Abstract
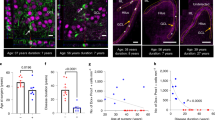
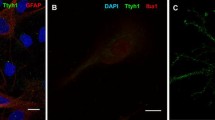
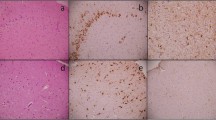
The amygdala displays neuronal cell loss and gliosis in human temporal lobe epilepsy (TLE). Therefore, we investigated a certain type of gliosis, called satellitosis, in the lateral amygdala (LA) of TLE patients with Ammon’s horn sclerosis (AHS, n = 15) and non-AHS (n = 12), and in autopsy controls. Satellite cells were quantified using light and electron microscopy at the somata of Nissl-stained and glutamic acid decarboxylase-negative projection neurons, and their functional properties were studied using electrophysiology. Non-AHS cases suffered from ganglioglioma, cortical dysplasia, Sturge–Weber syndrome, astrocytoma WHO III–IV, Rasmussen’s encephalitis, cerebral infarction and perinatal brain damage. TLE cases with AHS had a more prominent satellitosis as compared to non-AHS and/or autopsy cases, which correlated with epilepsy duration but not age. At ultrastructural level, the predominant type of satellite cells occurring in both AHS and non-AHS cases displayed a dark cytoplasm and an irregularly shaped dark nucleus, whereas perineuronal glial cells with a light cytoplasm and light oval nucleus were much rarer. Satellite cells expressed time- and voltage-dependent transmembrane currents as revealed by patch-clamp recordings typical for ‘complex’ glia, although only 44% of satellite cells were immunostained for the chondroitin sulfate proteoglycan NG2. Together, the perineuronal cells described here were a heterogenous cell population regarding their NG2 expression, although they resembled NG2 cells rather than bona fide oligodendrocytes and astrocytes based on their ultrastructural and electrophysiological characteristics. Thus, perineuronal satellitosis as studied in the LA seems to be a hallmark of AHS-associated TLE pathology in patients suffering from intractable epilepsy.







Similar content being viewed by others
References
Akopian G, Kuprijanova E, Kressin K, Steinhäuser C (1997) Analysis of ion channel expression by astrocytes in red nucleus brain stem slices of the rat. Glia 19:234–246. doi:10.1002/(SICI)1098-1136(199703)19:3<234::AID-GLIA6>3.0.CO;2-Z
Aliashkevich AF, Yilmazer-Hanke D, Van Roost D, Mundhenk B, Schramm J, Blümcke I (2003) Cellular pathology of amygdala neurons in human temporal lobe epilepsy. Acta Neuropathol 106:99–106. doi:10.1007/s00401-003-0707-0
Amaral DG, Price JL, Pitkänen A, Carmichael S (1992) Anatomical organization of the primate amygdaloid complex. In: Aggleton JP (ed) The amygdala: neurobiological aspects of emotion, memory, and mental dysfunction. Wiley-Liss, New York, pp 1–66
Armstrong DD (2005) Epilepsy-induced microarchitectural changes in the brain. Pediatr Dev Pathol 8:607–614. doi:10.1007/s10024-005-0054-3
Aroniadou-Anderjaska V, Fritsch B, Qashu F, Braga MF (2008) Pathology and pathophysiology of the amygdala in epileptogenesis and epilepsy. Epilepsy Res 78:102–116. doi:10.1016/j.eplepsyres.2007.11.011
Avoli M, Louvel J, Pumain R, Kohling R (2005) Cellular and molecular mechanisms of epilepsy in the human brain. Prog Neurobiol 77:166–200. doi:10.1016/j.pneurobio.2005.09.006
Binder DK, Steinhauser C (2006) Functional changes in astroglial cells in epilepsy. Glia 54:358–368. doi:10.1002/glia.20394
Blümcke I, Thom M, Wiestler OD (2002) Ammon’s horn sclerosis: a maldevelopmental disorder associated with temporal lobe epilepsy. Brain Pathol 12:199–211
