Abstract
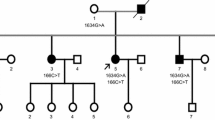
The mutation of vesicle-associated membrane protein-associated protein B (VAPB) was proved to cause family amyotrophic lateral sclerosis (FALS). Only two mutations of VAPB associated with ALS have been reported (p.Pro56Ser and p.Thr46Ile). Here we reported a Chinese Han FALS family caused by a novel VAPB point mutation. The clinical materials of one Chinese Han FALS family were collected. The genetic analysis was carried out by target sequencing and further verified by Sanger sequencing. One novel mutation of c.167C>A (p.Pro56His) on VAPB was found in the proband. The age at onset of the proband was 48 with the onset symptoms of weakness in the right arm, followed by progressive limb and trunk weakness with decreased deep-tendon reflexes, muscular cramps and fasciculation. But the disease duration was more than 15 years. He was under the tracheotomy for 1 year at last visit. Electromyography showed widespread acute and chronic neurogenic damages. His mother presented weakness in her limbs in 50 s and died 15 years later. One of his younger sisters diagnosed as ALS for 6 years also carried the same mutation. She presented the similar symptoms on 41. No dominant upper motor neuron sign was showed. The clinical features were similar to the patients carrying the known mutation of p.Pro56Ser. A novel mutation of VAPB was found in one Chinese Han FALS pedigree. The affected patients presented a much slower progression and the lesions were limited in lower motor neurons.



Similar content being viewed by others
References
Mitchell JD, Borasio GD (2007) Amyotrophic lateral sclerosis. Lancet 369(9578):2031–2041. doi:10.1016/S0140-6736(07)60944-1
Deng HX, Hentati A, Tainer JA, Iqbal Z, Cayabyab A, Hung WY, Getzoff ED, Hu P, Herzfeldt B, Roos RP et al (1993) Amyotrophic lateral sclerosis and structural defects in Cu, Zn superoxide dismutase. Science 261(5124):1047–1051
Corcia P, Couratier P, Blasco H, Andres CR, Beltran S, Meininger V, Vourc’h P (2017) Genetics of amyotrophic lateral sclerosis. Revue neurologique 173(5):254–262. doi:10.1016/j.neurol.2017.03.030
Nishimura AL, Mitne-Neto M, Silva HC, Richieri-Costa A, Middleton S, Cascio D, Kok F, Oliveira JR, Gillingwater T, Webb J, Skehel P, Zatz M (2004) A mutation in the vesicle-trafficking protein VAPB causes late-onset spinal muscular atrophy and amyotrophic lateral sclerosis. Am J Hum Genet 75(5):822–831. doi:10.1086/425287
Landers JE, Leclerc AL, Shi L, Virkud A, Cho T, Maxwell MM, Henry AF, Polak M, Glass JD, Kwiatkowski TJ, Al-Chalabi A, Shaw CE, Leigh PN, Rodriguez-Leyza I, McKenna-Yasek D, Sapp PC, Brown RH Jr (2008) New VAPB deletion variant and exclusion of VAPB mutations in familial ALS. Neurology 70(14):1179–1185. doi:10.1212/01.wnl.0000289760.85237.4e
Chadi G, Maximino JR, Jorge FMH, Borba FC, Gilio JM, Callegaro D, Lopes CG, Santos SND, Rebelo GNS (2017) Genetic analysis of patients with familial and sporadic amyotrophic lateral sclerosis in a Brazilian Research Center. Amyotroph Lateral Scler Frontotemporal Degener 18(3–4):249–255. doi:10.1080/21678421.2016.1254245
Nishimura AL, Al-Chalabi A, Zatz M (2005) A common founder for amyotrophic lateral sclerosis type 8 (ALS8) in the Brazilian population. Hum Genet 118(3–4):499–500. doi:10.1007/s00439-005-0031-y
