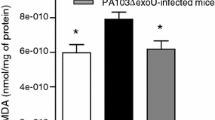
Abstract
The aim of this study was to determine the functional significance of peroxiredoxin V (PRXV) in defense against oxidative stress and changes of its expression in human lung inflammation. We used in vitro cell cultures and retrospective analyses of human sputum samples to perform the study. We found that stable clones of lung epithelial cell lines A549 and U1810 with reduced expression of PRXV were prone to oxidative damage. Upregulation of PRXV decreased induction of DNA double-strand breaks and protein oxidation by cigarette smoke extract and hydrogen peroxide. Transfection with PRXV-carrying plasmid protected Calu-3 confluent epithelial cell sheets from alterations in barrier permeability induced by oxidative stress. In human sputum proinflammatory cytokines, myeloperoxidase, and PRXV were increased during viral-induced inflammation. We conclude that PRXV is an important antioxidant protein of lung epithelial cells. Its expression in the human lung increases in inflammation.








Similar content being viewed by others
References
Ames BN, Shigenaga K, Hagen TM. (1993) Oxidants, antioxidants and the degenerative diseases of aging. Proc Natl Acad Sci U S A 90:7915–7922
Crapo JD (2003) Redox active agents in inflammatory lung injury. Am J Respir Crit Care Med 168:1027–1028
Fajardo I, Svensson L, Bucht A, Pejler G (2004) Increased levels of hypoxia-sensitive proteins in allergic airway inflammation. Am J Respir Crit Care Med 170:477–484
Kinnula VL (2005) Focus on antioxidant enzymes and antioxidant strategies in smoking related airway diseases. Thorax 60:693–700
Kinnula VL, Crapo JD (2003) Superoxide dismutases in the lung and human lung diseases. Am J Respir Crit Care Med 167:1600–1619
Banmeyer I, Marchand C, Verhaeghe C, Vucic B, Rees J-F, Knoops B (2004) Overexpression of human peroxiredoxin 5 in subcellular compartments of Chinese Hamster Ovary cells: effects on cytotoxicity and DNA damage caused by peroxides. Free Rad Biol Med 36:65–77
Das KC, Pahl PM, Guo XL, White CW (2001) Induction of peroxiredoxin gene expression by oxygen in lungs of newborn primates. Am J Respir Cell Mol Biol 25:226–232
Hosoya-Matsuda N, Motohashi K, Yoshimura H, Nozaki A, Inoue K, Ohmori M, Hisabori T (2005) Anti-oxidative stress system in cyanobacteria: significance of type II peroxiredoxin and the role of 1-cys peroxiredoxin in Synechocystis sp. strain PCC 6803. J Biol Chem 280:840–846
Kim HS, Kang SW, Rhee SG, Clerch LB (2001) Rat lung peroxiredoxins I and II are differentially regulated during development and by hyperoxia. Am J Physiol Lung Cell Mol Physiol 280:L1212–L1217
Kim H, Lee T-H, Park ES, Suh JM, Park SJ, Chung HK, Kwon O-Y, Kim YK, Ro HK, Shong M (2000) Role of peroxiredoxins in regulating intracellular hydrogen peroxide and hydrogen peroxide-induced apoptosis in thyroid cells. J Biol Chem 275:18266–18270
Kim H, Manevich Y, Feinstein SI, Pak JH, Ho YS, Fisher AB (2003) Induction of 1-cys peroxiredoxin expression by oxidative stress in lung epithelial cells. Am J Physiol Lung Cell Mol Physiol 285:L363–L369
Kinnula VL, Lehtonen S, Kaarteenaho-Wiik R, Lakari E, Paakko P, Kang SW, Rhee SG, Soini Y (2002) Cell specific expression of peroxiredoxins in human lung and pulmonary sarcoidosis. Thorax 57:157–164
Kinnula VL, Lehtonen S, Sormunen R, Kaarteenaho-Wiik R, Kang SW, Rhee SG, Soini Y (2002) Overexpression of peroxiredoxins I, II, III, V, and VI in malignant mesothelioma. J Pathol 196:316–323
Knoops B, Clippe A, Bogard C, Arsalane K, Wattiez R, Hermans C, Duconseille E, Falmagne P, Bernard A (1999) Cloning and characterization of AOEB166, a novel mammalian antioxidant enzyme of the peroxiredoxin family. J Biol Chem 274:30451–30458
Kropotov AV, Grudinkin PS, Pleskach NM, Gavrilov BA, Tomilin NV, Zhivotovsky BD (2004) Down-regulation of peroxiredoxin V stimulates formation of etoposide-induced double-strand DNA breaks. FEBS Lett 572:75–79
