Abstract
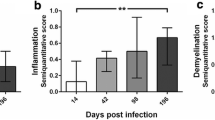
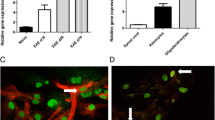
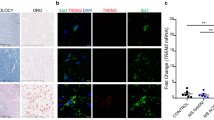
Matrix metalloproteinases (MMPs) are a family of extracellular proteases involved in the pathogenesis of demyelinating diseases like multiple sclerosis (MS). The aim of the present study was to investigate whether MMPs induce direct myelin degradation, leukocyte infiltration, disruption of the blood–brain barrier (BBB), and/or extracellular matrix remodeling in the pathogenesis of Theiler’s murine encephalomyelitis (TME), a virus-induced model of MS. During the demyelinating phase of TME, the highest transcriptional upregulation was detected for Mmp12, followed by Mmp3. Mmp12 −/− mice showed reduced demyelination, macrophage infiltration, and motor deficits compared with wild-type- and Mmp3 knock-out mice. However, BBB remained unaltered, and the amount of extracellular matrix deposition was similar in knock-out mice and wild-type mice. Furthermore, stereotaxic injection of activated MMP-3, -9, and -12 into the caudal cerebellar peduncle of adult mice induced a focally extensive primary demyelination prior to infiltration of inflammatory cells, as well as a reduction in the number of oligodendrocytes and a leakage of BBB. All these results demonstrate that MMP-12 plays an essential role in the pathogenesis of TME, most likely due to its primary myelin- or oligodendrocyte-toxic potential and its role in macrophage extravasation, whereas there was no sign of BBB damage or alterations to extracellular matrix remodeling/deposition. Thus, interrupting the MMP-12 cascade may be a relevant therapeutic approach for preventing chronic progressive demyelination.










Similar content being viewed by others
References
Adams RA, Bauer J, Flick MJ et al (2007) The fibrin-derived gamma 377–395 peptide inhibits microglia activation and suppresses relapsing paralysis in central nervous system autoimmune disease. J Exp Med 204:571–582
Agrawal SM, Lau L, Yong VW (2008) MMPs in the central nervous system: where the good guys go bad. Semin Cell Dev Biol 19:42–51
Anthony DC, Miller KM, Fearn S et al (1998) Matrix metalloproteinase expression in an experimentally-induced DTH model of multiple sclerosis in the rat CNS. J Neuroimmunol 87:62–72
Asahi M, Wang XY, Mori T et al (2001) Effects of matrix metalloproteinase-9 gene knock-out on the proteolysis of blood–brain barrier and white matter components after cerebral ischemia. J Neurosci 21:7724–7732
Bar-Or A (2008) The immunology of multiple sclerosis. Semin Neurol 28:29–45
Bar-Or A, Nuttall RK, Duddy M et al (2003) Analyses of all matrix metalloproteinase members in leukocytes emphasize monocytes as major inflammatory mediators in multiple sclerosis. Brain 126:2738–2749
Bartanusz V, Jezova D, Alajajian B, Digicaylioglu M (2011) The blood–spinal cord barrier: morphology and clinical implications. Ann Neurol 70:194–206
Bhat RV, Axt KJ, Fosnaugh JS et al (1996) Expression of the APC tumor suppressor protein in oligodendroglia. Glia 17:169–174
Canales RD, Luo Y, Willey JC et al (2006) Evaluation of DNA microarray results with quantitative gene expression platforms. Nat Biotechnol 24:1115–1122
Cauwe B, Opdenakker G (2010) Intracellular substrate cleavage: a novel dimension in the biochemistry, biology and pathology of matrix metalloproteinases. Crit Rev Biochem Mol Biol 45:351–423
Chamorro M, Aubert C, Brahic M (1986) Demyelinating lesions due to Theiler’s virus are associated with ongoing central nervous system infection. J Virol 57:992–997
