Abstract
Background
We postulate that in adults there is an established lung structure maintenance program and that lung alveolar septal cells are undergoing both continuous apoptosis and proliferation. Whereas lung cell apoptosis has been recognized in human emphysema, little is known about cell proliferation.
Methods
Using a novel rat model of emphysema, induced by intratracheal instillation of cigarette smoke extract (CSE), we investigated the dynamics of emphysematous lung destruction. Emphysematous lung destruction was determined by measuring mean linear intercept and destructive index. Lung injury and repair were assessed by immunohistochemistry and Western blot analysis for active caspase-3 and proliferating cell nuclear antigen (PCNA) after 4, 8, and 12 weeks of CSE instillations.
Results
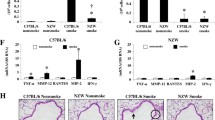
The emphysematous lung tissue destruction was present at 4 weeks of CSE treatment and progressed to 8 weeks. Spontaneous repair began at 12 weeks. Treatment with a peroxisome proliferator activated receptor (PPAR)α+γ agonist or granulocyte and macrophage-colony stimulating factor (GM-CSF) for 4 weeks prevented the progression of emphysematous lung destruction and decreased the number of caspase-3-positive cells.
Conclusion
Apoptosis and cell proliferation occur in this new model of emphysema. Treatment with a PPARα+γ agonist or GM-CSF can inhibit the progression of emphysematous alveolar septal destruction by decreasing alveolar cell apoptosis.








Similar content being viewed by others
References
Demedts IK, Demoor T, Bracke KR, Joos GF, Brusselle GG (2006) Role of apoptosis in the pathogenesis of COPD and pulmonary emphysema. Respir Res 7:53
Imai K, Mercer BA, Schulman LL, Sonett JR, D’Armiento JM (2005) Correlation of lung surface area to apoptosis and proliferation in human emphysema. Eur Respir J 25:250–258
Kasahara Y, Tuder RM, Taraseviciene-Stewart L, Le Cras TD, Abman S, Hirth PK, Waltenberger J, Voelkel NF (2000) Inhibition of VEGF receptors causes lung cell apoptosis and emphysema. J Clin Invest 106:1311–1319
Yokohori N, Aoshiba K, Nagai A (2004) Increased levels of cell death and proliferation in alveolar wall cells in patients with pulmonary emphysema. Chest 125:626–632
Massaro GD, Massaro D (1997) Retinoic acid treatment abrogates elastase-induced pulmonary emphysema in rats. Nat Med 3:675–677
Massaro D, Massaro GD, Baras A, Hoffman EP, Clerch LB (2004) Calorie-related rapid onset of alveolar loss, regeneration, and changes in mouse lung gene expression. Am J Physiol Lung Cell Mol Physiol 286:L896–L906
Churg A, Wang RD, Xie C, Wright JL (2003) Alpha-1-Antitrypsin ameliorates cigarette smoke-induced emphysema in the mouse. Am J Respir Crit Care Med 168:199–207
Foronjy RF, Mirochnitchenko O, Propokenko O, Lemaitre V, Jia Y, Inouye M, Okada Y, D’Armiento JM (2006) Superoxide dismutase expression attenuates cigarette smoke- or elastase-generated emphysema in mice. Am J Respir Crit Care Med 173:623–631
Lee JH, Lee DS, Kim EK, Choe KH, Oh YM, Shim TS, Kim SE, Lee YS, Lee SD (2005) Simvastatin inhibits cigarette smoking-induced emphysema and pulmonary hypertension in rat lungs. Am J Respir Crit Care Med 172:987–993
Martorana PA, Beume R, Lucattelli M, Wollin L, Lungarella G (2005) Roflomilast fully prevents emphysema in mice chronically exposed to cigarette smoke. Am J Respir Crit Care Med 172:848–853
