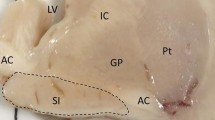
Abstract
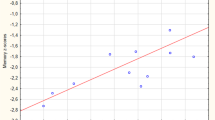
Similar to Alzheimer’s disease (AD), dementia with Lewy bodies (DLB) is characterized by a profound degeneration of cortically-projecting cholinergic neurons of the basal forebrain (BF) and associated depletion of cortical cholinergic activity. We aimed to investigate subregional atrophy of the BF in DLB in vivo and compare it to the pattern of BF atrophy in AD. Structural MRI scans of 11 patients with DLB, 11 patients with Alzheimer’s disease, and 22 healthy controls were analysed using a recently developed technique for automated BF morphometry based on high-dimensional image warping and cytoarchitectonic maps of BF cholinergic nuclei. For comparison, hippocampus volume was assessed within the same morphometric framework using recently published consensus criteria for the definition of hippocampus outlines on MRI. The DLB group demonstrated pronounced and subregion-specific atrophy of the BF which was comparable to BF atrophy in AD: volume of the nucleus basalis Meynert was significantly reduced by 20–25 %, whereas rostral BF nuclei were only marginally affected. By contrast, hippocampus volume was markedly less affected in DLB compared to AD. Global cognition as determined by MMSE score was associated with BF volume in AD, but not in DLB, whereas visuoperceptual function as determined by the trail making test was associated with BF volume in DLB, but not in AD. DLB may be characterized by a more selective degeneration of the cholinergic BF compared to AD, which may be related to the differential cognitive profiles in both conditions.


Similar content being viewed by others
References
Ballard C, Aarsland D, Francis P, Corbett A (2013) Neuropsychiatric symptoms in patients with dementias associated with cortical Lewy bodies: pathophysiology, clinical features, and pharmacological management. Drugs Aging 30:603–611
Barber R, Gholkar A, Scheltens P, Ballard C, McKeith IG, O’Brien JT (1999) Medial temporal lobe atrophy on MRI in dementia with Lewy bodies. Neurology 52:1153–1158
Basel MC (2005) CERAD-Plus: the consortium to establish a registry for Alzheimer’s disease. Univeristätsspital, Basel
Birks J (2006) Cholinesterase inhibitors for Alzheimer’s disease. Cochrane Database Syst Rev 1:CD005593
Boban M, Kostovic I, Simic G (2006) Nucleus subputaminalis: neglected part of the basal nucleus of Meynert. Brain 129:E42 (author reply E43)
Boccardi M, Bocchetta M, Ganzola R, Robitaille N, Redolfi A, Duchesne S, Jack CR Jr, Frisoni GB (2013) Operationalizing protocol differences for EADC-ADNI manual hippocampal segmentation. Alzheimers Dement. doi:10.1016/j.jalz.2013.03.001
Burton EJ, Barber R, Mukaetova-Ladinska EB, Robson J, Perry RH, Jaros E, Kalaria RN, O’Brien JT (2009) Medial temporal lobe atrophy on MRI differentiates Alzheimer’s disease from dementia with Lewy bodies and vascular cognitive impairment: a prospective study with pathological verification of diagnosis. Brain 132:195–203
Cagnin A, Gnoato F, Jelcic N, Favaretto S, Zarantonello G, Ermani M, Dam M (2013) Clinical and cognitive correlates of visual hallucinations in dementia with Lewy bodies. J Neurol Neurosurg Psychiatry 84:505–510
Candy J, Perry R, Perry E, Irving D, Blessed G, Fairbairn A, Tomlinson B (1983) Pathological changes in the nucleus of Meynert in Alzheimer’s and Parkinson’s diseases. J Neurol Sci 59:277–289
Chow N, Aarsland D, Honarpisheh H, Beyer MK, Somme JH, Elashoff D, Rongve A, Tysnes OB, Thompson PM, Apostolova LG (2012) Comparing hippocampal atrophy in Alzheimer’s dementia and dementia with Lewy bodies. Dement Geriatr Cogn Disord 34:44–50
Collerton D, Burn D, McKeith I, O’Brien J (2003) Systematic review and meta-analysis show that dementia with Lewy bodies is a visual-perceptual and attentional-executive dementia. Dement Geriatr Cogn Disord 16:229–237
