Abstract
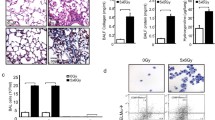
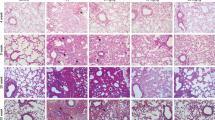
While radiation-induced lung injury (RILI) is known to be progressed by Th2 skewed, pro-inflammatory immune response, there have been few therapeutic attempts through Th1 immune modulation. We investigated whether the immunostimulant CpG-oligodeoxynucleotide (CpG-ODN) would be effective against RILI by way of measuring reactive oxygen species (ROS) and nitric oxides (NO), histopathology, micro-three-dimensional computer tomography (CT), and cytokine profiling. We found that KSK CpG-ODN (K-CpG) significantly reduced histopathological fibrosis when compared to the positive control (PC) group (p < 0.01). The levels of ROS production in serum and splenocyte of PC group were significantly higher than that of K-CpG group (p < 0.01). The production of nitric oxide (NO) in CpG-ODNs group was higher than that of PC group. Last, cytokine profiling illustrated that the protein concentrations of Th1-type cytokines such as IL-12 and TNF-α as well as Th2-type cytokine IL-5 in K-CpG group inclined to be significantly (p < 0.001 or p < 0.01) higher than those of in PC group. Collectively, our study clearly indicates that K-CpG is effective against RILI in mice by modulating the innate immune response. To our knowledge, this is the first note on anti-RILI effect of human type, K-CpG, clinically implying the potential of immunotherapy for RILI control.






Similar content being viewed by others
References
Brickey WJ, Neuringer IP, Walton W, Hua X, Wang EY, Jha S, Sempowski GD, Yang X, Kirby SL, Tilley SL, Ting JP (2012) MyD88 provides a protective role in long-term radiation-induced lung injury. Int J Radiat Biol 88(4):335–347. https://doi.org/10.3109/09553002.2012.652723
Brush J, Lipnick SL, Phillips T, Sitko J, McDonald JT, McBride WH (2007) Molecular mechanisms of late normal tissue injury. Semin Radiat Oncol 17(2):121–130. https://doi.org/10.1016/j.semradonc.2006.11.008
Burdelya LG, Krivokrysenko VI, Tallant TC, Strom E, Gleiberman AS, Gupta D, Kurnasov OV, Fort FL, Osterman AL, DiDonato JA, Feinstein E, Gudkov AV (2008) An agonist of toll-like receptor 5 has radioprotective activity in mouse and primate models. Science 320(5873):226–230. https://doi.org/10.1126/science.1154986
Chen J, Wang YC, Mei ZJ, Zhang SM, Yang J, Li X, Yao Y, Xie CH (2016) Radiation-induced lung fibrosis in a tumor-bearing mouse model is associated with enhanced Type-2 immunity. J Radiat Res 57(2):133–141. https://doi.org/10.1093/jrr/rrv077
Cho HC, Kim BH, Kim K, Park JY, Chang JH, Kim SK (2008) Cancer immunotherapeutic effects of novel CpG ODN in murine tumor model. Int Immunopharmacol 8(10):1401–1407. https://doi.org/10.1016/j.intimp.2008.05.010
Ding NH, Li JJ, Sun LQ (2013) Molecular mechanisms and treatment of radiation-induced lung fibrosis. Curr Drug Targets 14(11):1347–1356
Han G, Zhang H, Xie CH, Zhou YF (2011) Th2-like immune response in radiation-induced lung fibrosis. Oncol Rep 26(2):383–388. https://doi.org/10.3892/or.2011.1300
Hayashi T, Raz E (2006) TLR9-based immunotherapy for allergic disease. Am J Med. https://doi.org/10.1016/j.amjmed.2005.12.028
Hubner RH, Gitter W, El Mokhtari NE, Mathiak M, Both M, Bolte H, Freitag-Wolf S, Bewig B (2008) Standardized quantification of pulmonary fibrosis in histological samples. Biotechniques 44(4):507–511. https://doi.org/10.2144/000112729
Ignacio RM, Kim CS, Kim YD, Lee HM, Qi XF, Kim SK (2015) Therapeutic effect of Active Hexose-Correlated Compound (AHCC) combined with CpG-ODN (oligodeoxynucleotide) in B16 melanoma murine model. Cytokine 76(2):131–137. https://doi.org/10.1016/j.cyto.2015.06.002
Janot L, Sirard JC, Secher T, Noulin N, Fick L, Akira S, Uematsu S, Didierlaurent A, Hussell T, Ryffel B, Erard F (2009) Radioresistant cells expressing TLR5 control the respiratory epithelium's innate immune responses to flagellin. Eur J Immunol 39(6):1587–1596. https://doi.org/10.1002/eji.200838907
Krieg AM (2008) Toll-like receptor 9 (TLR9) agonists in the treatment of cancer. Oncogene 27(2):161–167. https://doi.org/10.1038/sj.onc.1210911
Li X, Xu GX, Qiao TK, Yuan SJ, Zhuang XB (2016) Effects of CpG oligodeoxynucleotide 1826 on acute radiation-induced lung injury in mice. Biol Res. https://doi.org/10.1186/s40659-016-0068-5
