Abstract
Purpose
The Stokes–Einstein equation predicts that diffusion is inversely correlated with viscosity of the medium. This predicts that diffusion fluxes should be lower in vitreous humour than in saline solution with lower viscosity. We test this hypothesis, which has implications for vitrectomy.
Methods
Diffusion cells were used, with a middle chamber filled with saline solution with different concentrations of hyaluronic acid (0 µg/ml, 30 µg/ml, 90 µg/ml and 180 µg/ml) or porcine vitreous humour, between cellophane membranes. HPLC was used to measure the concentration of the diffusing molecule, dexamethasone, and calculate the flux through the chamber filled with the various media. The diffusion coefficients were calculated, using Fick's law. Viscosity was measured with a Brookfield digital DV-I+viscometer.
Results
The diffusion coefficient for dexamethasone in vitreous humour is 0.065 ± 0.022 cm2/hour, 0.26 ± 0.12 cm2/hour for saline alone, 0.17 ± 0.04 cm2/hour for saline with 30 µg/ml hyaluronan, 0.076 ± 0.009 cm2/hour with 90 µg/ml hyaluronan, and with 180 µg/ml hyaluronan 0.072 ± 0.0018 cm2/hour (p < 0.001). The viscosity of liquid vitreous is 6.29 ± 2.3 cp, and the viscosity of saline alone is 1.01 ± 0.008 cp. For saline with 30 µg/ml of hyaluronan the viscosity is 1.04 ± 0.015, with 90 µg/ml hyaluronan is 1.06 ± 0.01, and with 180 µg/ml hyaluronan is 1.08 ± 0.014 (p < 0.001).
Conclusions
Dexamethasone diffuses four times faster through saline solution than vitreous humour. Liquid vitreous humour is about six times more viscous than saline. The results indicate that diffusion is faster in saline solution than in vitreous humour, in accordance to the Stokes–Einstein equation. This finding can help explain some of the physiological, pharmacological and clinical consequences of vitrectomy.






Similar content being viewed by others
References
Stefansson E, Loftsson T (2006) The Stokes-Einstein equation and the physiological effects of vitreous surgery. Acta Ophthalmol Scand 84(6):718–719. doi:10.1111/j.1600-0420.2006.00778.x
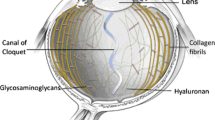
Bos KJ et al (2001) Collagen fibril organisation in mammalian vitreous by freeze etch/rotary shadowing electron microscopy. Micron 32:301–306. doi:10.1016/S0968-4328(00)00035-4
Gelse K, Pöschl E, Aigner T (2003) Collagens - structure, function, and biosynthesis. Adv Drug Deliv Rev 55:1531–1546. doi:10.1016/j.addr.2003.08.002
Bishop PN (2000) Structural macromolecules and supramolecular organisation of the vitreous gel. Prog Retin Eye Res 19(3):323–344. doi:10.1016/S1350-9462(99)00016-6
Ihanamäki T, Pelliniemi LJ, Vuorio E (2004) Collagens and collagen-related matrix components in the human and mouse eye. Prog Retin Eye Res 23:403–434. doi:10.1016/j.preteyeres.2004.04.002
Sebag J (1989) The vitreous: structure, function, and pathobiology. Springer-Verlag, New York, p 173
Itakura H et al (2005) Vitreous collagen metabolism before and after vitrectomy. Graefes Arch Clin Exp Ophthalmol 243(10):994–998. doi:10.1007/s00417-005-1150-9
Sinko P (2006) Martin’s physical pharmacy and pharmaceutical sciences. Lippincott Williams & Wilkins, Philadelphia
Brookfield Engineering Labs Education. More solutions to sticky problems. Brookfield Engineering Labs, Inc., Middleboro, MA, USA
Brookfield Engineering Labs Education. Brookfield Digital Viscometer Model DV-1+ Operating Instructions. Brookfield Engineering Labs, Inc., Middleboro, MA, USA
Weber H, Landwehr G (1982) A new method for the determination of the mechanical properties of the vitreous. Ophthalmic Res 14(5):326–334
Lee B, Litt M, Buchsbaum G (1992) Rheology of the vitreous body. Part I: Viscoelasticity of human vitreous. Biorheology 29(5–6):521–533
Sebag J, Ansari RR, Suh KI (2007) Pharmacologic vitreolysis with microplasmin increases vitreous diffusion coefficients. Graefes Arch Clin Exp Ophthalmol 245(4):576–580. doi:10.1007/s00417-006-0394-3
Sebag J (2005) Molecular biology of pharmacologic vitreolysis. Trans Am Ophthalmol Soc 103:473–494
Tuteja A et al (2007) Breakdown of the continuum stokes-einstein relation for nanoparticle diffusion. Nano Lett 7(5):1276–1281. doi:10.1021/nl070192x
Lee B, Litt M, Buchsbaum G (1994) Rheology of the vitreous body: part 3. Concentration of electrolytes, collagen and hyaluronic acid. Biorheology 31(4):339–351
Lee B, Litt M, Buchsbaum G (1994) Rheology of the vitreous body: Part 2. Viscoelasticity of bovine and porcine vitreous. Biorheology 31(4):327–338
Lee B (1992) Comparative rheological studies of the vitreous body of the eye. University of Pennsylvania dissertation, p 306
Soman N, Banerjee R (2003) Artificial vitreous replacements. Biomed Mater Eng 13(1):59–74
Barton KA et al (2007) Comment on: the Stokes-Einstein equation and the physiological effects of vitreous surgery. Acta Ophthalmol Scand 85(3):339–340. doi:10.1111/j.1600-0420.2007.00902.x
Itakura H et al (2009) Decreased vitreal hyaluronan levels with aging. Ophthalmologica 223(1):32–35. doi:10.1159/000165682
Stefansson E, Landers MB 3rd, Wolbarsht ML (1981) Increased retinal oxygen supply following pan-retinal photocoagulation and vitrectomy and lensectomy. Trans Am Ophthalmol Soc 79:307–334
Stefansson E, Novack RL, Hatchell DL (1990) Vitrectomy prevents retinal hypoxia in branch retinal vein occlusion. Invest Ophthalmol Vis Sci 31(2):284–289
Holekamp NM, Shui YB, Beebe DC (2005) Vitrectomy surgery increases oxygen exposure to the lens: a possible mechanism for nuclear cataract formation. Am J Ophthalmol 139(2):302–310. doi:10.1016/j.ajo.2004.09.046
Stefansson E (2009) Physiology of vitreous surgery. Graefes Arch Exp Clin Ophthalmol 247:147–163
Acknowledgments
This work was supported by grants from the Icelandic Fund for Prevention of Blindness, University of Iceland Research Fund and Helga Jónsdóttir and Sigurliði Kristjánsson Memorial Fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gisladottir, S., Loftsson, T. & Stefansson, E. Diffusion characteristics of vitreous humour and saline solution follow the Stokes Einstein equation. Graefes Arch Clin Exp Ophthalmol 247, 1677–1684 (2009). https://doi.org/10.1007/s00417-009-1141-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-009-1141-3