Chandler S, Coates R, Gearing A, Lury J, Wells G, Bone E (1995) Matrix metalloproteinases degrade myelin basic protein. Neurosci Lett 201:223–226
Chandler S, Cossins J, Lury J, Wells G (1996) Macrophage metalloelastase degrades matrix and myelin proteins and processes a tumour necrosis factor-alpha fusion protein. Biochem Biophys Res Commun 228:421–429
Clements JM, Cossins JA, Wells GM et al (1997) Matrix metalloproteinase expression during experimental autoimmune encephalomyelitis and effects of a combined matrix metalloproteinase and tumour necrosis factor-alpha inhibitor. J Neuroimmunol 74:85–94
Compston A, Coles A (2008) Multiple sclerosis. Lancet 372:1502–1517
Dal Canto MC, Kim BS, Miller SD, Melvold RW (1996) Theiler’s murine encephalomyelitis virus (TMEV)-induced demyelination: a model for human multiple sclerosis. Methods 10:453–461
Dal Canto MC, Lipton HL (1982) Ultrastructural immunohistochemical localization of virus in acute and chronic demyelinating Theiler’s virus infection. Am J Pathol 106:20–29
Dallas PB, Gottardo NG, Firth MJ et al (2005) Gene expression levels assessed by oligonucleotide microarray analysis and quantitative real-time RT-PCR—how well do they correlate? BMC Genomics 6:59–69
Del Valle J, Camins A, Pallas M, Vilaplana J, Pelegri C (2008) A new method for determining blood–brain barrier integrity based on intracardiac perfusion of an Evans Blue–Hoechst cocktail. J Neurosci Methods 174:42–49
Fanjul-Fernandez M, Folgueras AR, Cabrera S, Lopez-Otin C (2010) Matrix metalloproteinases: evolution, gene regulation and functional analysis in mouse models. Biochim Biophys Acta 1803:3–19
Fukuda S, Fini CA, Mabuchi T, Koziol JA, Eggleston LL Jr, del Zoppo GJ (2004) Focal cerebral ischemia induces active proteases that degrade microvascular matrix. Stroke 35:998–1004
Gerhauser I, Ulrich R, Alldinger S, Baumgärtner W (2007) Induction of activator protein-1 and nuclear factor-kappaB as a prerequisite for disease development in susceptible SJL/J mice after Theiler murine encephalomyelitis. J Neuropathol Exp Neurol 66:809–818
Goncalves Dasilva A, Yong VW (2008) Expression and regulation of matrix metalloproteinase-12 in experimental autoimmune encephalomyelitis and by bone marrow derived macrophages in vitro. J Neuroimmunol 199:24–34
Goncalves DaSilva A, Yong VW (2009) Matrix metalloproteinase-12 deficiency worsens relapsing-remitting experimental autoimmune encephalomyelitis in association with cytokine and chemokine dysregulation. Am J Pathol 174:898–909
Gröters S, Alldinger S, Baumgärtner W (2005) Up-regulation of mRNA for matrix metalloproteinases-9 and -14 in advanced lesions of demyelinating canine distemper leukoencephalitis. Acta Neuropathol 110:369–382
Gurney KJ, Estrada EY, Rosenberg GA (2006) Blood–brain barrier disruption by stromelysin-1 facilitates neutrophil infiltration in neuroinflammation. Neurobiol Dis 23:87–96
Haist V, Ulrich R, Kalkuhl A, Deschl U, Baumgärtner W (2011) Distinct spatio-temporal extracellular matrix accumulation within demyelinated spinal cord lesions in Theiler’s murine encephalomyelitis. Brain Pathol. doi:10.1111/j.1750-3639.2011.00518.x
Hamann GF, Okada Y, Fitridge R, del Zoppo GJ (1995) Microvascular basal lamina antigens disappear during cerebral ischemia and reperfusion. Stroke 26:2120–2126
Hansmann F, Herder V, Ernst H, Baumgärtner W (2011) Spinal epidermoid cyst in a SJL mouse: case report and literature review. J Comp Pathol 145:373–377
Hansmann F, Pringproa K, Ulrich R et al (2011) Highly malignant behavior of a murine oligodendrocyte precursor cell line following transplantation into the demyelinated and non-demyelinated central nervous system. Cell Transplant (accepted)
