting selection criteria (IOP one eye 19 mmHg and ≤32 mmHg and IOP both eyes ≤32 mmHg at 8:00 h) were switched to travoprost monotherapy for 4 weeks. Patients then insufficiently controlled on travoprost (IOP at 8:00 h ≥19 mmHg) at baseline were randomized to receive either travoprost and brinzolamide or travoprost and timolol in a double-masked fashion for 12 weeks.
Results
Two hundred and fifty-three patients underwent the 4-week run-in period. Switching to travoprost resulted in adequate IOP control (<19 mmHg) for 21.7% of patients. After 3 months of treatment, both drug combinations statistically significantly reduced the mean IOP at each time point (8:00, 12:00 and 16:00 h) and the mean diurnal IOP, which was 17.9 ± 2.6 mmHg for the brinzolamide group and 17.0 ± 3.2 mmHg for the timolol group. Both combinations were well-tolerated. However, a statistically significant difference occurred at 16:00 h, with pressures of 16.4 ± 3.2 mmHg and 17.3 ± 2.8 mmHg for the timolol and brinzolamide groups, respectively (p = 0.038). Fifty percent of patients reported one adverse event, whereas in 13.2% three or more adverse effects were named. Hyperemia was found most often (6.3% of the patients).
Conclusion
Both adjunctive combinations moderately reduced IOP in patients inadequately controlled with travoprost monotherapy, with timolol being slightly stronger 8 hours after instillation. Adjunctive treatment with brinzolamide and travoprost may be an alternative for patients not tolerant or not responsive to treatment with timolol and travoprost.
Similar content being viewed by others
References
AGIS Investigators. The Advanced Glaucoma Study Group (AGIS) (2000) The relationship between control of intraocular pressure and visual field deterioration. Am J Ophthalmol 130:429–440
Dubiner HB, Sircy MD, Landry T, Bergamini MV, Silver LH, Darell Turner F, Robertson S, Andrew RM, Weiner A, Przydryga J (2004) Comparison of the diurnal ocular hypotensive efficacy of travoprost and latanoprost over a 44-hour period in patients with elevated intraocular pressure. Clin Ther 26(1):84–91
Franks W, Group BS (2006) Ocular hypotensive efficacy and safety of brinzolamide ophthalmic suspension 1% added to travoprost ophthalmic suspension 0.004% therapy in patients with open-angle glaucoma or ocular hypertension. Curr Med Res Opin 22:1643–1649
Friedman DS, Wilson MR, Liebmann JM, Fechtner RD, Weinreb RN (2004) An evidence-based assessment of risk factors for the progression of ocular hypertension and glaucoma. Am J Ophthalmol 138:19–31
Garcia-Feijoo J, de la Martinez Casa JM, Castillo A, Mendez C, Fernandez-Vidal A, Garcia Sánchez J (2006) Circadian IOP-lowering efficacy of travoprost 0.004% ophthalmic solution compared to latanoprost 0.005%. Curr Med Res Opin 22(9):1689–1697
Goldberg I, Cunha-Vaz J, Jakobson JE et al (2001) Comparison of topical travoprost eye drops given once daily and timolol 0.5% given twice daily in patients with open-angle glaucoma or ocular hypertension. J Glaucoma 10:414–422
Hollo G, Chiselita D, Petkova N, Cvenkel B, Liehneova I, Izgi B, Berta A, Szaflik J, Turcli E, Stewart WC (2006) The efficacy and safety of timolol maleate versus brinzolamide each given twice daily added to travoprost in patients with ocular hypertension or primary open-angle glaucoma. Eur J Ophthalmol 16:816–823
Hollo G, Kothy P (2008) Intraocular pressure reduction with travoprost/timolol fixed combination, with and without adjunctive brinzolamide, in glaucoma. Curr Med Res Opin 24(6):1755–1761
Konstas AG, Karabatsas CH, Lallos N, Georgiadis N et al (2005) 24-hour intraocular pressures with brimonidine purite versus dorzolamide added to latanoprost in primary open-angle glaucoma. Ophthalmology 112:603–608
Mao LK, Stewart WC, Shields MB (1991) Correlation between intraocular pressure control and progressive glaucomatous damage in primary open-angle glaucoma. Am J Ophthalmol 111:51–55
Martínez de la Casa JM, Castillo A, García Feijoo J, Méndez Hernández C, Fernández Vidal A, García Sánchez J (2004) Concomitant administration of travoprost and brinzolamide versus fixed latanoprost/timolol maleate combined therapy: three-month comparison of efficacy and safety. Curr Med Res Opin 20(9):1333–1339
Miura K, Ito K, Okawa C, Sugimoto K, Matsunaga K, Uji Y (2008) Comparison of ocular hypotensive effect and safety of brinzolamide and timolol added to latanoprost. J Glaucoma 17(3):233–237
Netland PA, Landry T, Sullivan EK et al (2001) Travoprost compared with latanoprost and timolol in patients with open-angle glaucoma or ocular hypertension. Am J Ophthalmol 132:472–484
Netland PA, Robertson SM, Sullivan EK, Silver L, Bergamini MV, Krueger S, Weiner AL, Davis AA, Travoprost Study Groups (2003) Response to travoprost in black and nonblack patients with open-angle glaucoma or ocular hypertension. Adv Ther 20(3):149–163
Novack GD, O’Donnell MJ, Molloy DW (2002) New glaucoma medications in the geriatric population: efficacy and safety. J Am Geriatr Soc 50(5):956–962
