Abstract
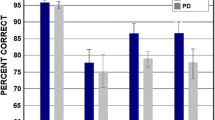
Empirical studies of taste function in multiple sclerosis (MS) are rare. Moreover, a detailed assessment of whether quantitative measures of taste function correlate with the punctate and patchy myelin-related lesions found throughout the CNS of MS patients has not been made. We administered a 96-trial test of sweet (sucrose), sour (citric acid), bitter (caffeine) and salty (NaCl) taste perception to the left and right anterior (CN VII) and posterior (CN IX) tongue regions of 73 MS patients and 73 matched controls. The number and volume of lesions were assessed using quantitative MRI in 52 brain regions of 63 of the MS patients. Taste identification scores were significantly lower in the MS patients for sucrose (p = 0.0002), citric acid (p = 0.0001), caffeine (p = 0.0372) and NaCl (p = 0.0004) and were present in both anterior and posterior tongue regions. The percent of MS patients with identification scores falling below the 5th percentile of controls was 15.07 % for caffeine, 21.9 % for citric acid, 24.66 % for sucrose, and 31.50 % for NaCl. Such scores were inversely correlated with lesion volumes in the temporal, medial frontal, and superior frontal lobes, and with the number of lesions in the left and right superior frontal lobes, right anterior cingulate gyrus, and left parietal operculum. Regardless of the subject group, women outperformed men on the taste measures. These findings indicate that a sizable number of MS patients exhibit taste deficits that are associated with MS-related lesions throughout the brain.



Similar content being viewed by others
References
Sorgun MH, Yucesan C, Tegin C (2014) Is malnutrition a problem for multiple sclerosis patients? J Clin Neurosci 21:1603–1605
Müller R (1949) Studies on disseminated sclerosis with special reference to symptomatology, course and prognosis. Acta Med Scand Suppl 20:68–75
Kurtzke JF, Beebe GW, Nagler B, Auth TL, Kurland LT, Nefzger MD (1972) Studies on the natural history of multiple sclerosis. Act Neurol Scand 48:19–46
Kahana E, Leibowitz U, Alter M (1973) Brainstem and cranial nerve involvement in multiple sclerosis. Act Neurol Scand 49:269–279
Wender M, Szmeja Z (1971) Badanie sluchu, czynnosci narzadu przedsionkowego, smaku I wechu u chorych na stwardnienie rozsiane. Neurol Neurochir Pol 5:179–184
Bromley SM, Doty RL (2015) Clinical disorders affecting taste: an update. In: RL Doty (ed) Handbook of Olfaction and Gustation. John Wiley Sons, Hoboken, pp 887–910
Deems DA, Doty RL, Settle RG, Moore-Gillon V, Shaman P, Mester AF, Kimmelman CP, Brightman VJ, Snow JB Jr (1991) Smell and taste disorders, a study of 750 patients from the University of Pennsylvania Smell and Taste Center. Arch Otolaryngol Head Neck Surg 117:519–528
Soter A, Kim J, Jackman A, Tourbier I, Kaul A, Doty RL (2008) Accuracy of self-report in detecting taste dysfunction. Laryngoscope 118:611–617
Rollin H (1976) Gustatory disturbances in multiple sclerosis. Laryngol Rhinol Otol 55:678–681 (German)
Dahlslett SB, Goektas O, Schmidt F, Harms L, Olze H, Fleiner F (2012) Psychophysiological and electrophysiological testing of olfactory and gustatory function in patients with multiple sclerosis. Eur Arch Otorhinolaryngol 269:1163–1169
Fleiner F, Dahlslett SB, Schmidt F, Harms L, Goektas O (2010) Olfactory and gustatory function in patients with multiple sclerosis. Am J Rhinol Allergy 24:e93–e97
Schmidt FA, Goktas O, Harms L, Bohner G, Erb K, Dahlslett B, Fleiner F (2011) Structural correlates of taste and smell loss in encephalitis disseminata. PLoS One 6:e19702
Doty RL, Tourbier I, Davis S, Rotz J, Cuzzocreo JL, Treem J, Shephard N, Pham DL (2012) Pure-tone auditory thresholds are not chronically elevated in multiple sclerosis. Behav Neurosci 126:314–324
Doty RL, Haxel BR (2005) Objective assessment of terbinafine-induced taste loss. Laryngoscope 115:2035–2037
Stinton N, Atif MA, Barkat N, Doty RL (2010) Influence of smell loss on taste function. Behav Neurosci 124:256–264
Doty RL, Cometto-Muniz JE, Jalowayski AA, Dalton P, Kendal-Reed M, Hodgson M (2004) Assessment of upper respiratory tract and ocular irritative effects of volatile chemicals in humans. Crit Rev Toxicol 34:85–142
