Abstract
The XX/XY system is the rule among mammals. However, many exceptions from this general pattern have been discovered since the last decades. One of these non-conventional sex chromosome mechanisms is the multiple sex chromosome system, which is evolutionary fixed among many bat species of the family Phyllostomidae, and has arisen by a translocation between one original gonosome (X or Y chromosome), and an autosome, giving rise to a “neo-XY body.” The aim of this work is to study the synaptic behavior and the chromatin remodeling of multiple sex chromosomes in different species of phyllostomid bats using electron microscopy and molecular markers. Testicular tissues from adult males of the species Artibeus lituratus, Artibeus planirostris, Uroderma bilobatum, and Vampyrodes caraccioli from the eastern Amazonia were analyzed by optical/electron microscopy and immunofluorescence of meiotic proteins involved in synapsis (SYCP3 and SYCE3), sister-chromatid cohesion (SMC3), and chromatin silencing (BRCA1, γ-H2AX, and RNApol 2). The presence of asynaptic axes—labeled by BRCA1 and γ-H2AX—at meiotic prophase in testes that have a normal development of spermatogenesis, suggests that the basic mechanism that arrests spreading of transcriptional silencing (meiotic sex chromosome inactivation (MSCI)) to the autosomal segments may be per se the formation of a functional synaptonemal complex between homologous or non-homologous regions, and thus, this SC barrier might be probably related to the preservation of fertility in these systems.




Similar content being viewed by others
References
Baker RJ (1973) Comparative cytogenetics of the New World leaf-nosed bats (Phyllostomatidae). Periodicumbiologorum 75:37–45
Baker RJ, Bass RA, Johnson MA (1979) Evolutionary implications of chromosomal homology in four genera of stenodermine bats (Phyllostomatidae, Chiroptera). Evolution 33:220–226
Beçak ML, Batistic RF, Vizotto LD, Beçak W (1969) Sex determining mechanism XY 1 Y 2 in Artibeus lituratus (Chiroptera, Phyllostomidae). Experientia 25:81–83
Burgoyne PS, Mahadevaiah SK, Turner JM (2009) The consequences of asynapsis for mammalian meiosis. Nat Rev Genet 10:207–216
Fernandez-Capetillo O, Mahadevaiah SK, Celeste A, Romanienko PJ, Camerini-Otero RD, Bonner WM, Manova K, Burgoyne P, Nussenzweig A (2003) H2AX is required for chromatin remodeling and inactivation of sex chromosomes in male mouse meiosis. Dev Cell 4:497–508
Franco MJ, Sciurano RB, Solari AJ (2007) Protein immunolocalization supports the presence of identical mechanisms of XY body formation in eutherians and marsupials. Chromosom Res 15:815–824
Fredga K (1970) Unusual sex chromosome inheritance in mammals. Philos Trans R Soc Lond B Biol Sci 259(828):15–36
Gomes AJB (2014) Evolução cromossômica e filogenia em morcegos pertencentes a família Phyllostomidae (Mammalia, Chiroptera). Ph.D thesis, Universidade Federal do Pará
Graves JA, Wakefield MJ, Toder R (1998) The origin and evolution of the pseudoautosomal regions of human sex chromosomes. Hum Mol Genet 7(13):1991–6
Handel MA (2004) The XY body: a specialized meiotic chromatin domain. Exp Cell Res 296:57–63
Homolka D, Jansa P, Forejt J (2012) Genetically enhanced asynapsis of autosomal chromatin promotes transcriptional dysregulation and meiotic failure. Chromosoma 121(1):91–104
Hsu T, Baker RJ, Utakoji T (1968) The multiple sex chromosome system of American leaf-nosed bats (Chiroptera, Phyllostomatidae). Cytogenetics 7:27–38
Ichijima Y, Sin HS, Namekawa SH (2012) Sex chromosome inactivation in germ cells: emerging roles of DNA damage response pathways. Cell Mol Life Sci 69(15):2559–72
Jaafar H, Gabriel-Robez O, Rumpler Y (1989) Pattern of ribonucleic acid synthesis in vitro in primary spermatocytes from mouse testis carrying an X-autosome translocation. Chromosoma 98:330–334
Kasahara S, Dutrillaux B (1983) Chromosome banding patterns of four species of bats, with special reference to a case of X-autosome translocation. Ann Genet 26:197–201
Khalil AM, Wahlestedt C (2008) Epigenetic mechanisms of gene regulation during mammalian spermatogenesis. Epigenetics 3(1):21–8
Kingswood SC, Kumamoto AT, Sudman PD, Fletcher KC, Greenbaum IF (1994) Meiosis in chromosomally heteromorphic goitered gazelle, Gazellasubgutturosa (Artiodactyla, Bovidae). Chromosom Res 2(1):37–6