Braak H, Braak E (1983) Neuronal types in the basolateral amygdaloid nuclei of man. Brain Res Bull 11:349–365. doi:10.1016/0361-9230(83)90171-5
Butt AM, Hamilton N, Hubbard P, Pugh M, Ibrahim M (2005) Synantocytes: the fifth element. J Anat 207:695–706. doi:10.1111/j.1469-7580.2005.00458.x
Chittajallu R, Aguirre A, Gallo V (2004) NG2-positive cells in the mouse white and grey matter display distinct physiological properties. J Physiol 561:109–122. doi:10.1113/jphysiol.2004.074252
Clusmann H, Kral T, Schramm J (2006) Present practice and perspective of evaluation and surgery for temporal lobe epilepsy. Zentralbl Neurochir 67:165–182. doi:10.1055/s-2006-942144
da Silva AV, Houzel JC, Targas Yacubian EM et al (2006) Dysmorphic neurons in patients with temporal lobe epilepsy. Brain Res 1072:200–207. doi:10.1016/j.brainres.2005.10.088
de Lanerolle NC, Lee TS (2005) New facets of the neuropathology and molecular profile of human temporal lobe epilepsy. Epilepsy Behav 7:190–203. doi:10.1016/j.yebeh.2005.06.003
Eid T, Thomas MJ, Spencer DD et al (2004) Loss of glutamine synthetase in the human epileptogenic hippocampus: possible mechanism for raised extracellular glutamate in mesial temporal lobe epilepsy. Lancet 363:28–37. doi:10.1016/S0140-6736(03)15166-5
Eriksson SH, Nordborg C, Rydenhag B, Malmgren K (2005) Parenchymal lesions in pharmacoresistant temporal lobe epilepsy: dual and multiple pathology. Acta Neurol Scand 112:151–156. doi:10.1111/j.1600-0404.2005.00467.x
Fauser S, Schulze-Bonhage A, Honegger J et al (2004) Focal cortical dysplasias: surgical outcome in 67 patients in relation to histological subtypes and dual pathology. Brain 127(Pt 11):2406–2418. doi:10.1093/brain/awh277
Gabriel S, Njunting M, Pomper JK et al (2004) Stimulus and potassium-induced epileptiform activity in the human dentate gyrus from patients with and without hippocampal sclerosis. J Neurosci 24:10416–10430. doi:10.1523/JNEUROSCI.2074-04.2004
Gloor P (1997) The amygdaloid system. In: Gloor P (ed) The temporal lobe and limbic system. Oxford University Press, Oxford, pp 591–721
Gonçalves CA, Leite MC, Nardin P (2008) Biological and methodological features of the measurement of S100B, a putative marker of brain injury. Clin Biochem 41:755–763. doi:10.1016/j.clinbiochem.2008.04.003
Hinterkeuser SW, Schröder G, Hager G et al (2000) Astrocytes in the hippocampus of patients with temporal lobe epilepsy display changes in potassium conductances. Eur J Neurosci 12:2087–2096. doi:10.1046/j.1460-9568.2000.00104.x
Hudson LP, Munoz DG, Miller L, McLachlanRS Girvin JP, Blume WT (1993) Amygdaloid sclerosis in temporal lobe epilepsy. Ann Neurol 33:622–631. doi:10.1002/ana.410330611
Hüttmann K, Yilmazer-Hanke D, Seifert G, Schramm J, Pape HC, Steinhäuser C (2006) Molecular and functional properties of neurons in the human lateral amygdala. Mol Cell Neurosci 31:210–217. doi:10.1016/j.mcn.2005.09.011
Jabs R, Pivneva T, Huttmann K, Wyczynski A, Nolte C, Kettenmann H, Steinhauser C (2005) Synaptic transmission onto hippocampal glial cells with hGFAP promoter activity. J Cell Sci 118:3791–3803. doi:10.1242/jcs.02515
Kalnins RM, McIntosh A, Saling MM, Berkovic SF, Jackson GD, Briellmann RS (2004) Subtle microscopic abnormalities in hippocampal sclerosis do not predict clinical features of temporal lobe epilepsy. Epilepsia 45:940–947. doi:10.1111/j.0013-9580.2004.57203.x
Kälviäinen R, Salmenperä T, Partanen K, Vainio P, Riekkinen P Sr, Pitkänen A (1997) MRI volumetry and T2 relaxometry of the amygdala in newly diagnosed and chronic temporal lobe epilepsy. Epilepsy Res 28:39–50. doi:10.1016/S0920-1211(97)00029-6