Mah K, Van Dyk J, Keane T, Poon PY (1987) Acute radiation-induced pulmonary damage: a clinical study on the response to fractionated radiation therapy. Int J Radiat Oncol Biol Phys 13(2):179–188
McDonald S, Rubin P, Phillips TL, Marks LB (1995) Injury to the lung from cancer therapy: clinical syndromes, measurable endpoints, and potential scoring systems. Int J Radiat Oncol Biol Phys 31(5):1187–1203. https://doi.org/10.1016/0360-3016(94)00429-O
Mingomataj EC, Bakiri AH (2016) Regulator Versus Effector Paradigm: Interleukin-10 as Indicator of the Switching Response. Clin Rev Allerg Immu 50(1):97–113. https://doi.org/10.1007/s12016-015-8514-7
Park SJ, Cho HC, Bae KS, Kim SK (2007) Immunoadjuvanticity of novel CpG ODN (Oligodeoxynucleotide). Mol Cell Toxicol 3(1):46–52
Rube CE, Uthe D, Wilfert F, Ludwig D, Yang KY, Konig J, Palm J, Schuck A, Willich N, Remberger K, Rube C (2005) The bronchiolar epithelium as a prominent source of pro-inflammatory cytokines after lung irradiation. Int J Radiat Oncol 61(5):1482–1492. https://doi.org/10.1016/j.ijrobp.2004.12.072
Saito-Fujita T, Iwakawa M, Nakamura E, Nakawatari M, Fujita H, Moritake T, Imai T (2011) Attenuated lung fibrosis in interleukin 6 knock-out mice after C-ion irradiation to lung. J Radiat Res 52(3):270–277. https://doi.org/10.1269/jrr.10094
Sanders CJ, Moore DA, Williams IR, Gewirtz AT (2008) Both radioresistant and hemopoietic cells promote innate and adaptive immune responses to flagellin. J Immunol 181(4):2933. https://doi.org/10.4049/jimmunol.181.4.2933
Takemura N, Kawasaki T, Kunisawa J, Sato S, Lamichhane A, Kobiyama K, Aoshi T, Ito J, Mizuguchi K, Karuppuchamy T, Matsunaga K, Miyatake S, Mori N, Tsujimura T, Satoh T, Kumagai Y, Kawai T, Standley DM, Ishii KJ, Kiyono H, Akira S, Uematsu S (2014) Blockade of TLR3 protects mice from lethal radiation-induced gastrointestinal syndrome. Nat Commun. https://doi.org/10.1038/ncomms4492
Travis EL, Harley RA, Fenn JO, Klobukowski CJ, Hargrove HB (1977) Pathologic changes in the lung following single and multi-fraction irradiation. Int J Radiat Oncol Biol Phys 2(5–6):475–490
Tsoutsou PG, Koukourakis MI (2006) Radiation pneumonitis and fibrosis: mechanisms underlying its pathogenesis and implications for future research. Int J Radiat Oncol Biol Phys 66(5):1281–1293. https://doi.org/10.1016/j.ijrobp.2006.08.058
Volpi C, Fallarino F, Pallotta MT, Bianchi R, Vacca C, Belladonna ML, Orabona C, De Luca A, Boon L, Romani L, Grohmann U, Puccetti P (2013) High doses of CpG oligodeoxynucleotides stimulate a tolerogenic TLR9-TRIF pathway. Nat Commun. https://doi.org/10.1038/ncomms2874
Vujaskovic Z, Marks LB, Anscher MS (2000) The physical parameters and molecular events associated with radiation-induced lung toxicity. Semin Radiat Oncol 10(4):296–307
Ward PA, Hunninghake GW (1998) Lung inflammation and fibrosis. Am J Respir Crit Care Med 157(4 Pt 2):S123–129. https://doi.org/10.1164/ajrccm.157.4.nhlbi-10
Westbury CB, Haviland J, Davies S, Gothard L, Abdi BA, Sydenham M, Bowen J, Stratton R, Short SC, Yarnold JR (2014) Cytokine levels as biomarkers of radiation fibrosis in patients treated with breast radiotherapy. Radiat Oncol. https://doi.org/10.1186/1748-717x-9-103
Yamaguchi M, Kashiwakura I (2013) Role of reactive oxygen species in the radiation response of human hematopoietic stem/progenitor cells. PLoS ONE. https://doi.org/10.1371/journal.pone.0070503
Yamamori T, Yasui H, Yamazumi M, Wada Y, Nakamura H, Inanami O (2012) Ionizing radiation induces mitochondrial reactive oxygen species production accompanied by upregulation of mitochondrial electron transport chain function and mitochondrial content under control of the cell cycle checkpoint. Free Radical Bio Med 53:S65–S65. https://doi.org/10.1016/j.freeradbiomed.2012.08.556
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Park, K., Dhupal, M., Kim, CS. et al. Ameliorating effect of CpG-ODN (oligodeoxynucleotide) against radiation-induced lung injury in mice. Radiat Environ Biophys 59, 733–741 (2020). https://doi.org/10.1007/s00411-020-00871-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00411-020-00871-w