Kropotov AV, Sedova V, Ivanov V, Sazeeva N, Tomilin A, Krutilina RI, Oei SL, Griesenbeck J, Buchlow G, Tomilin NV (1999) A novel human DNA-binding protein with sequence similarity to a subfamily of redox proteins, which is able to repress RNA-polymerase-III-driven transcription of the Alu-family retroposons in vitro. Eur J Biochem 260:336–346
Kropotov A, Gogvadze V, Shupliakov O, Tomilin N, Serikov V, Tomilin NV, Zhivotovsky B (2006) Peroxiredoxin V is essential for protection against apoptosis in human lung carcinoma cells. Exp Cell Res 312:2806–2815
Kropotov A, Serikov V, Suh J, Smirnova A, Bashkirov V, Zhivotovsky B, Tomilin NV (2006) Constitutive expression of the human peroxiredoxin V gene contributes to protection of the genome from oxidative DNA lesions and to suppression of transcription of noncoding DNA. FEBS J 273:2607–2617
Lehtonen ST, Markkanen PMH, Peltoniemi M, Kang SW, Kinnula VL (2005) Variable over-oxidation of peroxiredoxins in human lung cells in severe oxidative stress. Am J Physiol Lung Cell Mol Physiol 288:L997–L1001
Manevich Y, Feinstein SI, Fisher AB (2004) Activation of the antioxidant enzyme 1-CYS peroxiredoxin requires glutathionylation mediated by heterodimerization with GST. Proc Natl Acad Sci U S A 101:3780–3785
Manevich Y, Sweitzer T, Pak JH, Feinstein SI, Muzykantov V, Fisher AB (2002) 1-Cys peroxiredoxin overexpression protects cells against phospholipid peroxidation-mediated membrane damage. Proc Natl Acad Sci U S A 99:11599–11604
Pak JH, Manevich Y, Kim HS, Feinstein SI, Fisher AB (2002) An antisense oligonucleotide to 1-cys peroxiredoxin causes lipid peroxidation and apoptosis in lung epithelial cells. J Biol Chem 277:49927–49934
Hoshino T, Nakamura H, Okamoto M, Kato S, Araya S, Nomiyama K, Oizumi K, Young HA, Aizawa H, Yodoi J (2003) Redox-reactive protein Thioredoxin prevents proinflammatory cytokine- or bleomycin-induced lung injury. Am J Respir Crit Care Med 168:1075–1083
Wang Y, Manevich Y, Feinstein SI, Fisher AB (2004) Adenovirus-mediated transfer of the 1-cys peroxiredoxin gene to mouse lung protects against hyperoxic injury. Am J Physiol Lung Cell Mol Physiol 286:L1188-L1193
Wang X, Phelan SA, Forsman-Semb K, Taylor EF, Petros C, Brown A, Lerner CP, Paigen B (2003) Mice with targeted mutation of peroxiredoxin 6 develop normally but are susceptible to oxidative stress. J Biol Chem 278:25179–25190
Wattiez R, Hermans C, Bernard A, Lesur O, Falmagne P (1999) Human bronchoalveolar lavage fluid: two-dimensional gel electrophoresis, amino acid microsequencing and identification of major proteins. Electrophoresis 20:1634–1645
Wood ZA, Poole LB, Karplus PA (2003) Peroxiredoxin evolution and the regulation of hydrogen peroxide signaling. Science 300:650–653
Zhou Y, Kok KH, Chun AC, Wong CM, Wu HW, Lin MC, Fung PC, Kung H, Jin DY (2000) Mouse peroxiredoxin V is a thioredoxin peroxidase that inhibits p53-induced apoptosis. Biochem Biophys Res Commun 24:921–927
Serikov VB, Popov BV, Kropotov AV, Tomilin NV (2005) Bone marrow-derived cells restore expression of peroxiredoxin V in the airways following acute naphthalene injury in mice. Cytotherapy 336:762–769
Serikov VB, Leutenegger C, Krutilina RI, Kropotov AV, Pleskach NM, Suh JH, Tomilin NV (2006) Cigarette smoke extract inhibits expression of peroxiredoxin V and increases airway epithelial permeability. Inhal Toxicol 18:79–92
Buss H, Chan T, Sluis K, Dominigan N, Winterbourn CC (1997) Protein carbonyl measurement by a sensitive ELISA method. Free Radic Biol Med 23:361–366
Serikov VB, Choi H, Chmiel KJ, Wu R, Widdicombe JH (2004) Role of ERK in the increase in airway epithelial permeability during leukocyte transmigration. Am J Respir Cell Mol Biol 30:261–270
Avila PC, Abisheganaden JA, Wong H, Liu J, Yagi S, Schnurr D, Kishiyama JL, Boushey HA (2000) Effects of allergic inflammation of the nasal mucosa on the severity of rhinovirus 16 cold. J Allergy Clin Immunol 105:923–932
Lopez-Souza N, Avila PC, Widdicombe JH (2003) Polarized cultures of human airway epithelium from nasal scrapings and bronchial brushings. In Vitro Cell Dev Biol Anim 39:266–269
