Abstract
Background
The secretion of a variety of factors by the retinal pigment epithelium (RPE) is essential for the structural integrity of the neuronal retina and choroid, but also plays a pivotal role in the etiology of diseases such as choroidal neovascularisation. A recent study showed that the secretory activity of the RPE is regulated by the activity of a certain type of voltage-dependent Ca2+ channels, the L-type channel. In order to provide a better base for the understanding of the underlying Ca2+ signalling in these cells, we investigated the expression profile of voltage-dependent Ca2+ channel subunits in RPE cells.
Methods
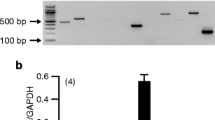
Using RT-PCR techniques with cDNA isolated from RPE cells, we investigated the expression pattern of Ca2+ channel subunits. Furthermore, we analysed Ba2+ currents through voltage-dependent Ca2+ channels in RPE cells by the patch-clamp technique.
Results
We detected the expression of two L-type channel subtypes and the expression of two different T-type channel subtypes. As accessory subunits, they expressed β2 and β4 and all known α2δ subunits. In general, we were able to confirm these data with cDNA from the ARPE-19 cell line. They only showed some differences in their expression pattern of accessory subunits. Since the expression of T-type channels was so far unknown in RPE cells, we confirmed their expression in the RPE using cDNA isolated from freshly isolated human RPE cells. Furthermore, the patch-clamp analysis of Ba2+ currents showed a heterogeneous pattern of voltage-dependent inward currents in RPE cells. In some cells, typical slowly inactivating L-type currents were detected, whereas in other cells fast inactivating T-type currents could be detected.
Conclusions
These data indicate the expression of a so far not detected subtype of voltage-dependent Ca2+ channels, the T-type channels. Together with the expression of L-type channels, RPE cells show a comparable expression pattern to that of other secretory cells, such as β-islets of the pancreas.




Similar content being viewed by others
References
Strauss O (2005) The retinal pigment epithelium in visual function. Physiol Rev 85:845–881
Sher E, Giovannini F, Codignola A et al (2003) Voltage-operated calcium channel heterogeneity in pancreatic beta cells: physiopathological implications. J Bioenerg Biomembr 35:687–696
Wen JF, Cui X, Ahn JS et al (2000) Distinct roles for L- and T-type Ca(2+) channels in regulation of atrial ANP release. Am J Physiol Heart Circ Physiol 279:H2879–H2888
Mears D (2004) Regulation of insulin secretion in islets of Langerhans by Ca(2+)channels. J Membr Biol 200:57–66
Shuttleworth TJ (1997) Intracellular Ca2+ signalling in secretory cells. J Exp Biol 200:303–314
Berridge MJ (2005) Unlocking the secrets of cell signaling. Annu Rev Physiol 67:1–21
Catterall WA (2000) Structure and regulation of voltage-gated Ca2+ channels. Annu Rev Cell Dev Biol 16:521–555
Striessnig J (1999) Pharmacology, structure and function of cardiac L-type Ca(2+) channels. Cell Physiol Biochem 9:242–269
Striessnig J, Hoda JC, Koschak A et al (2004) L-type Ca2+ channels in Ca2+ channelopathies. Biochem Biophys Res Commun 322:1341–1346
Ueda Y, Steinberg RH (1995) Dihydropyridine-sensitive calcium currents in freshly isolated human and monkey retinal pigment epithelial cells. Invest Ophthalmol Vis Sci 36:373–380
Strauss O, Wienrich M (1994) Ca(2+)-conductances in cultured rat retinal pigment epithelial cells. J Cell Physiol 160:89–96
Bollimuntha S, Cornatzer E, Singh BB (2005) Plasma membrane localization and function of TRPC1 is dependent on its interaction with beta-tubulin in retinal epithelium cells. Vis Neurosci 22:163–170
Wimmers S, Strauss O (2007) Basal calcium entry in retinal pigment epithelial cells is mediated by TRPC channels. Invest Ophthalmol Vis Sci
Wimmers S, Karl MO, Strauss O (2007) Ion channels in the RPE. Prog Retin Eye Res 26:263–301
Strauss O, Mergler S, Wiederholt M (1997) Regulation of L-type calcium channels by protein tyrosine kinase and protein kinase C in cultured rat and human retinal pigment epithelial cells. Faseb J 11:859–867
