Abstract
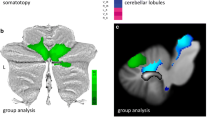
Friedreich’s ataxia (FA) is the most frequent autosomal recessive ataxia and essentially considered a disease of the dorsal root ganglia and spinal cord. It is caused by homozygous GAA expansions in the Frataxin gene in most cases. Although only a few studies have addressed cerebral involvement in FA, cognitive symptoms have lately been emphasized. To evaluate brain damage in vivo, we employed whole-brain VBM and analysis of pre-defined regions of interest (ROIs) over the cerebellum to compare 24 patients with 24 age-and-sex-matched normal controls. 1H-MRS of deep cerebral white matter (WM) was subsequently performed. Mean age of patients was 28 years (range 14–45), mean duration of disease was 14 years (range 5–28) and 11 were men. Mean length of shorter (GAA1) and longer (GAA2) alleles were 735 and 863, respectively. VBM analysis identified WM atrophy in the posterior cyngulate gyrus, paracentral lobule and middle frontal gyrus. ROIs over the infero-medial cerebellar hemispheres and dorsal brainstem presented gray matter atrophy, which correlated with duration of disease (r = −0.4). NAA/Cr ratios were smaller among patients (P = 0.006), but not Cho/Cr (P = 0.08). Our results provide evidence of axonal damage in the cerebellum, brainstem and subcortical WM in FA. This suggests that neuronal dysfunction is more widespread than previously thought in FA.



Similar content being viewed by others
References
Appenzeller S, Li LM, Costallat LT, Cendes F (2005) Evidence of reversible axonal dysfunction in systemic lupus erythematosus: a proton MRS study. Brain 128:2933–2940
Ashburner J, Friston KJ (2000) Voxel-based morphometry—the methods. Neuroimage 11:805–821
Babady NE, Carelle N, Wells RD et al (2007) Advancements in the pathophysiology of Friedreich’s Ataxia and new prospects for treatments. Mol Genet Metab 92:23–35
Bonilha L, Rorden C, Appenzeller S, Coan AC, Cendes F, Li LM (2006) Gray matter Atrophy associated with duration of temporal lobe epilepsy. Neuroimage 32:1070–1079
Bottomley PA (1987) Spatial localization in NMR spectroscopy in vivo. Ann NY Acad Sci 508:333–348
Brenneis C, Boesch SM, Egger KE et al (2006) Cortical atrophy in the cerebellar variant of multiple system atrophy: a voxel-based morphometry study. Mov Disord 21:159–165
Campuzano V, Montermini L, Molto MD et al (1996) Friedreich’s ataxia: autosomal recessive disease caused by an intronic GAA triplet repeat expansion. Science 271:1423–1427
Corben LA, Georgiou-Karistianis N, Fahey MC et al (2006) Towards an understanding of cognitive function in Friedreich ataxia. Brain Res Bull 70:197–202
Della Nave R, Ginestroni A, Giannelli M et al (2008) Brain structural damage in Friedreich ataxia. J Neurol Neurosurg Psychiatry 79:82–85
Della Nave R, Ginestroni A, Tessa C et al (2008) Brain white matter tracts degeneration in Friedreich ataxia an in vivo MRI study using tract-based spatial statistics and voxel-based morphometry. Neuroimage 40:19–25
De Michele G, Di Salle F, Filla A et al (1995) Magnetic resonance imaging in “typical” and “late onset” Friedreich’s disease and early onset cerebellar ataxia with retained tendon reflexes. Ital J Neurol Sci 16:303–308
Durr A, Cossee M, Agid Y et al (1996) Clinical and genetic abnormalities in patients with Friedreich’s ataxia. N Engl J Med 335:1169–1175
Eder K, Kish SJ, Kirchgessner M, Ross BM (1998) Brain phospholipids and fatty acids in Friedreich’s ataxia and spinocerebellar atrophy type-1. Mov Disord 13:813–819