Cummings J, Emre M, Aarsland D, Tekin S, Dronamraju N, Lane R (2010) Effects of rivastigmine in Alzheimer’s disease patients with and without hallucinations. J Alzheimers Dis 20:301–311
de Medeiros K, Robert P, Gauthier S, Stella F, Politis A, Leoutsakos J, Taragano F, Kremer J, Brugnolo A, Porsteinsson AP, Geda YE, Brodaty H, Gazdag G, Cummings J, Lyketsos C (2010) The Neuropsychiatric Inventory-Clinician rating scale (NPI-C): reliability and validity of a revised assessment of neuropsychiatric symptoms in dementia. Int Psychogeriatr 22:984–994
de Souza Silva MA, Lenz B, Rotter A, Biermann T, Peters O, Ramirez A, Jessen F, Maier W, Hull M, Schroder J, Frolich L, Teipel S, Gruber O, Kornhuber J, Huston JP, Muller CP, Schable S (2013) Neurokinin3 receptor as a target to predict and improve learning and memory in the aged organism. Proc Natl Acad Sci USA 110:15097–15102
Delli Pizzi S, Franciotti R, Tartaro A, Caulo M, Thomas A, Onofrj M, Bonanni L (2014) Structural alteration of the dorsal visual network in DLB patients with visual hallucinations: a cortical thickness MRI study. PLoS One 9:e86624
Eagger SA, Levy R, Sahakian BJ (1991) Tacrine in Alzheimer’s disease. Lancet 337:989–992
Ferman TJ, Smith GE, Boeve BF, Graff-Radford NR, Lucas JA, Knopman DS, Petersen RC, Ivnik RJ, Wszolek Z, Uitti R, Dickson DW (2006) Neuropsychological differentiation of dementia with Lewy bodies from normal aging and Alzheimer’s disease. Clin Neuropsychol 20:623–636
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Förstl H, Burns A, Luthert P, Cairns N, Levy R (1993) The Lewy-body variant of Alzheimer’s disease. Clinical and pathological findings. Br J Psychiatry 162:385–392
Fujishiro H, Umegaki H, Isojima D, Akatsu H, Iguchi A, Kosaka K (2006) Depletion of cholinergic neurons in the nucleus of the medial septum and the vertical limb of the diagonal band in dementia with Lewy bodies. Acta Neuropathol 111:109–114
Goldman JG, Stebbins GT, Dinh V, Bernard B, Merkitch D, deToledo-Morrell L, Goetz CG (2014) Visuoperceptive region atrophy independent of cognitive status in patients with Parkinson’s disease with hallucinations. Brain 137:849–859
Graff-Radford J, Boeve BF, Pedraza O, Ferman TJ, Przybelski S, Lesnick TG, Vemuri P, Senjem ML, Smith GE, Knopman DS, Lowe V, Jack CR Jr, Petersen RC, Kantarci K (2012) Imaging and acetylcholinesterase inhibitor response in dementia with Lewy bodies. Brain 135:2470–2477
Grothe M, Heinsen H, Teipel S (2013) Longitudinal measures of cholinergic forebrain atrophy in the transition from healthy aging to Alzheimer’s disease. Neurobiol Aging 34:1210–1220
Grothe M, Heinsen H, Teipel SJ (2012) Atrophy of the cholinergic Basal forebrain over the adult age range and in early stages of Alzheimer’s disease. Biol Psychiatry 71:805–813
Grothe M, Zaborszky L, Atienza M, Gil-Neciga E, Rodriguez-Romero R, Teipel SJ, Amunts K, Suarez-Gonzalez A, Cantero JL (2010) Reduction of basal forebrain cholinergic system parallels cognitive impairment in patients at high risk of developing Alzheimer’s disease. Cereb Cortex 20:1685–1695
Grothe MJ, Ewers M, Krause B, Heinsen H, Teipel SJ (2014) Basal forebrain atrophy and cortical amyloid deposition in nondemented elderly subjects. Alzheimers Dement
Halliday GM, Cullen K, Cairns MJ (1993) Quantitation and three-dimensional reconstruction of Ch4 nucleus in the human basal forebrain. Synapse 15:1–16
Hanyu H, Asano T, Sakurai H, Tanaka Y, Takasaki M, Abe K (2002) MR analysis of the substantia innominata in normal aging, Alzheimer disease, and other types of dementia. AJNR Am J Neuroradiol 23:27–32
Hanyu H, Shimizu S, Tanaka Y, Hirao K, Iwamoto T, Abe K (2007) MR features of the substantia innominata and therapeutic implications in dementias. Neurobiol Aging 28:548–554