Strauss O, Buss F, Rosenthal R et al (2000) Activation of neuroendocrine L-type channels (alpha1D subunits) in retinal pigment epithelial cells and brain neurons by pp60(c-src). Biochem Biophys Res Commun 270:806–810
Rosenthal R, Strauss O (2002) Ca2+-channels in the RPE. Adv Exp Med Biol 514:225–235
Wollmann G, Lenzner S, Berger W, Rosenthal R, Karl MO, Strauss O (2006) Voltage-dependent ion channels in the mouse RPE: comparison with Norrie disease mice. Vision Res 46:688–698
Rosenthal R, Bakall B, Kinnick T et al (2006) Expression of bestrophin-1, the product of the VMD2 gene, modulates voltage-dependent Ca2+ channels in retinal pigment epithelial cells. Faseb J 20:178–180
Rosenthal R, Heimann H, Agostini H, Martin G, Hansen LL, Strauss O (2007) Ca2+ channels in retinal pigment epithelial cells regulate vascular endothelial growth factor secretion rates in health and disease. Mol Vis 13:443–456
Wu J, Marmorstein AD, Striessnig J, Peachey NS (2007) Voltage-dependent calcium channel CaV1.3 subunits regulate the light peak of the electroretinogram. J Neurophysiol 973731–3735
Mergler S, Strauss O (2002) Stimulation of L-type Ca(2+) channels by increase of intracellular InsP3 in rat retinal pigment epithelial cells. Exp Eye Res 74:29–40
Rosenthal R, Thieme H, Strauss O (2001) Fibroblast growth factor receptor 2 (FGFR2) in brain neurons and retinal pigment epithelial cells act via stimulation of neuroendocrine L-type channels (Ca(v)1.3). Faseb J 15:970–977
Scholze A, Plant TD, Dolphin AC, Nurnberg B (2001) Functional expression and characterization of a voltage-gated CaV1.3 (alpha1D) calcium channel subunit from an insulin-secreting cell line. Mol Endocrinol 15:1211–1221
Barg S (2003) Mechanisms of exocytosis in insulin-secreting B-cells and glucagon-secreting A-cells. Pharmacol Toxicol 92:3–13
Yaney GC, Wheeler MB, Wei X et al (1992) Cloning of a novel alpha 1-subunit of the voltage-dependent calcium channel from the beta-cell. Mol Endocrinol 6:2143–2152
Glassmeier G, Hauber M, Wulfsen I, Weinsberg F, Bauer CK, Schwarz JR (2001) Ca2+ channels in clonal rat anterior pituitary cells (GH3/B6). Pflugers Arch 442:577–587
Koschak A, Reimer D, Huber I et al (2001) alpha 1D (Cav1.3) subunits can form l-type Ca2+ channels activating at negative voltages. J Biol Chem 276:22100–22106
Michna M, Knirsch M, Hoda JC et al (2003) Cav1.3 (alpha1D) Ca2+ currents in neonatal outer hair cells of mice. J Physiol 553:747–758
Rosenthal R, Malek G, Salomon N et al (2005) The fibroblast growth factor receptors, FGFR-1 and FGFR-2, mediate two independent signalling pathways in human retinal pigment epithelial cells. Biochem Biophys Res Commun 337:241–247
Catterall WA (1998) Structure and function of neuronal Ca2+ channels and their role in neurotransmitter release. Cell Calcium 24:307–323
Rossier MF, Burnay MM, Vallotton MB, Capponi AM (1996) Distinct functions of T- and L-type calcium channels during activation of bovine adrenal glomerulosa cells. Endocrinology 137:4817–4826
Frank RN (1997) Growth factors in age-related macular degeneration: pathogenic and therapeutic implications. Ophthalmic Res 29:341–353
Lopez PF, Sippy BD, Lambert HM, Thach AB, Hinton DR (1996) Transdifferentiated retinal pigment epithelial cells are immunoreactive for vascular endothelial growth factor in surgically excised age-related macular degeneration-related choroidal neovascular membranes. Invest Ophthalmol Vis Sci 37:855–868
Kliffen M, Sharma HS, Mooy CM, Kerkvliet S, de Jong PT (1997) Increased expression of angiogenic growth factors in age-related maculopathy. Br J Ophthalmol 81:154–162
Acknowledgements
The authors gratefully acknowledge the technical assistance of Stefanie Ehmer, Sabine Helbing and Birgit Grafelmann. The work was supported by the Deutsche Forschungsgemeinschaft grants DFG STR480/8-2 and STR480/9-1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wimmers, S., Coeppicus, L., Rosenthal, R. et al. Expression profile of voltage-dependent Ca2+ channel subunits in the human retinal pigment epithelium. Graefes Arch Clin Exp Ophthalmol 246, 685–692 (2008). https://doi.org/10.1007/s00417-008-0778-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-008-0778-7