Goldszal AF, Davatzikos C, Pham DL, Yan MX, Bryan RN, Resnick SM (1998) An image-processing system for qualitative and quantitative volumetric analysis of brain images. J Comput Assist Tomogr 22:827–837
Bazin PL, Cuzzocreo JL, Yassa MA, Gandler W, McAuliffe MJ, Bassett SS, Pham DL (2007) Volumetric neuroimage analysis extensions for the MIPAV software package. J Neurosci Methods 165:111–121
Pham DL, Prince JL (1999) Adaptive fuzzy segmentation of magnetic resonance images. IEEE Trans Med Imaging 18:737–752
Pham DL (2001) Spatial models for fuzzy clustering. Comput Med Imaging Graph 84:285–297
Van Leemput K, Maes F, Vandermeulen D, Colchester A, Suetens P (2001) Automated segmentation of multiple sclerosis lesions by model outlier detection. IEEE Trans Med Imaging 20:677–688
Van LK, Maes F, Vandermeulen D, Colchester A, Suetens P (2001) Automated segmentation of multiple sclerosis lesions by model outlier detection. IEEE Trans Med Imaging 20:677–688
Shen D, Davatzikos C (2002) HAMMER: hierarchical attribute matching mechanism for elastic registration. IEEE Trans Med Imaging 21:1421–1439
Wilkinson L (1990) SYSTAT: the system for statistics. SYSTAT, Inc., Evanston
Perneger TV (1998) What’s wrong with Bonferroni adjustments. BMJ 316:1236–1238
Rothman KJ (1990) No adjustments are needed for multiple comparisons. Epidemiology 1:43–46
Onoda K, Ikeda M (1999) Gustatory disturbance due to cerebrovascular disorder. Laryngoscope 109:123–128
Shikama Y, Kato T, Nagaoka U, Hosoya T, Katagiri T, Yamaguchi K, Sasaki H (1996) Localization of the gustatory pathway in the human midbrain. Neurosci Lett 218:198–200
McMahon EJ, Campbell KL, Bauer JD (2014) Taste perception in kidney disease and relationship to dietary sodium intake. Appetite 83:236–241
Doty RL (1978) Gender and reproductive state correlates of taste perception in humans. In: McGill TE, Dewsbury DA, Sachs BD (eds) Sex and behavior: status and prospectus. Plenum, New York, pp 337–362
Bartoshuk LM, Duffy VB, Miller IJ (1994) PTC/PROP tasting: anatomy, psychophysics, and sex effects. Physiol Behav 56:1165–1171
Brass SD, Zivadinov R, Bakshi R (2008) Acute demyelinating optic neuritis: a review. Front Biosci 13:2376–2390
Grénman R (1985) Involvement of the audiovestibular system in multiple sclerosis. An otoneurologic and audiologic study. Acta Otolaryngol Suppl 420:1–95
Lee J, Tucker RM, Tan SY, Running CA, Jones JB, Mattes RD (2015) Nutritional implications of taste and smell dysfunction. In: Doty RL (ed) Handbook of olfaction and gustation. Wiley, New York, pp 831–863
Devanand DP, Lee S, Manly J, Andrews H, Schupf N, Masurkar A, Stern Y, Mayeux R, Doty RL (2015) Olfactory identification deficits and increased mortality in the community. Ann Neurol 78:401–411
Solemdal K, Moinichen-Berstad C, Mowe M, Hummel T, Sandvik L (2014) Impaired taste and increased mortality in acutely hospitalized older people. Chem Senses 39:263–269
Catalanotto FA, Dore-Duffy P, Donaldson JO, Testa M, Peterson M, Ostrom KM (1984) Quality-specific taste changes in multiple sclerosis. Ann Neurol 16:611–615
Small DM, Gregory MD, Mak YE, Gitelman D, Mesulam MM, Parrish T (2003) Dissociation of neural representation of intensity and affective valuation in human gustation. Neuron 39:701–711
Rovee CK, Cohen RY, Shlapack W (1975) Life-span stability in olfactory sensitivity. Dev Psychol 11:311–318
Methven L, Allen VJ, Withers CA, Gosney MA (2012) Ageing and taste. Proc Nutr Soc 71:1–10
Onoda K, Kobayakawa T, Ikeda M, Saito S, Kida A (2005) Laterality of human primary gustatory cortex studied by MEG. Chem Senses 30:657–666
Tsivgoulis G, Ioannis H, Vadikolias K, Galetta SL, Piperidou C (2011) Bilateral ageusia caused by a unilateral midbrain and thalamic infarction. J Neuroimaging 21:263–265
Nakajima M, Ohtsuki T, Minematsu K (2010) Bilateral hypogeusia in a patient with a unilateral paramedian thalamic infarction. J Neurol Neurosurg Psychiatry 81:700–701
Yildiz M, Tettenborn B, Radue EW, Bendfeldt K, Borgwardt S (2014) Association of cognitive impairment and lesion volumes in multiple sclerosis–a MRI study. Clin Neurol Neurosurg 127:54–58