Kasper BS, Stefan H, Paulus W (2003) Microdysgenesis in mesial temporal lobe epilepsy: a clinicopathological study. Ann Neurol 54:501–506. doi:10.1002/ana.10694
Kaufman DL, Houser CR, Tobin AJ (1991) Two forms of the gamma-aminobutyric acid synthetic enzyme glutamate decarboxylase have distinct intraneuronal distributions and cofactor interactions. J Neurochem 56:720–723. doi:10.1111/j.1471-4159.1991.tb08211.x
Lin SC, Bergles DE (2002) Physiological characteristics of NG2-expressing glial cells. J Neurocytol 31:537–549. doi:10.1023/A:1025799816285
Lin SC, Huck JH, Roberts JD, Macklin WB, Somogyi P, Bergles DE (2005) Climbing fiber innervation of NG2-expressing glia in the mammalian cerebellum. Neuron 46:773–785. doi:10.1016/j.neuron.2005.04.025
Luskin MB, Parnavelas JG, Barfield JA (1993) Neurons astrocytes and oligodendrocytes of the rat cerebral cortex originate from separate progenitor cells: an ultrastructural analysis of clonally related cells. J Neurosci 13:1730–1750
Mathern GW, Adelson PD, Cahan LD, Leite JP (2002) Hippocampal neuron damage in human epilepsy: Meyer’s hypothesis revisited. Prog Brain Res 135:237–251. doi:10.1016/S0079-6123(02)35023-4
Matthias K, Kirchhoff F, Seifert G, Hüttmann K, Matyash M, Kettenmann H, Steinhäuser C (2003) Segregated expression of AMPA-type glutamate receptors and glutamate transporters defines distinct astrocyte populations in the mouse hippocampus. J Neurosci 23:1750–1758
Miller LA, McLachlan RS, Bouwer MS, Hudson LP, Munoz MG (1994) Amygdalar sclerosis: preoperative indicators and outcome after temporal lobe lobectomy. J Neurol Neurosurg Psychiatry 57:1099–1105. doi:10.1136/jnnp.57.9.1099
Monzon-Mayor M, Yanes C, James JL, Sturrock RR (1990) An ultrastructural study of the development of astrocytes in the midbrain of the lizard. J Anat 170:33–41
Nishiyama A (2007) Polydendrocytes: NG2 cells with many roles in development and repair of the CNS. Neuroscientist 13:62–76. doi:10.1177/1073858406295586
Paukert M, Bergles DE (2006) Synaptic communication between neurons and NG2+ cells. Curr Opin Neurobiol 16:515–521. doi:10.1016/j.conb.2006.08.009
Peters A (2004) A fourth type of neuroglial cell in the adult central nervous system. J Neurocytol 33:345–357. doi:10.1023/B:NEUR.0000044195.64009.27
Peters A, Sethares C (2004) Oligodendrocytes, their progenitors and other neuroglial cells in the aging primate cerebral cortex. Cereb Cortex 14:995–1007. doi:10.1093/cercor/bhh060
Peters A, Palay LS, Webster HF (1976) The neuroglial cells. In: Peters A, Palay LS, Webster HF (eds) The fine structure of the nervous system. The neurons and supporting cells. Saunders, Philadelphia, pp 231–263
Pitkänen A, Tuunanen J, Kälviäinen R, Partanen K, Salmenperä T (1998) Amygdala damage in experimental and human temporal lobe epilepsy. Epilepsy Res 32:233–253. doi:10.1016/S0920-1211(98)00055-2
Schools GP, Zhou M, Kimelberg HK (2003) Electrophysiologically “complex” glial cells freshly isolated from the hippocampus are immunopositive for the chondroitin sulfate proteoglycan NG2. J Neurosci Res 73:765–777. doi:10.1002/jnr.10680
Seifert G, Huttmann K, Schramm J, Steinhauser C (2004) Enhanced relative expression of glutamate receptor 1 flip AMPA receptor subunits in hippocampal astrocytes of epilepsy patients with Ammon’s horn sclerosis. J Neurosci 24:1996–2003. doi:10.1523/JNEUROSCI.3904-03.2004
Seifert G, Schilling K, Steinhauser C (2006) Astrocyte dysfunction in neurological disorders: a molecular perspective. Nat Rev Neurosci 7:194–206. doi:10.1038/nrn1870
