Abstract
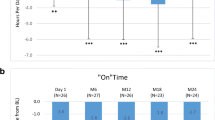
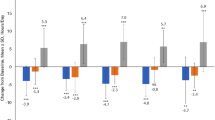
The short-term benefits of levodopa/carbidopa intestinal gel (LCIG) in patients with advanced Parkinson’s disease (PD) are well documented, but the long-term benefits are still uncertain. The aim of this study was to investigate the motor and cognitive outcome of LCIG treatment in advanced PD after a follow-up period of at least 24 months. We assessed 29 patients with advanced PD who started LCIG infusion at our centre between 2007 and 2013. Motor fluctuations, parkinsonian symptoms, activities of daily living and impact on quality of life were evaluated. We also investigated the cognitive outcome using a battery of neuropsychological tests. All adverse events were recorded. Of the 29 PD patients who initiated LCIG, 16 patients reached the follow-up evaluation (24 months), after a mean time period of 32.2 ± 12.4 months. Six patients did not fulfil the 24-month follow-up visit and were evaluated after a mean time period of 8.6 ± 5.4 months. Seven patients discontinued the treatment before the scheduled visit. “Off” time and “On” dyskinesia duration were significantly reduced. LCIG improved quality of life and non motor symptoms, despite overall unchanged total levodopa doses prior to LCIG beginning. Motor and cognitive decline were detected. A relatively high number of adverse events occurred during the follow-up, above all, technical problems with the infusion device and mild problems related with gastrostomy. There were four cases of peripheral neuropathy (PN), 2 of which were considered serious. Our data confirm that LCIG is beneficial in the long-term treatment of advanced PD patients despite a decline in cognitive functions in a subgroup of patients, probably due to disease progression. PN in patients with LCIG may be more frequent than the published date suggest.




Similar content being viewed by others
References
Schrag A, Quinn N (2000) Dyskinesias and motor fluctuations in Parkinson’s disease. A community-based study. Brain 123:2297–2305
Obeso JA, Rodriguez-Oroz M, Marín C et al (2004) The origin of motor fluctuations in Parkinson’s disease: importance of dopaminergic innervation and basal ganglia circuits. Neurology 13:17–30
Castrioto A, Lozano AM (2011) Ten year outcome of subthalamic stimulation in Parkinson disease: a blinded evaluation. Arch Neurol 68:1550–1556
Nilsson D, Nyholm D, Aquilonius SM (2001) Duodenal levodopa infusion in Parkinson’s disease- long-term experience. Acta Neurol Scand 104:343–348
Nyholm D, Nilsson Remahl AI, Dizdar N et al (2005) Duodenal levodopa infusion monotherapy vs oral polypharmacy in advanced Parkinson’s disease. Neurology 25:216–223
Hughes AJ, Daniel SE, Kilford L et al (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinicopathological study of 100 cases. J Neurol Neurosurg Psychiatry 55:181–184
Crum RM, Anthony JC, Bassett SS et al (1993) Population based norms for the Mini-Mental State Examination by age and education level. JAMA 269:2386–2391
Caviness JN, Driver-Dunckley E, Connor DJ et al (2007) Defining mild cognitive impairment in Parkinson’s disease. Mov Disord 15:1272–1277
Zibetti M, Merola A, Ricchi V et al (2013) Long-term duodenal levodopa infusion in Parkinson’s disease: a 3-year motor and cognitive follow-up study. J Neurol 260:101–114
Emre M, Aarsland D, Brown R et al (2007) Clinica diagnostic criteria for dementia associated with Parkinson’s disease. Mov Disord 15:1689–1707
Santos-García D, Sanjurjo LF, Macías M et al (2012) Long-term exposure to duodenal levodopa/carbidopa infusion therapy improves quality of life in relation especially to mobility, activities of daily living, and emotional well-being. Acta Neurol Scand 125:187–191