Lund H, Jonsson A, Andresen J, Rostrup E, Paulson OB, Sorensen PS (2012) Cognitive deficits in multiple sclerosis: correlations with T2 changes in normal appearing brain tissue. Acta Neurol Scand 125:338–344
Harrison DM, Roy S, Oh J, Izbudak I, Pham D, Courtney S, Caffo B, Jones CK, van Zijl P, Calabresi PA (2015) Association of cortical lesion burden on 7-T magnetic resonance imaging with cognition and disability in multiple sclerosis. JAMA Neurol 72:1004–1012
Dolezal O, Dwyer MG, Horakova D, Havrdova E, Minagar A, Balachandran S, Bergsland N, Seidl Z, Vaneckova M, Fritz D, Krasensky J, Zivadinov R (2007) Detection of cortical lesions is dependent on choice of slice thickness in patients with multiple sclerosis. Int Rev Neurobiol 79:475–489
Reddy H, Narayanan S, Arnoutelis R, Jenkinson M, Antel J, Matthews PM, Arnold DL (2000) Evidence for adaptive functional changes in the cerebral cortex with axonal injury from multiple sclerosis. Brain 123(Pt 11):2314–2320
Loevner LA, Grossman RI, Cohen JA, Lexa FJ, Kessler D, Kolson DL (1995) Microscopic disease in normal-appearing white matter on conventional MR images in patients with multiple sclerosis: assessment with magnetization-transfer measurements. Radiology 196:511–515
Gasperini C, Horsfield MA, Thorpe JW, Kidd D, Barker GJ, Tofts PS, MacManus DG, Thompson AJ, Miller DH, McDonald WI (1996) Macroscopic and microscopic assessments of disease burden by MRI in multiple sclerosis: relationship to clinical parameters. J Magn Reson Imaging 6:580–584
Chaudhari N, Roper SD (2010) The cell biology of taste. J Cell Biol 190:285–296
Chandrashekar J, Hoon MA, Ryba NJ, Zuker CS (2006) The receptors and cells for mammalian taste. Nature 444:288–294
Meyerhof W, Batram C, Kuhn C, Brockhoff A, Chudoba E, Bufe B, Appendino G, Behrens M (2010) The molecular receptive ranges of human TAS2R bitter taste receptors. Chem Senses 35:157–170
Roudnitzky N, Bufe B, Thalmann S, Kuhn C, Gunn HC, Xing C, Crider BP, Behrens M, Meyerhof W, Wooding SP (2011) Genomic, genetic and functional dissection of bitter taste responses to artificial sweeteners. Hum Mol Genet 20:3437–3449
Schattling B, Steinbach K, Thies E, Kruse M, Menigoz A, Ufer F, Flockerzi V, Bruck W, Pongs O, Vennekens R, Kneussel M, Freichel M, Merkler D, Friese MA (2012) TRPM4 cation channel mediates axonal and neuronal degeneration in experimental autoimmune encephalomyelitis and multiple sclerosis. Nat Med 18:1805–1811
Bouafia A, Golmard JL, Thuries V, Sazdovitch V, Hauw JJ, Fontaine B, Seilhean D (2014) Axonal expression of sodium channels and neuropathology of the plaques in multiple sclerosis. Neuropathol Appl Neurobiol 40:579–590
Velle W (1987) Sex differences in sensory functions. Perspect Biol Med 30:490–522
Goldstein JM, Seidman LJ, Horton NJ, Makris N, Kennedy DN, Caviness VS Jr, Faraone SV, Tsuang MT (2001) Normal sexual dimorphism of the adult human brain assessed by in vivo magnetic resonance imaging. Cereb Cortex 11:490–497
Ochoa-Reparaz J, Mielcarz DW, Begum-Haque S, Kasper LH (2011) Gut, bugs, and brain: role of commensal bacteria in the control of central nervous system disease. Ann Neurol 69:240–247
Acknowledgments
We are indebted to the subjects who participated in this study. We thank Nancy A. Lee, M.D., Andre S. Souza, M.D., and Hussam Tallab, M.D., for their contributions to this work, and Greg Smutzer, Ph.D., for comments on an earlier version of the manuscript. Special thanks goes out to Inna Chung, who played a significant role in preparing the manuscript for journal submission. This research was supported by the U.S. National Institutes of Health Grants RO1 DC 02974 (Doty), R01 NS 37172 (Doty), and R01 NS070906 (Pham).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None of the authors of this study declare any conflicts of interest.
Rights and permissions
About this article
Cite this article
Doty, R.L., Tourbier, I.A., Pham, D.L. et al. Taste dysfunction in multiple sclerosis. J Neurol 263, 677–688 (2016). https://doi.org/10.1007/s00415-016-8030-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-016-8030-6