Fogel BL, Perlman S (2007) Clinical features and molecular genetics of autosomal recessive cerebellar ataxias. Lancet Neurol 6:245–257
Genovese CR, Lazar NA, Nichols T (2002) Thresholding of statistical maps in functional neuroimaging using the false discovery rate. Neuroimage 15:870–878
Good CD, Johnsrude IS, Ashburner J, Henson RNA, Friston KJ, Frackowiak RSJ (2001) A voxel-based morphometric study of ageing in 465 normal adult human brains. Neuroimage 14:21–36
Jiralerspong S, Liu Y, Montermini L, Stifani S, Pandolfo M (1997) Frataxin shows developmentally regulated tissue-specific expression in the mouse embryo. Neurobiol Dis 4:103–113
Junck L, Gilman S, Gebarski SS, Koeppe RA, Kluin KJ, Markel DS (1994) Structural and functional brain imaging in Friedreich’s ataxia. Arch Neurol 51:349–355
Lamarche JB, Lemieux B, Lieu HB (1984) The neuropathology of “typical” Friedreich’s ataxia in Quebec. Can J Neurol Sci 11:592–600
Lasek K, Lencer R, Gaser C et al (2006) Morphological basis for the spectrum of clinical deficits in spinocerebellar ataxia 17 (SCA17). Brain 129:2341–2352
Mantovan MC, Martinuzzi A, Squarzanti F et al (2006) Exploring mental status in Friedreich’s ataxia: a combined neuropsychological, behavioral and neuroimaging study. Eur J Neurol 13:827–835
Mascalchi M, Salvi F, Piacentini S, Bartolozzi C (1994) Friedreich’s ataxia: MR findings involving the cervical portion of the spinal cord. AJR Am J Roentgenol 163:187–191
Moffett JR, Ross B, Arun P, Madhavarao CN, Namboodiri AM (2007) N-Acetylaspartate in the CNS: from neurodiagnostics to neurobiology. Prog Neurobiol 8:89–131
Oppenheimer DR (1979) Brain lesions in Friedreich’s ataxia. Can J Neurol Sci 6:173–176
Ormerod IE, Harding AE, Miller DH et al (1994) Magnetic resonance imaging in degenerative ataxic disorders. J Neurol Neurosurg Psychiatry 57:51–57
Rudkin TM, Arnold DL (1999) Proton magnetic resonance spectroscopy for the diagnosis and management of cerebral disorders. Arch Neurol 56:919–926
Ruocco HH, Bonilha L, Li LM, Lopes-Cendes I, Cendes F (2008) Longitudinal analysis of regional gray matter loss in Huntington disease: effects of the length of the CAG repeat. J Neurol Neurosurg Psychiatry 79:130–135
Subramony SH, May W, Lynch D et al (2005) Cooperative Ataxia Group measuring Friedreich ataxia: interrater reliability of a neurologic rating scale. Neurology 64:1261–1262
Tzourio-Mazoyer N, Landeau B, Papathanassiou D et al (2002) Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 15:273–289
Wessel K, Schroth G, Diener HC, Muller-Forell W, Dichgans J (1989) Significance of MRI-confirmed atrophy of the cranial spinal cord in Friedreich’s ataxia. Eur Arch Psychiatry Neurol Sci 238:225–230
White M, Lalonde R, Botez-Marquard T (2000) Neuropsychologic and neuropsychiatric characteristics of patients with Friedreich’s ataxia. Acta Neurol Scand 102:222–226
Wullner U, Klockgether T, Petersen D, Naegele T, Dichgans J (1993) Magnetic resonance imaging in hereditary and idiopathic ataxia. Neurology 43:318–325
Acknowledgments
Supported by a grant from Fundação de Amparo à Pesquisa do Estado de São Paulo – FAPESP, São Paulo, Brazil, Grant # 04/13725-4.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
França, M.C., D’Abreu, A., Yasuda, C.L. et al. A combined voxel-based morphometry and 1H-MRS study in patients with Friedreich’s ataxia. J Neurol 256, 1114–1120 (2009). https://doi.org/10.1007/s00415-009-5079-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-009-5079-5