Nilsson D, Hansson LE, Johansson K et al (1998) Long-term intraduodenal infusion of a water based levodopa–carbidopa dispersion in very advanced Parkinson’s disease. Acta Neurol Scand 97:175–183
Tomlinson CL, Stowe R, Patel S et al (2010) Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Mov Disord 25:2649–2653
Antonini A, Isaias IU, Canesi M et al (2007) Duodenal levodopa infusion for advanced Parkinson’s disease: 12-month treatment outcome. Mov Disord 22:1145–1149
Puente V, De Fabregues O, Oliveras C et al (2010) Eighteen month study of continuous intraduodenal levodopa infusion in patients with advanced Parkinson’s disease: impact on control of fluctuations and quality of life. Parkinsonism Relat Disord 16:218–221
Levy G, Schupf N, Tang MX et al (2002) Combined effect of age and severity on the risk of dementia in Parkinson’s disease. Ann Neurol 51:722–729
Foltynie T, Brayne C, Barker RA (2002) The heterogeneity of idiopathic Parkinson’s disease. J Neurol 249:138–145
Guimarães J, Moura E, Vieira-Coelho MA et al (2012) Weight variation before and after surgery in Parkinson’s disease: a noradrenergic modulation? Mov Disord 27:1078–1082
Toth C, Brown MS, Furtado S et al (2008) Neuropathy as a potential complication of levodopa use in Parkinson’s disease. Mov Disord 23:1850–1859
Santos-García D, de la Fuente-Fernández R, Valldeoriola F et al (2012) Polyneuropathy while on duodenal levodopa infusion in Parkinson’s disease patients: we must be alert. J Neurol 259:1668–1672
Müller T, van Laar T, Cornblath DR et al (2013) Peripheral neuropathy in Parkinson’s disease: levodopa exposure and implications for duodenal delivery. Parkinsonism Relat Disord 19:501–507
Toth C, Breithaupt K, Ge S et al (2010) Levodopa, methylmalonic acid, and neuropathy in idiopathic Parkinson disease. Ann Neurol 68:28–36
Müller T (2008) Role of homocysteine in the treatment of Parkinson’s disease. Expert Rev Neurother 8:957–967
Müller T, Kuhn W (2009) Homocysteine levels after acute levodopa intake in patients with Parkinson’s disease. Mov Disord 24:1339–1343
Ceravolo R, Cossu G, Bandettini di Poggio M et al (2013) Neuropathy and levodopa in Parkinson’s disease: evidence from a multicenter study. Mov Disord 28:1931–1937
Acknowledgments
This work was supported by grants from the Ministerio de Economía y Competitividad de España [SAF2007-60700]; the Instituto de Salud Carlos III [PI10/01674, CP08/00174], the Consejería de Economía, Innovación, Ciencia y Empleo de la Junta de Andalucía [CVI-02526, CTS-7685], the Consejería de Salud y Bienestar Social de la Junta de Andalucía [PI-0377/2007, PI-0741/2010, PI-0437-2012], the Sociedad Andaluza de Neurología, the Jacques and Gloria Gossweiler Foundation, the Fundación Alicia Koplowitz and the Fundación Mutua Madrileña.
Conflicts of interest
María T. Cáceres-Redondo, Fátima Carrillo, María J. Lama and Pablo Mir have received honoraria, payments for consulting, advisory services, speaking services and research from Abbvie Laboratories.
Ismael Huertas-Fernández: none.
Laura Vargas-González: none.
Manuel Carballo: none.
Ethical standard statement
Ethical approval was obtained and all patients, gave informed consent for this study. This investigation was approved by the local ethics committee.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cáceres-Redondo, M.T., Carrillo, F., Lama, M.J. et al. Long-term levodopa/carbidopa intestinal gel in advanced Parkinson’s disease. J Neurol 261, 561–569 (2014). https://doi.org/10.1007/s00415-013-7235-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-013-7235-1