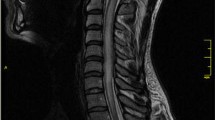
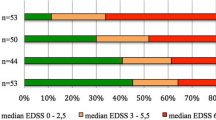
Abstract
The aim of the study was to characterize the demographic, clinical, and prognostic features of Chinese patients with acute transverse myelitis (ATM). The clinical data from ATM patients in a demyelinating disease database were analyzed retrospectively. Sixty-seven ATM patients with a follow-up duration longer than 2 years were identified. The frequency of neuromyelitis optica-related ATM (NMO-ATM) was high in our cohort (40.3%). Recurrent ATM (R-ATM), with a female predominance, was common in total idiopathic ATM (69.0%, 20/29). In R-ATM with longitudinally extensive spinal cord lesions (LESCLs), the high seropositivity of NMO-IgG, spinal cord lesions mostly involved the central gray matter and severer long-term disability were similar to NMO-ATM. In RTM without LESCLs, low seropositivity of NMO-IgG, preferentially involvement of the peripheral white matter and relative better neurological recovery were consistent with multiple sclerosis-related ATM (MS-ATM). The transition rates to MS in patients with acute partial transverse myelitis (APTM) and acute complete transverse myelitis (ACTM) were not significant (16.7 vs. 6.3%, P = 0.753), while LESCLs (OR = 11.4, P = 0.028) were significantly correlated with transition to NMO. The presence of LESCLs was the only variable showing a higher risk for reaching Rankin 3 (hazard ratio: 2.5, 95% CI: 1.0–6.1). Chinese patients with ATM had demographic, clinical, and prognostic features different from those in Western populations. Idiopathic R-ATM, common in Chinese, is a heterogeneous entity that shares partial clinical, spinal MRI and prognostic features with MS-ATM and NMO-ATM. The length of spinal cord lesion, rather than APTM/ACTM, may be a prognostic factor associated with clinical outcome and long-term disability in our population.



Similar content being viewed by others
References
TMCW Group (2002) Proposed diagnostic criteria and nosology of acute transverse myelitis. Neurology 59:499–505
Kahl KG, Naumann M, Oertele E, Warmuth-Metz M, Toyka KV (1998) Recurrent transverse myelitis. 2 cases and review of the literature. Nervenarzt 69:1115–1122
Wingerchuk DM, Hogancamp WF, O’Brien PC, Weinshenker BG (1999) The clinical course of neuromyelitis optica (Devic’s syndrome). Neurology 53:1107–1114
Mok CC, Lee KW, Wong RW, Lau CS (1999) Acute lupus myelitis affecting the clonus medullaris. Clin Exp Rheumatol 17:123–124
Weiss A, Brandl HG (1999) Cervical myelitis in herpes simplex virus (HSV) infections. Dtsch Med Wochenschr 124:781
Dodson D (1990) Transverse myelitis and spastic paraparesis in a patient with HIV infection. N Engl J Med 322:1322
Alvarenga MP, Thuler LC, Neto SP, Vasconcelos CC, Camargo SG, Papais-Alvarenga RM (2010) The clinical course of idiopathic acute transverse myelitis in patients from Rio de Janeiro. J Neurol 257:992–998
Lu Z, Qiu W, Zou Y, Lv K, Long Y, You W, Zheng X, Hu X (2010) Characteristic linear lesions and longitudinally extensive spinal cord lesions in Chinese patients with neuromyelitis optica. J Neurol Sci 293:92–96
Polman CH, Reingold SC, Edan G, Filippi M, Hartung HP, Kappos L, Lublin FD, Metz LM, McFarland HF, O’Connor PW, Sandberg-Wollheim M, Thompson AJ, Weinshenker BG, Wolinsky JS (2005) Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria”. Ann Neurol 58:840–846