Harkness KA, Adamson P, Sussman JD, Davies-Jones GAB, Greenwood J, Woodroofe MN (2000) Dexamethasone regulation of matrix metalloproteinase expression in CNS vascular endothelium. Brain 123:698–709
Herder V, Hansmann F, Stangel M, Skripuletz T, Baumgärtner W, Beineke A (2011) Lack of cuprizone-induced demyelination in the murine spinal cord despite oligodendroglial alterations substantiates the concept of site-specific susceptibilities of the central nervous system. Neuropathol Appl Neurobiol 37:676–684
Hewson AK, Smith T, Leonard JP, Cuzner ML (1995) Suppression of experimental allergic encephalomyelitis in the Lewis rat by the matrix metalloproteinase inhibitor Ro31-9790. Inflamm Res 44:345–349
Ho MK, Springer TA (1983) Tissue distribution, structural characterization, and biosynthesis of Mac-3, a macrophage surface glycoprotein exhibiting molecular weight heterogeneity. J Biol Chem 258:636–642
Jucker M, Tian M, Ingram DK (1996) Laminins in the adult and aged brain. Mol Chem Neuropathol 28:209–218
Kummerfeld M, Meens J, Haas L, Baumgärtner W, Beineke A (2009) Generation and characterization of a polyclonal antibody for the detection of Theiler’s murine encephalomyelitis virus by light and electron microscopy. J Virol Methods 160:185–188
Kumnok J, Ulrich R, Wewetzer K et al (2008) Differential transcription of matrix-metalloproteinase genes in primary mouse astrocytes and microglia infected with Theiler’s murine encephalomyelitis virus. J. Neurovirol 14:205–217
Lee MA, Palace J, Stabler G, Ford J, Gearing A, Miller K (1999) Serum gelatinase B, TIMP-1 and TIMP-2 levels in multiple sclerosis—a longitudinal clinical and MRI study. Brain 122:191–197
Lindberg RLP, De Groot CJA, Montagne L et al (2001) The expression profile of matrix metalloproteinases (MMPs) and their inhibitors (TIMPs) in lesions and normal appearing white matter of multiple sclerosis. Brain 124:1743–1753
Lo EH, Wang XY, Cuzner ML (2002) Extracellular proteolysis in brain injury and inflammation: role for plasminogen activators and matrix metalloproteinases. J Neurosci Res 69:1–9
Miao Q, Baumgärtner W, Failing K, Alldinger S (2003) Phase-dependent expression of matrix metalloproteinases and their inhibitors in demyelinating canine distemper encephalitis. Acta Neuropathol 106:486–494
Mudgett JS, Hutchinson NI, Chartrain NA et al (1998) Susceptibility of stromelysin 1-deficient mice to collagen-induced arthritis and cartilage destruction. Arthritis Rheum 41:110–121
Mutch DM, Berger A, Mansourian R, Rytz A, Roberts MA (2002) The limit fold change model: a practical approach for selecting differentially expressed genes from microarray data. BMC Bioinforma 3:17
Navarrete-Talloni MJ, Kalkuhl A, Deschl U et al (2010) Transient peripheral immune response and central nervous system leaky compartmentalization in a viral model for multiple sclerosis. Brain Pathol 20:890–901
Pagenstecher A, Stalder AK, Kincaid CL, Shapiro SD, Campbell IL (1998) Differential expression of matrix metalloproteinase and tissue inhibitor of matrix metalloproteinase genes in the mouse central nervous system in normal and inflammatory states. Am J Pathol 152:729–741
Prendergast CT, Anderton SM (2009) Immune cell entry to central nervous system—current understanding and prospective therapeutic targets. Endocr Metab Immune Disord Drug Targets 9:315–327
Rodriguez M, Rivera-Quinones C, Murray PD, Kariuki Njenga M, Wettstein PJ, Mak T (1997) The role of CD4+ and CD8+ T cells in demyelinating disease following Theiler’s virus infection: a model for multiple sclerosis. J Neurovirol 3(Suppl 1):43–45