Wright JL, Farmer SG, Churg A (2002) Synthetic serine elastase inhibitor reduces cigarette smoke induced emphysema in guinea pigs. Am J Respir Crit Care Med 166:954–960
Petrache I, Fiualkowska I, Zhen L, Medler TR, Brown E, Vruz P, Choe KH, Tarasevicience-Stewart L, Scerbavicius R, Shapiro L, Zhang B, Song S, Hicklin D, Voelkel NF, Flotte T, Tuder RM (2006) A novel antiapoptotic role for alpha-1 antitrypsin in the prevention of pulmonary emphysema. Am J Respir Crit Care Med 173:1222–1228
Petrache I, Fiualkowska I, Medler TR, Skirball J, Cruz P, Zhen L, Petrache HI, Flotte T, Tuder RM (2006) Alpha-1 antitrypsin inhibits caspase-3 activity, preventing lung endothelial cell apoptosis. Am J Pathol 169:1155–1166
Hoshino Y, Mio T, Nagai S, Miki H, Ito I, Izumi T (2001) Cytotoxic effects of cigarette smoke extract on an alveolar type II cell-derived cell line. Am J Physiol Lung Cell Mol Physiol 281:L509–L516
Thurlbeck WM (1967) Measurement of pulmonary emphysema. Am Rev Respir Dis 95:752–764
Saetta M, Shiner RJ, Angus GE, Kim WD, Wang NS, King M, Ghezzo H, Cosio MG (1985) Destructive index: a measurement of lung parenchymal destruction in smokers. Am Rev Respir Dis 131:764–769
Rangel-Moreno J, Hartson L, Navarro C, Gaxiola M, Selman M, Randall TD (2006) Inducible bronchus-associated lymphoid tissue (iBALT) in patients with pulmonary complications of rheumatoid arthritis. J Clin Invest 116:3183–3194
Elias JA, Kang MJ, Crouthers K, Homer R, Lee CG (2006) State of the art. Mechanistic heterogeneity in chronic obstructive pulmonary disease: insight from transgenic mice. Proc Am Thorac Soc 3:494–498
Taraseviciene-Stewart L, Scerbavicius R, Choe KH, Moore M, Sullivan A, Nicolls MR, Fontenot AP, Tuder RM, Voelkel NF (2005) An animal model of autoimmune emphysema. Am J Respir Crit Care Med 171:734–742
Mahadeva R, Shapiro SD (2002) Chronic obstructive pulmonary disease: Experimental animal models of pulmonary emphysema. Thorax 57:908–914
Nana-Sinkam SP, Lee JD, Sotto-Santiago S, Stearman RS, Keith RL, Choudhury Q, Cool C, Parr J, Moore MD, Bull TM, Voelkel NF, Geraci MW (2007) Prostacyclin prevents pulmonary endothelial cell apoptosis induced by cigarette smoke. Am J Respir Crit Care Med 175:676–685
D’hulst AI, Vermaelen KY, Brusselle GG, Joos GF, Pauwels RA (2005) Time course of cigarette smoke-induced pulmonary inflammation in mice. Eur Respir J 26:204–213
Tschernig T, Pabst R (2000) Bronchus-associated lymphoid tissue (BALT) is not present in the normal adult lung but in different diseases. Pathobiology 68:1–8
Richmond I, Pritchard GE, Ashcroft T, Avery A, Corris PA, Walters EH (1993) Bronchus associated lymphoid tissue (BALT) in human lung: its distribution in smokers and non-smokers. Thorax 48:1130–1134
Aoshiba K, Yokohori N, Nagai A (2003) Alveolar cell apoptosis causes lung destruction and emphysematous changes. Am J Respir Cell Mol Biol 28:555–562
Chen Y, Hanaoka M, Droma Y, Chen P, Voelkel NF, Kubo K (2010) Endothelin-1 receptor antagonists prevent the development of pulmonary emphysema in rats. Eur Respir J 35:904–912
Nakamachi T, Nomiyama T, Gizard F, Heywood EB, Jones KL, Zhao Y, Fuentes L, Takebayashi K, Aso Y, Staels B, Inukai T, Bruemmer D (2007) PPAR alpha agonists suppress osteopontin expression in macrophages and decrease plasma levels in patients with type 2 diabetes. Diabetes 56:1662–1670