Hanyu H, Tanaka Y, Shimizu S, Sakurai H, Iwamoto T, Abe K (2005) Differences in MR features of the substantia innominata between dementia with Lewy bodies and Alzheimer’s disease. J Neurol 252:482–484
Hepp DH, Ruiter AM, Galis Y, Voorn P, Rozemuller AJ, Berendse HW, Foncke EM, van de Berg WD (2013) Pedunculopontine cholinergic cell loss in hallucinating Parkinson disease patients but not in dementia with Lewy bodies patients. J Neuropathol Exp Neurol 72:1162–1170
Hirano S, Shinotoh H, Eidelberg D (2012) Functional brain imaging of cognitive dysfunction in Parkinson’s disease. J Neurol Neurosurg Psychiatry 83:963–969
Iraizoz I, Guijarro JL, Gonzalo LM, de Lacalle S (1999) Neuropathological changes in the nucleus basalis correlate with clinical measures of dementia. Acta Neuropathol 98:186–196
Janzen J, van‘t Ent D, Lemstra AW, Berendse HW, Barkhof F, Foncke EM (2012) The pedunculopontine nucleus is related to visual hallucinations in Parkinson’s disease: preliminary results of a voxel-based morphometry study. J Neurol 259:147–154
Jicha GA, Schmitt FA, Abner E, Nelson PT, Cooper GE, Smith CD, Markesbery WR (2010) Prodromal clinical manifestations of neuropathologically confirmed Lewy body disease. Neurobiol Aging 31:1805–1813
Kantarci K, Ferman TJ, Boeve BF, Weigand SD, Przybelski S, Vemuri P, Murray MM, Senjem ML, Smith GE, Knopman DS (2012) Focal atrophy on MRI and neuropathologic classification of dementia with Lewy bodies. Neurology 79:553–560
Kilimann I, Grothe M, Heinsen H, Alho EJ, Grinberg L, Amaro E Jr, Dos Santos GA, da Silva RE, Mitchell AJ, Frisoni GB, Bokde AL, Fellgiebel A, Filippi M, Hampel H, Kloppel S, Teipel SJ (2014) Subregional basal forebrain atrophy in Alzheimer’s disease: a multicenter study. J Alzheimers Dis 40:687–700
Klein JC, Eggers C, Kalbe E, Weisenbach S, Hohmann C, Vollmar S, Baudrexel S, Diederich NJ, Heiss WD, Hilker R (2010) Neurotransmitter changes in dementia with Lewy bodies and Parkinson disease dementia in vivo. Neurology 74:885–892
Levy R, Eagger S, Griffiths M, Perry E, Honavar M, Dean A, Lantos P (1994) Lewy bodies and response to tacrine in Alzheimer’s disease. Lancet 343:176
Liberini P, Valerio A, Memo M, Spano PF (1996) Lewy-body dementia and responsiveness to cholinesterase inhibitors: a paradigm for heterogeneity of Alzheimer’s disease? Trends Pharmacol Sci 17:155–160
Lippa CF, Smith TW, Perry E (1999) Dementia with Lewy bodies: choline acetyltransferase parallels nucleus basalis pathology. J Neural Transm 106:525–535
Marra C, Quaranta D, Profice P, Pilato F, Capone F, Iodice F, Di Lazzaro V, Gainotti G (2012) Central cholinergic dysfunction measured “in vivo” correlates with different behavioral disorders in Alzheimer’s disease and dementia with Lewy body. Brain Stimul 5:533–538
Marui W, Iseki E, Kato M, Akatsu H, Kosaka K (2004) Pathological entity of dementia with Lewy bodies and its differentiation from Alzheimer’s disease. Acta Neuropathol 108:121–128
McKeith I, Dickson D, Lowe J, Emre M, O’brien J, Feldman H, Cummings J, Duda J, Lippa C, Perry E (2005) Diagnosis and management of dementia with Lewy bodies third report of the DLB consortium. Neurology 65:1863–1872
McKeith I, Mintzer J, Aarsland D, Burn D, Chiu H, Cohen-Mansfield J, Dickson D, Dubois B, Duda JE, Feldman H, Gauthier S, Halliday G, Lawlor B, Lippa C, Lopez OL, Machado JC, O’Brien J, Playfer J (2004) Dementia with Lewy bodies. Lancet Neurol 3:19–28
McKeith I, O’Brien J, Walker Z, Tatsch K, Booij J, Darcourt J, Padovani A, Giubbini R, Bonuccelli U, Volterrani D, Holmes C, Kemp P, Tabet N, Meyer I, Reininger C (2007) Sensitivity and specificity of dopamine transporter imaging with 123I-FP-CIT SPECT in dementia with Lewy bodies: a phase III, multicentre study. Lancet Neurol 6:305–313