Sloviter RS, Kudrimoti HS, Laxer KD et al (2004) “Tectonic” hippocampal malformations in patients with temporal lobe epilepsy. Epilepsy Res 59:123–153. doi:10.1016/j.eplepsyres.2004.04.001
Sorvari H, Miettinen R, Soininen H, Pitkanen A (1998) Parvalbumin-immunoreactive neurons make inhibitory synapses on pyramidal cells in the human amygdala: a light and electron microscopic study. Neurosci Lett 217:93–96. doi:10.1016/0304-3940(96)13067-6
Steinhäuser C, Jabs R, Kettenmann H (1994) Properties of GABA and glutamate responses in identified glial cells of the mouse hippocampal slice. Hippocampus 4:19–36. doi:10.1002/hipo.450040105
Streit WJ (2005) Microglial cells. In: Kettenmann H, Ransom BR (eds) Neuroglia, 2nd edn. Oxford University Press, New York, pp 60–71
Vijayan VK, Zhou SS, Russell MJ, Geddes J, Ellis W, Cotman CW (1993) Perineuronal satellitosis in the human hippocampal formation. Hippocampus 3:239–250. doi:10.1002/hipo.450030215
Wennström M, Hellsten J, Ekdahl CT, Tingström A (2003) Electroconvulsive seizures induce proliferation of NG2-expressing glial cells in adult rat hippocampus. Biol Psychiatry 54:1015–1024. doi:10.1016/S0006-3223(03)00693-0
Wennström M, Hellsten J, Tingström A (2004) Electroconvulsive seizures induce proliferation of NG2-expressing glial cells in adult rat amygdala. Biol Psychiatry 55:464–471. doi:10.1016/j.biopsych.2003.11.011
Wolf HK, Aliashkevich AF, Blümcke I, Wiestler OD, Zentner J (1997) Neuronal loss and gliosis of the amygdaloid nucleus in temporal lobe epilepsy. Acta Neuropathol 93:606–610. doi:10.1007/s004010050658
Wyler AR, Dohan FC, Schweitzer JB, Berry AD (1992) A grading system for mesial temporal pathology (hippocampal sclerosis) from anterior temporal lobectomy. J Epilepsy 5:220–225. doi:10.1016/S0896-6974(05)80120-3
Yilmazer-Hanke DM, Wolf HK, Schramm J, Elger CE, Wiestler OD, Blümcke I (2000) Subregional pathology of the amygdala complex and entorhinal region in surgical specimens from patients with pharmacoresistant temporal lobe epilepsy. J Neuropathol Exp Neurol 59:907–920
Yilmazer-Hanke DM, Faber-Zuschratter H, Blümcke I et al (2007) Axo-somatic inhibition of projection neurons in the lateral nucleus of amygdala in human temporal lobe epilepsy: an ultrastructural study. Exp Brain Res 177:384–399. doi:10.1007/s00221-006-0680-7
Yokota O, Tsuchiya K, Hayashi M et al (2008) Glial clusters and perineuronal glial satellitosis in the basal ganglia of neurofibromatosis type 1. Acta Neuropathol 116:57–66. doi:10.1007/s00401-008-0390-2
Acknowledgments
Ms. Röhl and Ms. Schneider (Institute of Anatomy, Magdeburg, Germany) and Mr. O’Leary (Department of Anatomy, Cork, Ireland) are acknowledged for expert technical assistance, Ms. Stausberg and Ms. Ullmann for their aid with the clinical data. We thank C. Mawrin and K. Dietzmann (Institute of Neuropathology, University of Magdeburg Germany), and RAI de Vos (Laboratorium voor Pathologie Enschede Netherlands) for providing autopsy material. This study was supported by the Deutsche Forschungsgemeinschaft (SFB/TR3, TP C3 to DYH and CS, TP 1 to JS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Faber-Zuschratter, H., Hüttmann, K., Steinhäuser, C. et al. Ultrastructural and functional characterization of satellitosis in the human lateral amygdala associated with Ammon’s horn sclerosis. Acta Neuropathol 117, 545–555 (2009). https://doi.org/10.1007/s00401-009-0504-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-009-0504-5