Lemos-Pinto MMP, Calixto MS, Souza MJ, Araújo APT, Langguth A, Santos N (2012) Cytotaxonomy of the subgenus Artibeus (Phyllostomidae, Chiroptera) by characterization of species-specific markers. Comp Cytogenet 6(1):17–28
Martin RH (2008) Cytogenetic determinants of male fertility. Hum Reprod Update 14:379–390
Monesi V (1965) Differential rate of ribonucleic acid synthesis in the autosomes and sex chromosomes during male meiosis in the mouse. Chromosoma 17(1):11–21
Moreira da Silva A, Marques-Aguiar SA, Souza Barros RM, Nagamachi CY, Pieczarka JC (2005) Comparative cytogenetic analysis in the species Uroderma magnirostrum and U. bilobatum (cytotype 2n = 42) (Phyllostomidae, Stenodermatinae) in the Brazilian Amazon. Genet Mol Biol 28(2):248–253
Namekawa SH, Park PJ, Zhang LF, Shima JE, McCarrey JR et al (2006) Postmeiotic sex chromatin in the male germline of mice. Curr Biol 16:660–667
Noronha RCR, Nagamachi CY, Pieczarka JC, Marques-Aguiar S, Barros RMS (2001) Sex-autosome translocations: meiotic behavior suggests an inactivation block with permanence of autosomal gene activity in Phyllostomid bats. Caryologia 3:267–277
Noronha RCR, Nagamachi CY, Pieczarka JC, Marques-Aguiar S, Barros RMS (2004) Meiotic analyses of the sex chromosomes in Carolliinae-Phyllostomidae (Chiroptera): NOR separates the XY 1 Y 2 into two independent parts. Caryologia 57:1–9
Noronha RCR, Nagamachi CY, O’Brien PC, Ferguson-Smith MA, Pieczarka JC (2009) Neo-XY body: an analysis of XY 1 Y 2 meiotic behavior in Carollia (Chiroptera, Phyllostomidae) by chromosome painting. Cytogenet Genome Res 124:37–43
Noronha RC, Nagamachi CY, O’Brien PC, Ferguson-Smith MA, Pieczarka JC (2010) Meiotic analysis of XX/XY and neo-XX/XY sex chromosomes in Phyllostomidae by cross-species chromosome painting revealing a common chromosome 15-XY rearrangement in Stenodermatinae. Chromosom Res 18(6):667–76
Pigozzi MI, Solari AJ (1994) Regular segregation of the XY 1 Y 2 trivalent and univalency in male meiosis of Artibeus lituratus (Phyllostomidae, Chiroptera). Biocell 18(1):41–47
Pinton A, Calgaro A, Bonnet N, Ferchaud S, Billoux S, Dudez AM et al (2009) Influence of sex on the meiotic segregation of a t(13;17) Robertsonian translocation: a case study in the pig. Hum Reprod 24:2034–2043
Ratomponirina C, Viegas-Péquignot E, Dutrillaux B, Petter F, Rumpler Y (1986) Synaptonemal complexes in Gerbillidae: probable role of intercalated heterochromatin in gonosome-autosome translocations. Cytogenet Cell Genet 43(3-4):161–7
Russell LB (1963) Mammalian X-chromosome action: inactivation limited in spread and in region of origin. Science 140:976
Sciurano RB, Solari AJ (2014) Ultraestructural and immunofluorescent methods for the study of XY body as a biomarker. In: Stockert, Espada, Blázquez-Castro (eds) Functional analysis of DNA and chromatin, Methods in Molecular Biology, Chapter 11, vol 2094. Springer Science + Bussiness Media, NY, pp 137–149
Sciurano RB, Rahn MI, Rey Valzacchi G, Solari AJ (2007) The asynaptic chromatin in spermatocytes of translocation carriers contains the histone variant γ-H2AX and associates with the XY body. Hum Reprod 22:142–150
Sciurano RB, Rahn IM, Rey-Valzacchi G, Coco R, Solari AJ (2012a) The role of asynapsis in human spermatocyte failure. Int J Androl 35(4):541–549
Sciurano RB, Rahn IM, Rossi L, Luaces JP, Merani MS, Solari AJ (2012b) Synapsis, recombination and chromatin remodeling in the XY body of armadillos. Chromosom Res 20:293–302
Sciurano RB, Rahn MI, Cavicchia JC, Solari AJ (2013) Dissociation of the X chromosome from the synaptonemal complex in the XY body of the rodent Galea musteloides. Chromosom Res 21(8):753–63
Sharp P (1982) Sex chromosome pairing during male meiosis in marsupials. Chromosoma 86(1):27–47
Solari AJ (1971) The behaviour of chromosomal axes in Searle’s X-autosome translocation. Chromosoma 34:99–112
Solari AJ (1994) Sex chromosomes and sex determination in vertebrates. CRC, Florida
Solari AJ, Pigozzi MI (1994) Fine structure of the XY body in the XY 1 Y 2 trivalent of the bat Artibeus lituratus. Chromosom Res 2:53–58