Orengo-Nania S, Landry T, Von Tress M, Silver LH, Weiner A, Davis AA (2001) Travoprost Study Group. Evaluation of travoprost as adjunctive therapy in patients with uncontrolled intraocular pressure while using timolol 0.5%. Am J Ophthalmol 132(6):860–868
Orzalesi N, Rossetti L, Invernizzi T, Bottoli A, Autelitano A (2000) Effect of timolol, latanoprost and dorzolamide on circadian IOP in glaucoma or ocular hypertension. Invest Ophthalmol Vis Sci 41:2566–2573
Pfeiffer N, European Latanoprost Fixed Combination Study Group (2002) A comparison of the fixed combination of latanoprost and timolol with its individual components. Graefes Arch Clin Exp Ophthalmol 240:893–899
Reis R, Queiroz CF, Santos LC, Avila MP, Magacho L (2006) A randomized, investigator-masked, 4-week study comparing timolol maleate 0.5%, brinzolamide 1%, and brimonidine tartrate 0.2% as adjunctive therapies to travoprost 0.004% in adults with primary open-angle glaucoma or ocular hypertension. Clin Ther 28:552–559
Shoji N, Ogata H, Suyama H, Ishikawa H, Suzuki H, Morita T, Kawai H, Nishimoto H, Nemoto T, Shimizu K (2005) Intraocular pressure lowering effect of brinzolamide 1.0% as adjunctive therapy to latanoprost 0.005% in patients with open angle glaucoma or ocular hypertension: an uncontrolled, open-label study. Curr Med Res Opin 21:503–507
Sorensen SJ, Abel SR (1996) Comparison of the ocular beta-blockers. Ann Pharmacother 30(1):43–54
Stewart WC, Day DG, Stewart JA et al (2004) Short-term ocular tolerability of dorzolamide 2% and brinzolamide 1% vs placebo in primary open-angle glaucoma and ocular hypertension subjects. Eye 18(9):905–910
Williams T, Ginther WH (1982) Hazard of ophthalmic timolol maleate. N Engl J Med 306(24):1485–1486
Zhang WY, Li Wan Po A, Dua HS, Azuara-Blanco A (2001) Meta-analysis of randomised controlled trials comparing latanoprost with timolol in the treatment of patients with open angle glaucoma or ocular hypertension. Br J Ophthalmol 85:983–990
Acknowledgments
The Study was sponsored by Alcon, Forth Worth, TX, USA. The company only had influence on the design of the study. The collection, management, analysis and interpretation of the data were investigator driven. The study group had no financial relationship (e.g., contractors, consultants, stockholders) to the sponsor.
Investigators and co-investigators of the TATS study group
Philippe Denis, Cornut Pierre-Loïc (Department of Ophthalmology, Hôpital Edouard Herriot, Lyon, France); Yves Lachkar, Aoife Doyle (Department of Ophthalmology, Institut du Glaucome, Hopital St Joseph, Paris, France); Silke Kinnebrock, Marlene Maser-Wahle (Department of Ophthalomology, Augenklinik und Universität Mainz, Mainz, Germany); Ines Lanzl, Andreas Pichler (Department of Ophthalomology, Augenklinik und Poliklinik der, Technischen Universität München, München, Germany); Klaus-Dieter Quentin, Sandra Hache (Department of Ophthalmology, Universitätsaugenklinik Göttingen, Göttingen, Germany); Christian Vorwerk, Cordula Pump-Schmidt (Department of Ophthalmology, Otto von Guericke Universität, Magdeburg, Germany); Ilse Strempel, Claudia Knop (Department of Ophthalmology, Klinikum der Philipps-Universität, Marburg, Germany); Carl Erb, Michael Meyer (Department of Ophthalmology, Medizinische Hochschule, Hannover, Germany); Giancarlo Bertoni, Blini Mirella, Alessandro David, Daniella Salerno (Department of Ophthalmology, U.O. Oculistica Azienda Ospedaliera L. Sacco-Polo Universitario, Milano, Italy); Renato Alberto Meduri, Lucia Scorolli, Sergio Zaccaria Scanlinci, Daniela Giardina (Centro per la Prevenzione del Glaucoma, Dipartamento de Scienze Chirurgiche ed Anestesiologiche, Università degli Studi di Bologna, Policlinico S. Orsola-Malpigui, Bologna, Italy); Alessandro Galan (Department of Ophthalmology, U.O.C. di Oculistica, Hospédale S. Antonio Abate, Padova, Italy); Antonio Martínez García (Department of Ophthalmology, Fundación Pública Instituto Galego de Oftalmoloxia, Hospital Provincial de Conxo, Santiago de Compostela, Spain); Manuel Díaz Llopis, Salvador García-Delpech, María García-Pous (Department of Ophthalmology, Hospital General Universitario de Valencia, Spain); Francisco Honrubia, Luis Pablo, LM Larrosa, V Polo (Hospital Universitario Miguel Servet, Zaragoza, Spain); Susana Duch Tuesta, Elena Milla (Department of Ophthalmology, Instituto Condal de Oftalmología, Barcelona, Spain).
Preparation of the study and manuscript writing
Norbert Pfeiffer, Sabine Kurz (Department of Ophthalmology, Augenklinik und Universität Mainz, Mainz, Germany)
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
The Study was sponsored by Alcon, Forth Worth, Texas, USA
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pfeiffer, N., The TATS (Travatan Adjunctive Treatment Study) group. Timolol versus brinzolamide added to travoprost in glaucoma or ocular hypertension. Graefes Arch Clin Exp Ophthalmol 249, 1065–1071 (2011). https://doi.org/10.1007/s00417-011-1650-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-011-1650-8