Abstract
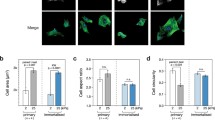
Substrate stiffness is a key regulator of cell behavior. To investigate how mechanical properties of cell microenvironment affect the human keratinocyte, primary cells were seeded on polyacrylamide hydrogels of different compliances (soft: 4 kPa, medium: 14 kPa, rigid: 45 kPa) in comparison with glass coverslip (> GPa). Keratinocyte spreading and proliferation were strongly decreased on the softest hydrogel, while no significant difference was observed between medium, rigid hydrogels and glass coverslip, and cells’ viability was comparable in all conditions after 72 h. We then performed a RNA-seq to compare the transcriptomes from keratinocytes cultured for 72 h on the softest hydrogel or on coverslips. The cells on the soft hydrogel showed a strong increase in the expression of late differentiation marker genes from the epidermal differentiation complex (1q21) and the antioxidant machinery. In parallel, these cells displayed a significant loss of expression of the matrix receptors (integrin α6 and β1) and the EGF receptor. However, when these cells were replated on a plastic culture plate (> GPa), they were able to re-engage the proliferation machinery with a strong colony-formation efficiency. Overall, using low-calcium differentiation monolayers at confluence, the lesser the rigidity, the stronger the markers of late differentiation are expressed, while the inverse is observed regarding the markers of early differentiation. In conclusion, below a certain rigidity, human keratinocytes undergo genome reprogramming indicating terminal differentiation that can switch back to proliferation in contact with a stiffer environment.



Similar content being viewed by others
References
Abhishek S, Palamadai Krishnan S (2016) Epidermal differentiation complex: a review on its epigenetic regulation and potential drug targets. Cell J 18:1–6
Adams JC, Watt FM (1989) Fibronectin inhibits the terminal differentiation of human keratinocytes. Nature 340:307–309. https://doi.org/10.1038/340307a0
Blanpain C, Fuchs E (2009) Epidermal homeostasis: a balancing act of stem cells in the skin. Nat Rev Mol Cell Biol 10:207–217. https://doi.org/10.1038/nrm2636
Chen S, Shi J, Xu X, Ding J, Zhong W, Zhang L, Xing M, Zhang L (2016) Study of stiffness effects of poly(amidoamine)-poly(n-isopropyl acrylamide) hydrogel on wound healing. Coll Surf B Biointerfaces 140:574–582. https://doi.org/10.1016/j.colsurfb.2015.08.041
Eden E, Navon R, Steinfeld I, Lipson D, Yakhini Z (2009) GOrilla: a tool for discovery and visualization of enriched GO terms in ranked gene lists. BMC Bioinform 10:48. https://doi.org/10.1186/1471-2105-10-48
Edwards C, Marks R (1995) Evaluation of biomechanical properties of human skin. Clin Dermatol 13:375–380
El-Domyati M, Attia S, Saleh F, Brown D, Birk DE, Gasparro F, Ahmad H, Uitto J (2002) Intrinsic aging vs. photoaging: a comparative histopathological, immunohistochemical, and ultrastructural study of skin. Exp Dermatol 11:398–405
Evans ND, Oreffo RO, Healy E, Thurner PJ, Man YH (2013) Epithelial mechanobiology, skin wound healing, and the stem cell niche. J Mech Behav Biomed Mater 28:397–409. https://doi.org/10.1016/j.jmbbm.2013.04.023
Hinz B, Mastrangelo D, Iselin CE, Chaponnier C, Gabbiani G (2001) Mechanical tension controls granulation tissue contractile activity and myofibroblast differentiation. Am J Pathol 159:1009–1020. https://doi.org/10.1016/S0002-9440(10)61776-2
Hsu CK, Lin HH, Harn HI, Hughes MW, Tang MJ, Yang CC (2018) Mechanical forces in skin disorders. J Dermatol Sci 90:232–240. https://doi.org/10.1016/j.jdermsci.2018.03.004
Jor JW, Parker MD, Taberner AJ, Nash MP, Nielsen PM (2013) Computational and experimental characterization of skin mechanics: identifying current challenges and future directions. Wiley Interdiscip Rev Syst Biol Med 5:539–556. https://doi.org/10.1002/wsbm.1228
Kenny FN, Connelly JT (2015) Integrin-mediated adhesion and mechano-sensing in cutaneous wound healing. Cell Tissue Res 360:571–582. https://doi.org/10.1007/s00441-014-2064-9
Kenny FN, Drymoussi Z, Delaine-Smith R, Kao AP, Laly AC, Knight MM, Philpott MP, Connelly JT (2018) Tissue stiffening promotes keratinocyte proliferation through activation of epidermal growth factor signaling. J Cell Sci 131:jcs215780. https://doi.org/10.1242/jcs.215780
Law CW, Chen Y, Shi W, Smyth GK (2014) voom: precision weights unlock linear model analysis tools for RNA-seq read counts. Genome Biol 15:R29. https://doi.org/10.1186/gb-2014-15-2-r29