Solari AJ, Rahn MI (2005) Fine structure and meiotic behaviour of the male multiple sex chromosomes in the genus Alouatta. Cytogenet Genome Res 108(1-3):262–7
Tucker PK, Bickham JW (1986) Sex chromosome-autosome translocation in the leaf nosed bats. II. Meiotic analysis of the subfamilies Sternodermatinae and Phyllostominae. Cytogenet Cell Genet 43:28–27
Turner JM (2007) Meiotic sex chromosome inactivation. Development 134(10):1823–1831
Turner JM, Aprelikova O, Xu X, Wang R, Kim S, Chandramouli GV, Barrett JC, Burgoyne PS, Deng CX (2004) BRCA1, histone H2AX phosphorylation, and male meiotic sex chromosome inactivation. Curr Biol 14:2135–2142
Turner JMA, Mahadevaiah SK, Fernandez-Capetillo O, Nussenzweig A, Xu X, Deng C, Burgoyne PS (2005) Silencing of unsynapsed meiotic chromosomes in the mouse. Nat Genet 37:41–47
Turner JM, Mahadevaiah SK, Ellis PJ, Mitchell MJ, Burgoyne PS (2006) Pachytene asynapsis drives meiotic sex chromosome inactivation and leads to substantial postmeiotic repression in spermatids. Dev Cell 10:521–529
Van Assche E, Bonduelle M, Tournaye H, Joris H, Verheyen G, Debroey P, Van Steirteghem A, Liebaers I (1996) Cytogenetics of infertile men. Hum Reprod 11(Suppl 4):1–26
Villagómez DA, Pinton A (2008) Chromosomal abnormalities, meiotic behavior and fertility in domestic animals. Cytogenet Genome Res 120(1-2):69–80
Yonenaga Y, Frota-Pessoa O, Lewis KR (1969) Karyotypes of seven species of Brazilian bats. Caryologia 22:63–78
Zamudio NM, Chong S, O’Bryan MK (2008) Epigenetic regulation in male germ cells. Reproduction 136(2):131–46
Acknowledgments
We specially thank MSSc. C. Deparci for her excellent assistance in EM techniques (UBA, Buenos Aires, Argentina), MSc. T.F.A. Ribas and Dr. A. J.B. Gomes (UFPA, Belem, Brazil) for capturing and handling animals, and Mg. Angie Barajas Ruiz (UBA, Buenos Aires, Argentina) for the technical assistance. We are also very grateful to Prof. M. Alsheimer (University of Würzburg, Germany) for the generous gift of SYCE3 antibody.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by the Consejo Nacional de Investigaciones Científicas y Técnicas, CONICET (grant no. PIP11220120100683 to RBS and AJS), the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), and the Fundação de Amparo à Pesquisa do Estado do Pará (FAPESPA) (Edital BIONORTE—grant no. Proc 552032/2010-7 to CYN and Edital Vale—grant no. Proc 2010/110447 to JCP).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
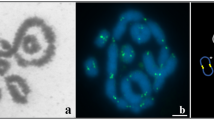
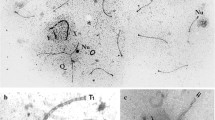
The presence of multiple sex chromosomes in phyllostomid bats. a An electron micrograph of spermatocyte spread staining by phosphotungstic acid shows the presence of one recombination nodule (RN) in the long synaptonemal complex (SC) formed between the X and Y 2 chromosomes in Artibeus lituratus during pachytene stage (a RN in the shorter SC is not visible). k kinetochore. b–f The fluorescent immunolocalization of protein markers of synapsis and chromatin remodeling on pachytene spermatocyte spreads of the four analyzed species of phyllostomid bats reveal that (1) the cohesin SMC3 (b and d–f) and the SYCP3 protein of the lateral element of SC (c, red axes) are present on the single-chromosome axes in autosomes, as well as in multiple sex chromosomes; (2) the SYCE3 protein, a marker of the central element of SC, is located on synapsed regions (b, green axes); (3) the asynaptic axes of the sexual chromosomes are labeled by the DNA damage repair protein, BRCA1 (d, green axes), and the corresponding chromatin, by the phosphorylated variant histone, γ-H2AX (e, red area), indicating that these regions are transcriptionally silenced, as shown by the depletion of RNApol2 (f, asterisk). b–f Multiple sex chromosomes, white arrows; b CREST, kinetochores. Al, Artibeus lituratus; Ap, A. planirostris; Ub, Uroderma bilobatum; Vc, Vampyrodes caraccioli. Bars a 1 μm and b–f 3 μm. (GIF 192 kb)
Rights and permissions
About this article
Cite this article
Rahn, M.I., Noronha, R.C., Nagamachi, C.Y. et al. Protein markers of synaptic behavior and chromatin remodeling of the neo-XY body in phyllostomid bats. Chromosoma 125, 701–708 (2016). https://doi.org/10.1007/s00412-015-0566-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-015-0566-1