Abstract
Purpose Lung cancer remains the leading cause of cancer deaths with intricate mechanisms. In the present study, we evaluated the clinical significance and biological role of ANGPTL4 in non-small cell lung cancer (NSCLC), the most common lung cancer subtype.
Methods Quantitative reverse transcription-polymerase chain reaction (qRT-PCR) was used for examining the mRNA level of ANGPTL4 in NSCLC tissues and adjacent non-tumor tissues, NSCLC cell lines, and the immortalized human bronchial epithelial cell line HBE, respectively. A tissue microarray was used for analyzing the relationship between ANGPTL4 expression and the clinicopathological parameters of NSCLC patients. Commercial lentivirus expressing shRNAs was used for silencing ANGPTL4. Cell Counting Kit-8 (CCK-8) was employed for evaluating the cell proliferation ability and transwell with or without matrigel was used for cell migration and invasion assay.
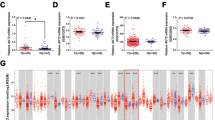
Results As the result, ANGPTL4 was over-expressed in NSCLC tissues compared with benign lung tissues. Silencing ANGPTL4 expression strongly inhibited the proliferation, migration, and invasion of A549 and H520 cells, which was in accordance with the increase of epithelial marker E-cadherin and decrease of mesenchymal marker vimentin. By screening the ERK, AKT, EGFR, and STAT3 pathways, we found that cell growth, migration, and invasion arrest induced by loss of ANGPTL4 expression was partially attributable to down-regulation of ERK signaling.
Conclusion These results suggested that ANGPTL4 was essential for proliferation and metastasis of lung cancer cells and might serve as a novel target for the treatment of lung cancer.






Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A (2013) Cancer statistics, 2013. CA Cancer J Clin 63(1):11–30
Wood SL, Pernemalm M, Crosbie PA et al (2015) Molecular histology of lung cancer: from targets to treatments. Cancer Treat Rev 41(4):361–375
Lubos E, Schnabel R, Rupprecht HJ et al (2006) Prognostic value of tissue inhibitor of metalloproteinase-1 for cardiovascular death among patients with cardiovascular disease: results from the AtheroGene study. Eur Heart J 27(2):150–156
Henschke CI, Yankelevitz DF, Libby DM et al (2006) Survival of patients with stage I lung cancer detected on CT screening. N Engl J Med 355(17):1763–1771
Albain KS, Swann RS, Rusch VW et al (2009) Radiotherapy plus chemotherapy with or without surgical resection for stage III non-small-cell lung cancer: a phase III randomised controlled trial. Lancet 374(9687):379–386
Kim S-H, Park Y-Y, Kim S-W et al (2011) ANGPTL4 induction by prostaglandin E2 under hypoxic conditions promotes colorectal cancer progression. Cancer Res 71(22):7010–7020
Nakayama T, Hirakawa H, Shibata K et al (2011) Expression of angiopoietin-like 4 (ANGPTL4) in human colorectal cancer: ANGPTL4 promotes venous invasion and distant metastasis. Oncol Rep 25(4):929–935
Tanaka J, Irié T, Yamamoto G et al (2015) ANGPTL4 regulates the metastatic potential of oral squamous cell carcinoma. J Oral Pathol Med 44(2):126–133
Okochi-Takada E, Hattori N, Tsukamoto T et al (2014) ANGPTL4 is a secreted tumor suppressor that inhibits angiogenesis. Oncogene 33(17):2273–2278
Hattori N, Okochi-Takada E, Kikuyama M et al (2011) Methylation silencing of angiopoietin-like 4 in rat and human mammary carcinomas. Cancer Sci 102(7):1337–1343
Zhu P, Goh YY, Chin HFA et al (2012) Angiopoietin-like 4: a decade of research. Biosci Rep 32(3):211–219
Guo L, Li S-Y, Ji F-Y et al (2014) Role of ANGPTL4 in vascular permeability and inflammation. Inflamm Res 63(1):13–22
Hermann L, Pinkerton M, Jennings K et al (2005) Angiopoietin-like-4 is a potential angiogenic mediator in arthritis. Clin Immunol 115(1):93–101
Wu Y, Zhou BP (2009) Inflammation: a driving force speeds cancer metastasis. Cell Cycle 8(20):3267–3273
Hanahan D, Weinberg RA (2000) The hallmarks of cancer. Cell 100(1):57–70
Reya T, Morrison SJ, Clarke MF et al (2001) Stem cells, cancer, and cancer stem cells. Nature 414(6859):105–111
Lamouille S, Xu J, Derynck R (2014) Molecular mechanisms of epithelial–mesenchymal transition. Nat Rev Mol Cell Biol 15(3):178
Gonzalez DM, Medici D (2014) Signaling mechanisms of the epithelial-mesenchymal transition. Sci Signal 7(344):re8
Nakaya Y, Sheng G (2013) EMT in developmental morphogenesis. Cancer Lett 341(1):9–15
Nowrin K, Sohal SS, Peterson G et al (2014) Epithelial-mesenchymal transition as a fundamental underlying pathogenic process in COPD airways: fibrosis, remodeling and cancer. Expert Rev Respir Med 8(5):547–559
Sato M, Shames DS, Hasegawa Y (2012) Emerging evidence of epithelial-to-mesenchymal transition in lung carcinogenesis. Respirology 17(7):1048–1059
Santaliz-Ruiz I, Luis E, Xie X et al (2014) Emerging role of nanog in tumorigenesis and cancer stem cells. Int J Cancer 135(12):2741–2748
Nurwidya F, Takahashi F, Murakami A et al (2012) Epithelial mesenchymal transition in drug resistance and metastasis of lung cancer. Cancer Res Treat 44(3):151–156
Gomez-Casal R, Bhattacharya C, Ganesh N et al (2013) Non-small cell lung cancer cells survived ionizing radiation treatment display cancer stem cell and epithelial-mesenchymal transition phenotypes. Mol Cancer 12(1):1
Reka AK, Chen G, Jones RC et al (2014) Epithelial–mesenchymal transition-associated secretory phenotype predicts survival in lung cancer patients. Carcinogenesis 35(6):1292–1300.
Fumarola C, Bonelli MA, Petronini PG et al (2014) Targeting PI3K/AKT/mTOR pathway in non small cell lung cancer. Biochem Pharmacol 90(3):197–207
Yamada T, Takeuchi S, Fujita N et al (2013) AKT kinase-interacting protein1, a novel therapeutic target for lung cancer with EGFR-activating and gatekeeper mutations. Oncogene 32(37):4427–4435
Weerasinghe P, Garcia GE, Zhu Q et al (2007) Inhibition of Stat3 activation and tumor growth suppression of non-small cell lung cancer by G-quartet oligonucleotides. Int J Oncol 31(1):129–136
Maheswaran S, Sequist LV, Nagrath S et al (2008) Detection of mutations in EGFR in circulating lung-cancer cells. N Engl J Med 359(4):366–377
Nguyen T, Tran E, Nguyen T et al (2004) The role of activated MEK-ERK pathway in quercetin-induced growth inhibition and apoptosis in A549 lung cancer cells. Carcinogenesis 25(5):647–659
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Zhu, X., Guo, X., Wu, S. et al. ANGPTL4 Correlates with NSCLC Progression and Regulates Epithelial-Mesenchymal Transition via ERK Pathway. Lung 194, 637–646 (2016). https://doi.org/10.1007/s00408-016-9895-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-016-9895-y