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM (1984) Clinical diagnosis of Alzheimer’s disease Report of the NINCDS-ADRDA Work Group* under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34:939
Mesulam MM, Mufson EJ, Levey AI, Wainer BH (1983) Cholinergic innervation of cortex by the basal forebrain: cytochemistry and cortical connections of the septal area, diagonal band nuclei, nucleus basalis (substantia innominata), and hypothalamus in the rhesus monkey. J Comp Neurol 214:170–197
Mesulam MM, Mufson EJ, Wainer BH, Levey AI (1983) Central cholinergic pathways in the rat: an overview based on an alternative nomenclature (Ch1-Ch6). Neuroscience 10:1185–1201
Mori E, Ikeda M, Kosaka K (2012) Donepezil for dementia with Lewy bodies: a randomized, placebo-controlled trial. Ann Neurol 72:41–52
Morris J, Heyman A, Mohs R, Hughes J, Van Belle G, Fillenbaum G, Mellits E, Clark C, the CERAD investigators (1989) The consortium to establish a registry for Alzheimer’s disease (CERAD). Part I. Clinical and neuropsychological assessment of Alzheimer’s disease. Neurology 39:1159–1165
Muir JL (1997) Acetylcholine, aging, and Alzheimer’s disease. Pharmacol Biochem Behav 56:687–696
Narayanan NS, Rodnitzky RL, Uc EY (2013) Prefrontal dopamine signaling and cognitive symptoms of Parkinson’s disease. Rev Neurosci 24:267–278
Pappas BA, Bayley PJ, Bui BK, Hansen LA, Thal LJ (2000) Choline acetyltransferase activity and cognitive domain scores of Alzheimer’s patients. Neurobiol Aging 21:11–17
Perry EK, Haroutunian V, Davis KL, Levy R, Lantos P, Eagger S, Honavar M, Dean A, Griffiths M, McKeith IG et al (1994) Neocortical cholinergic activities differentiate Lewy body dementia from classical Alzheimer’s disease. NeuroReport 5:747–749
Rolinski M, Fox C, Maidment I, McShane R (2012) Cholinesterase inhibitors for dementia with Lewy bodies, Parkinson’s disease dementia and cognitive impairment in Parkinson’s disease. Cochrane Database Syst Rev 3:CD006504
Sanchez-Cubillo I, Perianez JA, Adrover-Roig D, Rodriguez-Sanchez JM, Rios-Lago M, Tirapu J, Barcelo F (2009) Construct validity of the Trail Making Test: role of task-switching, working memory, inhibition/interference control, and visuomotor abilities. J Int Neuropsychol Soc 15:438–450
Sarter M, Albin RL, Kucinski A, Lustig C (2014) Where attention falls: increased risk of falls from the converging impact of cortical cholinergic and midbrain dopamine loss on striatal function. Exp Neurol 257C:120–129
Scherfler C, Esterhammer R, Nocker M, Mahlknecht P, Stockner H, Warwitz B, Spielberger S, Pinter B, Donnemiller E, Decristoforo C, Virgolini I, Schocke M, Poewe W, Seppi K (2013) Correlation of dopaminergic terminal dysfunction and microstructural abnormalities of the basal ganglia and the olfactory tract in Parkinson’s disease. Brain 136:3028–3037
Shimada H, Hirano S, Shinotoh H, Aotsuka A, Sato K, Tanaka N, Ota T, Asahina M, Fukushi K, Kuwabara S (2009) Mapping of brain acetylcholinesterase alterations in Lewy body disease by PET. Neurology 73:273–278
Shulman KI (2000) Clock-drawing: is it the ideal cognitive screening test? Int J Geriatr Psychiatry 15:548–561
Takahashi R, Ishii K, Miyamoto N, Yoshikawa T, Shimada K, Ohkawa S, Kakigi T, Yokoyama K (2010) Measurement of gray and white matter atrophy in dementia with Lewy bodies using diffeomorphic anatomic registration through exponentiated lie algebra: a comparison with conventional voxel-based morphometry. AJNR Am J Neuroradiol 31:1873–1878
Tam CW, Burton EJ, McKeith IG, Burn DJ, O’Brien JT (2005) Temporal lobe atrophy on MRI in Parkinson disease with dementia: a comparison with Alzheimer disease and dementia with Lewy bodies. Neurology 64:861–865
Tanaka Y, Hanyu H, Sakurai H, Takasaki M, Abe K (2003) Atrophy of the substantia innominata on magnetic resonance imaging predicts response to donepezil treatment in Alzheimer’s disease patients. Dement Geriatr Cogn Disord 16:119–125