Albino AP, Huang X, Jorgensen ED, Gietl D, Traganos F, Darzynkewicz Z (2006) Induction of DNA double-strand breaks in A549 and normal human pulmonary epithelial cells by cigarette smoke is mediated by free radicals. Int J Oncol 28:1491–1505
Albino AP, Huang X, Jorgensen ED, Yang J, Gietl D, Traganos F, Darzynkewicz Z (2004) Induction of H2AX phosphorylation in pulmonary cells by tobacco smoke. Cell Cycle 3:1062–1068
Grunberg K, Sterk PJ (1999) Rhinovirus infections: induction and modulation of airways inflammation in asthma. Clin Exp Allergy 29:65–73
Gurn JE, Vrtis R, Grindle KA, Swenson C, Busse WW (2000) Relationship of upper and lower airway cytokines to outcome of experimental rhinovirus infection. Am J Respir Crit Care Med 162:2226–2231
Krutilina RI, Kropotov A, leutenegger CM, Serikov VB (2006) Migrating leukocytes are the source of Peroxiredoxin V during inflammation in the airways. J Inflamm (Lond) 3:13–21
Acknowledgments
Research described in this article was supported by Philip Morris USA Inc. and by Philip Morris International, and by INTAS Genomics grant 05-1000004-7755. Clinical samples were collected under funding from NIAID (AI50496 and AI057506). P. Avila is currently funded by the Ernest S. Bazley Grant to Northwestern University. The authors thank Dr. Homer Boushey, Ms. Theresa Ward, and Ms. Jane Liu for their assistance in conducting the clinical common cold study, and Ms. Junqing Shen for technical work with rhinovirus infection of epithelial cell cultures.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
408_2007_9066_MOESM4_ESM.tif
[DTT is known to affect measurements of some cytokines. Study was performed to determine whether the presence of this reducing agent could influence measurements of PRXV levels by Western blot. DTT and sputum did not affect measurements of PRXV. Representative Western blot analyses for PRXV. Ten nanograms of recombinant PRXV were incubated in PBS, PBS with 10 mM DTT (A), or different sputum samples (B) with known very low or zero concentration of endogenous PRXV for 30 min at 37°C]
408_2007_9066_MOESM5_ESM.tif
[Level of myeloperoxidase (MPO) is an index of the presence of neutrophils, indicative of the level of inflammation. Correlation between MPO levels and PRXV levels was investigated to determine whether levels of PRXV upregulation are related to inflammatory reaction. PRXV levels correlated strongly with MPO levels in induced-sputum samples]
408_2007_9066_MOESM6_ESM.tif
[Experiments were performed in vitro to determine whether inoculation of epithelial cell cultures with RV16 virus changes expression of PRXV without the presence of immune cells. Expression of PRXV was not changed by RV16 infection in vitro in epithelial cells. These data indicate that upregulation of PRXV requires the presence of activated immune cells, which may trigger secretion of PRXV by paracrine mechanisms or become an alternative source of PRXV. A Confocal microscopy images of nasal epithelial cultures stained for PRXV with FITC-labeled secondary antibody. A1 Control staining without primary antibody, no nonspecific green fluorescence is present, costaining with propidium iodine (red fluorescence). Upper left panel – PI red fluorescence; upper right panel – FITC green fluorescence; lower panel – combined image. A2 Control noninfected culture. A3 Culture infected with RV16. Original magnification 40×. Diagrams at the bottom demonstrate distribution of green and red fluorescence signals along the median horizontal axis. B Western blot analyses of tissue lysates for PRXV. Actin was used as control]
Rights and permissions
About this article
Cite this article
Avila, P.C., Kropotov, A.V., Krutilina, R. et al. Peroxiredoxin V Contributes to Antioxidant Defense of Lung Epithelial Cells. Lung 186, 103–114 (2008). https://doi.org/10.1007/s00408-007-9066-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-007-9066-2