Wingerchuk DM, Lennon VA, Pittock SJ, Lucchinetti CF, Weinshenker BG (2006) Revised diagnostic criteria for neuromyelitis optica. Neurology 66:1485–1489
Scott TF, Kassab SL, Singh S (2005) Acute partial transverse myelitis with normal cerebral magnetic resonance imaging: transition rate to clinically definite multiple sclerosis. Mult Scler 11:373–377
Kira J (2008) Neuromyelitis optica and asian phenotype of multiple sclerosis. Ann N Y Acad Sci 1142:58–71
Roberts L, Counsell C (1998) Assessment of clinical outcomes in acute stroke trials. Stroke 29:986–991
de Seze J, Stojkovic T, Breteau G, Lucas C, Michon-Pasturel U, Gauvrit JY, Hachulla E, Mounier-Vehier F, Pruvo JP, Leys D, Destee A, Hatron PY, Vermersch P (2001) Acute myelopathies: clinical, laboratory and outcome profiles in 79 cases. Brain 124:1509–1521
Harzheim M, Schlegel U, Urbach H, Klockgether T, Schmidt S (2004) Discriminatory features of acute transverse myelitis: a retrospective analysis of 45 patients. J Neurol Sci 217:217–223
Jeffery DR, Mandler RN, Davis LE (1993) Transverse myelitis. Retrospective analysis of 33 cases, with differentiation of cases associated with multiple sclerosis and parainfectious events. Arch Neurol 50:532–535
de Seze J, Lanctin C, Lebrun C, Malikova I, Papeix C, Wiertlewski S, Pelletier J, Gout O, Clerc C, Moreau C, Defer G, Edan G, Dubas F, Vermersch P (2005) Idiopathic acute transverse myelitis: application of the recent diagnostic criteria. Neurology 65:1950–1953
Bhat A, Naguwa S, Cheema G, Gershwin ME (2010) The epidemiology of transverse myelitis. Autoimmun Rev 9:A395–A399
al Deeb SM, Yaqub BA, Bruyn GW, Biary NM (1997) Acute transverse myelitis. A localized form of postinfectious encephalomyelitis. Brain 120:1115–1122
Berman M, Feldman S, Alter M, Zilber N, Kahana E (1981) Acute transverse myelitis: incidence and etiologic considerations. Neurology 31:966–971
Christensen PB, Wermuth L, Hinge HH, Bomers K (1990) Clinical course and long-term prognosis of acute transverse myelopathy. Acta Neurol Scand 81:431–435
Prabhakar S, Syal P, Singh P, Lal V, Khandelwal N, Das CP (1999) Non-compressive myelopathy: clinical and radiological study. Neurol India 47:294–299
Andronikou S, Albuquerque-Jonathan G, Wilmshurst J, Hewlett R (2003) MRI findings in acute idiopathic transverse myelopathy in children. Pediatr Radiol 33:624–629
Choi KH, Lee KS, Chung SO, Park JM, Kim YJ, Kim HS, Shinn KS (1996) Idiopathic transverse myelitis: MR characteristics. AJNR Am J Neuroradiol 17:1151–1160
Kim KK (2003) Idiopathic recurrent transverse myelitis. Arch Neurol 60:1290–1294
Kim SH, Kim SM, Vincent A, Ahn SW, Hong YH, Park KS, Sung JJ, Lee KW (2010) Clinical characteristics, prognosis, and seropositivity to the anti-aquaporin-4 antibody in Korean patients with longitudinally extensive transverse myelitis. J Neurol 257:920–925
Ravaglia S, Bastianello S, Franciotta D, Ceroni M, Pichiecchio A, Tavazzi E, Moglia A, Marchioni E (2009) NMO-IgG-negative relapsing myelitis. Spinal Cord 47:531–537
Qiu W, Wu JS, Zhang MN, Matsushita T, Kira JI, Carroll WM, Mastaglia FL, Kermode AG (2010) Longitudinally extensive myelopathy in Caucasians: a West Australian study of 26 cases from the Perth Demyelinating Diseases Database. J Neurol Neurosurg Psychiatry 81:209–212