Tsai CP, Soong BW, Lin KP, Tu PH, Lin JL, Lee YC (2011) FUS, TARDBP, and SOD1 mutations in a Taiwanese cohort with familial ALS. Neurobiol Aging 32(3):553 e513–553 e521. doi:10.1016/j.neurobiolaging.2010.04.009
Ingre C, Pinto S, Birve A, Press R, Danielsson O, de Carvalho M, Gudmundsson G, Andersen PM (2013) No association between VAPB mutations and familial or sporadic ALS in Sweden, Portugal and Iceland. Amyotroph Lateral Scler Frontotemporal Degener 14(7–8):620–627. doi:10.3109/21678421.2013.822515
Conforti FL, Sprovieri T, Mazzei R, Ungaro C, Tessitore A, Tedeschi G, Patitucci A, Magariello A, Gabriele A, Labella V, Simone IL, Majorana G, Monsurro MR, Valentino P, Muglia M, Quattrone A (2006) Sporadic ALS is not associated with VAPB gene mutations in Southern Italy. J Negat Results Biomed 5:7. doi:10.1186/1477-5751-5-7
Funke AD, Esser M, Kruttgen A, Weis J, Mitne-Neto M, Lazar M, Nishimura AL, Sperfeld AD, Trillenberg P, Senderek J, Krasnianski M, Zatz M, Zierz S, Deschauer M (2010) The p. P56S mutation in the VAPB gene is not due to a single founder: the first European case. Clin Genet 77(3):302–303. doi:10.1111/j.1399-0004.2009.01319.x
Chen HJ, Anagnostou G, Chai A, Withers J, Morris A, Adhikaree J, Pennetta G, de Belleroche JS (2010) Characterization of the properties of a novel mutation in VAPB in familial amyotrophic lateral sclerosis. J Biol Chem 285(51):40266–40281. doi:10.1074/jbc.M110.161398
Millecamps S, Salachas F, Cazeneuve C, Gordon P, Bricka B, Camuzat A, Guillot-Noel L, Russaouen O, Bruneteau G, Pradat PF, Le Forestier N, Vandenberghe N, Danel-Brunaud V, Guy N, Thauvin-Robinet C, Lacomblez L, Couratier P, Hannequin D, Seilhean D, Le Ber I, Corcia P, Camu W, Brice A, Rouleau G, LeGuern E, Meininger V (2010) SOD1, ANG, VAPB, TARDBP, and FUS mutations in familial amyotrophic lateral sclerosis: genotype–phenotype correlations. J Med Genet 47(8):554–560. doi:10.1136/jmg.2010.077180
Di L, Chen H, Da Y, Wang S, Shen XM (2016) Atypical familial amyotrophic lateral sclerosis with initial symptoms of pain or tremor in a Chinese family harboring VAPB-P56S mutation. J Neurol 263(2):263–268. doi:10.1007/s00415-015-7965-3
van Blitterswijk M, van Es MA, Koppers M, van Rheenen W, Medic J, Schelhaas HJ, van der Kooi AJ, de Visser M, Veldink JH, van den Berg LH (2012) VAPB and C9orf72 mutations in 1 familial amyotrophic lateral sclerosis patient. Neurobiol Aging 33(12):2950 e2951–2950 e2954. doi:10.1016/j.neurobiolaging.2012.07.004
Kabashi E, El Oussini H, Bercier V, Gros-Louis F, Valdmanis PN, McDearmid J, Mejier IA, Dion PA, Dupre N, Hollinger D, Sinniger J, Dirrig-Grosch S, Camu W, Meininger V, Loeffler JP, Rene F, Drapeau P, Rouleau GA, Dupuis L (2013) Investigating the contribution of VAPB/ALS8 loss of function in amyotrophic lateral sclerosis. Hum Mol Genet 22(12):2350–2360. doi:10.1093/hmg/ddt080
de Carvalho M, Dengler R, Eisen A, England JD, Kaji R, Kimura J, Mills K, Mitsumoto H, Nodera H, Shefner J, Swash M (2008) Electrodiagnostic criteria for diagnosis of ALS. Clin Neurophysiol 119(3):497–503. doi:10.1016/j.clinph.2007.09.143
Xiong WX, Sun YM, Guan RY, Luo SS, Chen C, An Y, Wang J, Wu JJ (2016) The heterozygous A53T mutation in the alpha-synuclein gene in a Chinese Han patient with Parkinson disease: case report and literature review. J Neurol 263(10):1984–1992. doi:10.1007/s00415-016-8213-1