Romeis B, Böck P (1989) Azanfärbung nach Heidenhain. In: Romeis B, Böck P (eds) Mikroskopische Technik. Urban & Fischer, Munich, p 501
Rosenberg GA (2002) Matrix metalloproteinases and neuroinflammation in multiple sclerosis. Neuroscientist 8:586–595
Rosenberg GA (2002) Matrix metalloproteinases in neuroinflammation. Glia 39:279–291
Rosenberg GA (2005) Matrix metalloproteinases biomarkers in multiple sclerosis. Lancet 365:1291–1293
Rosenberg GA (2009) Matrix metalloproteinases and their multiple roles in neurodegenerative diseases. Lancet Neurol 8:205–216
Saria A, Lundberg JM (1983) Evans blue fluorescence: quantitative and morphological evaluation of vascular permeability in animal tissues. J Neurosci Methods 8:41–49
Sato F, Tanaka H, Hasanovic F, Tsunoda I (2011) Theiler’s virus infection: pathophysiology of demyelination and neurodegeneration. Pathophysiology 18:31–41
Shipley JM, Wesselschmidt RL, Kobayashi DK, Ley TJ, Shapiro SD (1996) Metalloelastase is required for macrophage-mediated proteolysis and matrix invasion in mice. Proc Natl Acad Sci USA 93:3942–3946
Si-Tayeb K, Monvoisin A, Mazzocco C et al (2006) Matrix metalloproteinase 3 is present in the cell nucleus and is involved in apoptosis. Am J Pathol 169:1390–1401
Sixt M, Engelhardt B, Pausch F, Hallmann R, Wendler O, Sorokin LM (2001) Endothelial cell laminin isoforms, laminins 8 and 10, play decisive roles in T cell recruitment across the blood–brain barrier in experimental autoimmune encephalomyelitis. J Cell Biol 153:933–946
Skuljec J, Gudi V, Ulrich R et al (2011) Matrix metalloproteinases and their tissue inhibitors in cuprizone-induced demyelination and remyelination of brain white and gray matter. J Neuropathol Exp Neurol 70:758–769
Sobel RA (2001) The extracellular matrix in multiple sclerosis: an update. Braz J Med Biol Res 34:603–609
Teesalu T, Hinkkanen AE, Vaheri A (2001) Coordinated induction of extracellular proteolysis systems during experimental autoimmune encephalomyelitis in mice. Am J Pathol 159:2227–2237
Toft-Hansen H, Nuttall RK, Edwards DR, Owens T (2004) Key metalloproteinases are expressed by specific cell types in experimental autoimmune encephalomyelitis. J Immunol 173:5209–5218
Trapp BD, Nave KA (2008) Multiple sclerosis: an immune or neurodegenerative disorder? Annu Rev Neurosci 31:247–269
Ulrich R, Baumgärtner W, Gerhauser I et al (2006) MMP-12. MMP-3, and TIMP-1 are markedly upregulated in chronic demyelinating Theiler murine encephalomyelitis. J Neuropathol Exp Neurol 65:783–793
Ulrich R, Gerhauser I, Seeliger F, Baumgärtner W, Alldinger S (2005) Matrix metalloproteinases and their inhibitors in the developing mouse brain and spinal cord: a reverse transcription quantitative polymerase chain reaction study. Dev Neurosci 27:408–418
Ulrich R, Kalkuhl A, Deschl U, Baumgärtner W (2010) Machine learning approach identifies new pathways associated with demyelination in a viral model of multiple sclerosis. J Cell Mol Med 14:434–448
Ulrich R, Seeliger F, Kreutzer M, Germann PG, Baumgärtner W (2008) Limited remyelination in Theiler’s murine encephalomyelitis due to insufficient oligodendroglial differentiation of nerve/glial antigen 2 (NG2)-positive putative oligodendroglial progenitor cells. Neuropathol Appl Neurobiol 34:603–620
van Horssen J, Bo L, Vos CM, Virtanen I, de Vries HE (2005) Basement membrane proteins in multiple sclerosis-associated inflammatory cuffs: potential role in influx and transport of leukocytes. J Neuropathol Exp Neurol 64:722–729
van Horssen J, Dijkstra CD, de Vries HE (2007) The extracellular matrix in multiple sclerosis pathology. J Neurochem 103:1293–1301