Wingerchuk DM, Lennon VA, Lucchinetti CF, Pittock SJ, Weinshenker BG (2007) The spectrum of neuromyelitis optica. Lancet Neurol 6:805–815
Seifert T, Enzinger C, Ropele S, Storch MK, Strasser-Fuchs S, Fazekas F (2005) Relapsing acute transverse myelitis: a specific entity. Eur J Neurol 12:681–684
Pandit L, Rao S (1996) Recurrent myelitis. J Neurol Neurosurg Psychiatry 60:336–338
Scott TF, Bhagavatula K, Snyder PJ, Chieffe C (1998) Transverse myelitis. Comparison with spinal cord presentations of multiple sclerosis. Neurology 50:429–433
Osoegawa M, Kira J, Fukazawa T, Fujihara K, Kikuchi S, Matsui M, Kohriyama T, Sobue G, Yamamura T, Itoyama Y, Saida T, Sakata K, Ochi H, Matsuoka T (2009) Temporal changes and geographical differences in multiple sclerosis phenotypes in Japanese: nationwide survey results over 30 years. Mult Scler 15:159–173
Chan KH, Tsang KL, Fong GC, Ho SL, Cheung RT, Mak W (2006) Idiopathic inflammatory demyelinating disorders after acute transverse myelitis. Eur J Neurol 13:862–868
Chan KH, Tsang KL, Fong GC, Cheung RT, Ho SL (2005) Idiopathic severe recurrent transverse myelitis: a restricted variant of neuromyelitis optica. Clin Neurol Neurosurg 107:132–135
Gajofatto A, Monaco S, Fiorini M, Zanusso G, Vedovello M, Rossi F, Turatti M, Benedetti MD (2010) Assessment of outcome predictors in first-episode acute myelitis: a retrospective study of 53 cases. Arch Neurol 67:724–730
Rubiera M, Rio J, Tintore M, Nos C, Rovira A, Tellez N, Montalban X (2006) Neuromyelitis optica diagnosis in clinically isolated syndromes suggestive of multiple sclerosis. Neurology 66:1568–1570
Barkhof F, Filippi M, Miller DH, Scheltens P, Campi A, Polman CH, Comi G, Ader HJ, Losseff N, Valk J (1997) Comparison of MRI criteria at first presentation to predict conversion to clinically definite multiple sclerosis. Brain 120(Pt 11):2059–2069
Tintore M, Rovira A, Martinez MJ, Rio J, Diaz-Villoslada P, Brieva L, Borras C, Grive E, Capellades J, Montalban X (2000) Isolated demyelinating syndromes: comparison of different MR imaging criteria to predict conversion to clinically definite multiple sclerosis. AJNR Am J Neuroradiol 21:702–706
Cordonnier C, de Seze J, Breteau G, Ferriby D, Michelin E, Stojkovic T, Pruvo JP, Vermersch P (2003) Prospective study of patients presenting with acute partial transverse myelopathy. J Neurol 250:1447–1452
Sellner J, Luthi N, Buhler R, Gebhardt A, Findling O, Greeve I, Mattle HP (2008) Acute partial transverse myelitis: risk factors for conversion to multiple sclerosis. Eur J Neurol 15:398–405
Scott TF, Kassab SL, Pittock SJ (2006) Neuromyelitis optica IgG status in acute partial transverse myelitis. Arch Neurol 63:1398–1400
Acknowledgments
The study was supported by Technology Project of Guangzhou City (No. 2060402).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
R. Li and W. Qiu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Li, R., Qiu, W., Lu, Z. et al. Acute transverse myelitis in demyelinating diseases among the Chinese. J Neurol 258, 2206–2213 (2011). https://doi.org/10.1007/s00415-011-6093-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-011-6093-y