Liao Y, Smyth GK, Shi W (2013) The Subread aligner: fast, accurate and scalable read mapping by seed-and-vote. Nucleic Acids Res 41:e108. https://doi.org/10.1093/nar/gkt214
Liem RK (2016) Cytoskeletal Integrators: the spectrin superfamily. Cold Spring Harb Perspect Biol 8:a018259. https://doi.org/10.1101/cshperspect.a018259
Mainzer C, Remoue N, Molinari J, Rousselle P, Barricchello C, Lago JC, Sommer P, Sigaudo-Roussel D, Debret R (2018) In vitro epidermis model mimicking IGF-1-specific age-related decline. Exp Dermatol 27:537–543. https://doi.org/10.1111/exd.13547
McCarthy DJ, Chen Y, Smyth GK (2012) Differential expression analysis of multifactor RNA-Seq experiments with respect to biological variation. Nucleic Acids Res 40:4288–4297. https://doi.org/10.1093/nar/gks042
Poumay Y, Pittelkow MR (1995) Cell density and culture factors regulate keratinocyte commitment to differentiation and expression of suprabasal K1/K10 keratins. J Invest Dermatol 104:271–276
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK (2015) limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43:e47. https://doi.org/10.1093/nar/gkv007
Robinson MD, McCarthy DJ, Smyth GK (2010) edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26:139–140. https://doi.org/10.1093/bioinformatics/btp616
Trappmann B, Gautrot JE, Connelly JT, Strange DG, Li Y, Oyen ML, Cohen Stuart MA, Boehm H, Li B, Vogel V, Spatz JP, Watt FM, Huck WT (2012) Extracellular-matrix tethering regulates stem-cell fate. Nat Mater 11:642–649. https://doi.org/10.1038/nmat3339
Varani J, Dame MK, Rittie L, Fligiel SE, Kang S, Fisher GJ, Voorhees JJ (2006) Decreased collagen production in chronologically aged skin: roles of age-dependent alteration in fibroblast function and defective mechanical stimulation. Am J Pathol 168:1861–1868. https://doi.org/10.2353/ajpath.2006.051302
Waldera Lupa DM, Kalfalah F, Safferling K, Boukamp P, Poschmann G, Volpi E, Gotz-Rosch C, Bernerd F, Haag L, Huebenthal U, Fritsche E, Boege F, Grabe N, Tigges J, Stuhler K, Krutmann J (2015) Characterization of skin aging-associated secreted proteins (SAASP) produced by dermal fibroblasts isolated from intrinsically aged human skin. J Invest Dermatol 135:1954–1968. https://doi.org/10.1038/jid.2015.120
Wang Y, Wang G, Luo X, Qiu J, Tang C (2012) Substrate stiffness regulates the proliferation, migration, and differentiation of epidermal cells. Burns 38:414–420. https://doi.org/10.1016/j.burns.2011.09.002
Watt FM, Kubler MD, Hotchin NA, Nicholson LJ, Adams JC (1993) Regulation of keratinocyte terminal differentiation by integrin-extracellular matrix interactions. J Cell Sci 106(Pt 1):175–182
Wong VW, Akaishi S, Longaker MT, Gurtner GC (2011) Pushing back: wound mechanotransduction in repair and regeneration. J Invest Dermatol 131:2186–2196. https://doi.org/10.1038/jid.2011.212
Wu XT, Sun LW, Yang X, Ding D, Han D, Fan YB (2017) The potential role of spectrin network in the mechanotransduction of MLO-Y4 osteocytes. Sci Rep 7:40940. https://doi.org/10.1038/srep40940
You H, Padmashali RM, Ranganathan A, Lei P, Girnius N, Davis RJ, Andreadis ST (2013) JNK regulates compliance-induced adherens junctions formation in epithelial cells and tissues. J Cell Sci 126:2718–2729. https://doi.org/10.1242/jcs.122903
Zahouani H, Pailler-Mattei C, Sohm B, Vargiolu R, Cenizo V, Debret R (2009) Characterization of the mechanical properties of a dermal equivalent compared with human skin in vivo by indentation and static friction tests. Skin Res Technol 15:68–76. https://doi.org/10.1111/j.1600-0846.2008.00329.x
Zhao KN, Masci PP, Lavin MF (2011) Disruption of spectrin-like cytoskeleton in differentiating keratinocytes by PKCdelta activation is associated with phosphorylated adducin. PLoS One 6:e28267. https://doi.org/10.1371/journal.pone.0028267
Acknowledgements
This work is supported by Isispharma France. Choua Ya is a recipient of a PhD grant from the French National Association of Research and Technology (ANRT). Mariana Carrancá is supported by a PhD grant from the Région Auvergne Rhône Alpes.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Bérengère Fromy and Romain Debret are co-last authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ya, C., Carrancá, M., Sigaudo-Roussel, D. et al. Substrate softness promotes terminal differentiation of human keratinocytes without altering their ability to proliferate back into a rigid environment. Arch Dermatol Res 311, 741–751 (2019). https://doi.org/10.1007/s00403-019-01962-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-019-01962-5