Vos CMP, van Haastert ES, de Groot CJA, van der Valk P, de Vries HE (2003) Matrix metalloproteinase-12 is expressed in phagocytotic macrophages in active multiple sclerosis lesions. J Neuroimmunol 138:106–114
Wang T, Town T, Alexopoulou L, Anderson JF, Fikrig E, Flavell RA (2004) Toll-like receptor 3 mediates West Nile virus entry into the brain causing lethal encephalitis. Nat Med 10:1366–1373
Yong VW, Power C, Forsyth P, Edwards DR (2001) Metalloproteinases in biology and pathology of the nervous system. Nat Rev Neurosci 2:502–511
Yong VW, Zabad RK, Agrawal S, Goncalves Dasilva A, Metz LM (2007) Elevation of matrix metalloproteinases (MMPs) in multiple sclerosis and impact of immunomodulators. J Neurol Sci 259:79–84
Yuen T, Wurmbach E, Pfeffer RL, Ebersole BJ, Sealfon SC (2002) Accuracy and calibration of commercial oligonucleotide and custom cDNA microarrays. Nucleic Acids Res 30:e48
Zhou J, Marten NW, Bergmann CC, Macklin WB, Hinton DR, Stohlman SA (2005) Expression of matrix metalloproteinases and their tissue inhibitor during viral encephalitis. J Virol 79:4764–4773
Acknowledgments
The authors thank Bettina Buck, Thomas Feidl, Martin Gamber, Petra Grünig, Michael Müller, Kerstin Rohn, Caroline Schütz, Anuschka Unold, and Heinz Theobald for excellent technical assistance. Furthermore, we are grateful to Dr. Karl Rohn, Department of Biometry, Epidemiology and Information Processing, University of Veterinary Medicine Hannover, Germany, for statistical assistance; Prof. Christiane Pfarrer, Department of Anatomy, University of Veterinary Medicine Hannover, Germany for providing the fluorescence microscope; Dr. Maren Luchtefeld, Department of Cardiology and Angiology, Hannover Medical School, Germany for providing the fluorescence reader; Prof. Klaus Schughart, Department of Infection Genetics, Helmholtz Centre for Infection Research, Braunschweig, Germany for help with planning the cross-breeding of the Mmp knockout animals; and Frances Sherwood-Brock for her helpful comments on the manuscript. The BeAn strain of TMEV was a generous gift of Dr. Howard L. Lipton, Department of Microbiology–Immunology, University of Illinois, Chicago, USA. Florian Hansmann and Vanessa Herder were supported by scholarships from the National Academic Research Foundation (Germany) or the Ministry of Lower Saxony (Germany), respectively. Reiner Ulrich received a grant from the Centre for Systems Neuroscience Hannover (Germany). The authors have no conflicting financial interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1 includes the primer sequences and reaction conditions used for qPCR. Supplementary Table 2 and Supplementary Fig. 1 show the transcriptional changes related to the canonical Metacore™ cell adhesion and extracellular matrix-remodeling pathway. The canonical cell adhesion and ECM remodeling map was taken from the Metacore™ database (GeneGo, St. Joseph, MO, USA). Supplementary Table 3 shows probe sets, official gene symbols, and fold changes along with the respective p-values of all Mmps and Timps included in the Affymetrix mouse genome 430 2.0 array. Supplementary Table 4 displays a comparison of the fold changes detected by gene-expression microarray and RT-qPCR, respectively.
Rights and permissions
About this article
Cite this article
Hansmann, F., Herder, V., Kalkuhl, A. et al. Matrix metalloproteinase-12 deficiency ameliorates the clinical course and demyelination in Theiler’s murine encephalomyelitis. Acta Neuropathol 124, 127–142 (2012). https://doi.org/10.1007/s00401-012-0942-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-012-0942-3