Teipel S, Heinsen H, Amaro E Jr, Grinberg LT, Krause B, Grothe M (2014) Cholinergic basal forebrain atrophy predicts amyloid burden in Alzheimer’s disease. Neurobiol Aging 35:482–491
Teipel SJ, Flatz W, Ackl N, Grothe M, Kilimann I, Bokde AL, Grinberg L, Amaro E Jr, Kljajevic V, Alho E, Knels C, Ebert A, Heinsen H, Danek A (2014) Brain atrophy in primary progressive aphasia involves the cholinergic basal forebrain and Ayala’s nucleus. Psychiatry Res 221:187–194
Teipel SJ, Flatz WH, Heinsen H, Bokde AL, Schoenberg SO, Stöckel S, Dietrich O, Reiser MF, Möller H-J, Hampel H (2005) Measurement of basal forebrain atrophy in Alzheimer’s disease using MRI. Brain 128:2626–2644
Teipel SJ, Meindl T, Grinberg L, Grothe M, Cantero JL, Reiser MF, Moller HJ, Heinsen H, Hampel H (2011) The cholinergic system in mild cognitive impairment and Alzheimer’s disease: an in vivo MRI and DTI study. Hum Brain Mapp 32:1349–1362
Tiraboschi P, Hansen LA, Alford M, Merdes A, Masliah E, Thal LJ, Corey-Bloom J (2002) Early and widespread cholinergic losses differentiate dementia with Lewy bodies from Alzheimer disease. Arch Gen Psychiatry 59:946–951
Tiraboschi P, Salmon DP, Hansen LA, Hofstetter RC, Thal LJ, Corey-Bloom J (2006) What best differentiates Lewy body from Alzheimer’s disease in early-stage dementia? Brain 129:729–735
Vogels OJ, Broere CA, ter Laak HJ, ten Donkelaar HJ, Nieuwenhuys R, Schulte BP (1990) Cell loss and shrinkage in the nucleus basalis Meynert complex in Alzheimer’s disease. Neurobiol Aging 11:3–13
Whitehouse PJ, Hedreen JC, White CL, Price DL (1983) Basal forebrain neurons in the dementia of Parkinson disease. Ann Neurol 13:243–248
Whitehouse PJ, Price DL, Clark AW, Coyle JT, DeLong MR (1981) Alzheimer disease: evidence for selective loss of cholinergic neurons in the nucleus basalis. Ann Neurol 10:122–126
Whitwell JL, Weigand SD, Shiung MM, Boeve BF, Ferman TJ, Smith GE, Knopman DS, Petersen RC, Benarroch EE, Josephs KA, Jack CR Jr (2007) Focal atrophy in dementia with Lewy bodies on MRI: a distinct pattern from Alzheimer’s disease. Brain 130:708–719
Wider C, Ross OA, Nishioka K, Heckman MG, Vilarino-Guell C, Jasinska-Myga B, Erketin-Taner N, Rademakers R, Graff-Radford NR, Mash DC, Papapetropoulos S, Duara R, Uchikado H, Wszolek ZK, Farrer MJ, Dickson DW (2012) An evaluation of the impact of MAPT, SNCA and APOE on the burden of Alzheimer’s and Lewy body pathology. J Neurol Neurosurg Psychiatry 83:424–429
Zaborszky L, Csordas A, Mosca K, Kim J, Gielow MR, Vadasz C, Nadasdy Z (2013) Neurons in the basal forebrain project to the cortex in a complex topographic organization that reflects corticocortical connectivity patterns: an experimental study based on retrograde tracing and 3D reconstruction. Cereb Cortex
Zaborszky L, Hoemke L, Mohlberg H, Schleicher A, Amunts K, Zilles K (2008) Stereotaxic probabilistic maps of the magnocellular cell groups in human basal forebrain. Neuroimage 42:1127–1141
Acknowledgments
The study was supported by the Department ‘Aging of the individual and the Society’ of the University of Rostock.
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical standards
All participants gave written informed consent in agreement with the local ethics committee; the study was conducted according to the 1964 Declaration of Helsinki and its later amendments.
Author information
Authors and Affiliations
Corresponding author
Additional information
C. Schuster and M. J. Grothe contributed equally to the manuscript
Rights and permissions
About this article
Cite this article
Grothe, M.J., Schuster, C., Bauer, F. et al. Atrophy of the cholinergic basal forebrain in dementia with Lewy bodies and Alzheimer’s disease dementia. J Neurol 261, 1939–1948 (2014). https://doi.org/10.1007/s00415-014-7439-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-014-7439-z