Wu ZY, Lin MT, Murong SX, Wang N (2003) Molecular diagnosis and prophylactic therapy for presymptomatic Chinese patients with Wilson disease. Arch Neurol 60(5):737–741. doi:10.1001/archneur.60.5.737
Sun YM, Li HL, Guo QH, Wu P, Hong Z, Lu CZ, Wu ZY (2012) The polymorphism of the ATP-binding cassette transporter 1 gene modulates Alzheimer disease risk in Chinese Han ethnic population. Am J Geriatr Psychiatry 20(7):603–611. doi:10.1097/JGP.0b013e3182423b6a
Tomiyama H (2013) [C9orf72 in Japanese amyotrophic lateral sclerosis (ALS)]. Rinsho shinkeigaku (Clin Neurol) 53(11):1074–1076
Jiao B, Tang B, Liu X, Yan X, Zhou L, Yang Y, Wang J, Xia K, Shen L (2014) Identification of C9orf72 repeat expansions in patients with amyotrophic lateral sclerosis and frontotemporal dementia in mainland China. Neurobiol Aging 35(4):936 e919–936 e922. doi:10.1016/j.neurobiolaging.2013.10.001
DeJesus-Hernandez M, Mackenzie IR, Boeve BF, Boxer AL, Baker M, Rutherford NJ, Nicholson AM, Finch NA, Flynn H, Adamson J, Kouri N, Wojtas A, Sengdy P, Hsiung GY, Karydas A, Seeley WW, Josephs KA, Coppola G, Geschwind DH, Wszolek ZK, Feldman H, Knopman DS, Petersen RC, Miller BL, Dickson DW, Boylan KB, Graff-Radford NR, Rademakers R (2011) Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron 72(2):245–256. doi:10.1016/j.neuron.2011.09.011
Jiao B, Guo JF, Wang YQ, Yan XX, Zhou L, Liu XY, Zhang FF, Zhou YF, Xia K, Tang BS, Shen L (2013) C9orf72 mutation is rare in Alzheimer’s disease, Parkinson’s disease, and essential tremor in China. Front Cell Neurosci 7:164. doi:10.3389/fncel.2013.00164
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, Grody WW, Hegde M, Lyon E, Spector E, Voelkerding K, Rehm HL, Committee ALQA (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17(5):405–424. doi:10.1038/gim.2015.30
Venselaar H, Te Beek TA, Kuipers RK, Hekkelman ML, Vriend G (2010) Protein structure analysis of mutations causing inheritable diseases. An e-Science approach with life scientist friendly interfaces. BMC Bioinform 11:548. doi:10.1186/1471-2105-11-548
Nishimura Y, Hayashi M, Inada H, Tanaka T (1999) Molecular cloning and characterization of mammalian homologues of vesicle-associated membrane protein-associated (VAMP-associated) proteins. Biochem Biophys Res Commun 254(1):21–26. doi:10.1006/bbrc.1998.9876
Navone F, Genevini P, Borgese N (2015) Autophagy and neurodegeneration: insights from a cultured cell model of ALS. Cells 4(3):354–386. doi:10.3390/cells4030354
Marques VD, Barreira AA, Davis MB et al (2006) Expanding the phenotypes of the Pro56Ser VAPB mutation: proximal SMA with dysautonomia. Muscle Nerve 34:731–739
Acknowledgements
This study was supported by the Founding of Shanghai Municipal Commission of Health and Family Planning (20124222).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors report no conflicts of interest.
Ethical standards
This study has been approved by the ethics committee of Huashan hospital and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sun, Ym., Dong, Y., Wang, J. et al. A novel mutation of VAPB in one Chinese familial amyotrophic lateral sclerosis pedigree and its clinical characteristics. J Neurol 264, 2387–2393 (2017). https://doi.org/10.1007/s00415-017-8628-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-017-8628-3