Wohlfert EA, Nichols FC, Nevius E, Clark RB (2007) Peroxisome proliferator-activated receptor γ and immunoregulation: Enhancement of regulatory T cells through PPARγ-dependent and independent mechanisms. J Immunol 178:4129–4135
De Ciuceis C, Amiri F, Iglarz M, Cohn JS, Touyz RM, Schiffrin EL (2007) Synergistic vascular protective effects of combined low doses of PPARα and PPARγ activators in angiotensin II-induced hypertension in rats. Brit J Pharmacol 151:45–53
Chintalgattu V, Harris GS, Akula SM, Katwa LC (2007) PPAR-γ agonists induce the expression of VEGF and its receptors in cultured cardiac myofibroblasts. Cardiovasc Res 74:140–150
van Eeden SF, Kitagawa Y, Sato Y, Hogg JC (1999) Polymorphonuclear leukocytes released from the bone marrow and acute lung injury. Chest 116:43S–46S
Azoulay E, Herigault S, Levame M, Brochard L, Schlemmer B, Harf A, Delclaux C (2003) Effect of granulocyte colony-stimulating factor on bleomycin-induced acute lung injury and pulmonary fibrosis. Crit Care Med 31:1442–1448
Vlahos R, Bozinovski S, Hamilton JA, Andoerson GP (2006) Therapeutic potential of treating chronic obstructive pulmonary disease (COPD) by neutralising granulocyte macrophage-colony stimulating factor (GM-CSF). Pharmacol Ther 112:106–115
Nathan C (2002) Point of control in inflammation. Nature 420:846–852
Jackson JD, Yan Y, Brunda MJ, Kelsey LS, Talmadge JE (1995) Interleukin-12 enhances peripheral hematopoiesis in vivo. Blood 85:2371–2376
Roberts AW, Metcalf D (1994) Granulocyte colony-stimulating factor induces selective elevations of progenitor cells in the peripheral blood of mice. Exp Hematol 22:1156–1163
Schenk S, Mal N, Finan A, Zhang M, Kiedrowski M, Popovic Z, McCarthy PM, Penn MS (2007) Monocyte chemotactic protein-3 is a myocardial mesenchymal stem cell homing factor. Stem Cells 25:245–251
Cantürk NZ, Vural B, Esen N, Cantürk Z, Oktay G, Kirkali G, Solkoglu S (1999) Effects of granulocyte-macrophage colony-stimulating factor on incisional wound healing in an experimental diabetic rat model. Endocr Res 25:105–116
Jaschke E, Zabernigg A, Gattringer C (1999) Recombinant human granulocyte-macrophage colony-stimulating factor applied locally in low doses enhances healing and prevents recurrence of chronic venous ulcers. Int J Dermatol 38:380–386
Ishizawa K, Kubo H, Yamada M, Kobayashi S, Numasaki M, Ueda S, Suzuki T, Sasaki H (2004) Bone marrow-derived cells contribute to lung regeneration after elastase-induced pulmonary emphysema. FEBS Lett 556:249–252
Ishikawa T, Aoshiba K, Yokohori N, Nagai A (2006) Macrophage colony-stimulating factor aggravates rather than regenerates emphysematous lungs in mice. Respiration 73:538–545
Shapiro SD (2004) Smoke gets in your cells. Am J Respir Cell Mol Biol 31:481–482
Vandivier RW, Henson PM, Douglas IS (2006) Burying the dead: the impact of failed apoptotic cell removal (efferocytosis) on chronic inflammatory lung disease. Chest 129:1673–1682
Acknowledgment
This work was supported by a NIH grant [NIH ROI HL 72235 (NFV)], the Bixler Family COPD Foundation (AHA SDG 0735388 N), and an annual grant provided by Glaxo-Smith-Kline (King of Prussia, PA).
Conflict of interest
Authors in the manuscript has no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, JH., Hanaoka, M., Kitaguchi, Y. et al. Imbalance of Apoptosis and Cell Proliferation Contributes to the Development and Persistence of Emphysema. Lung 190, 69–82 (2012). https://doi.org/10.1007/s00408-011-9326-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-011-9326-z