Abstract
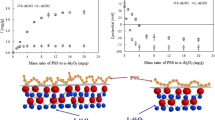
Adsorption of sodium dodecyl sulfate (SDS) onto large α-alumina beads was systematically analyzed as functions of pH and NaCl concentration. The maximum adsorption density of SDS onto α-Al2O3 increases with decreasing pH due to the increase in equilibrium surface charge. Adsorption isotherms at different salt concentrations demonstrated a common intersection point (CIP) corresponding to charge neutralization. The obtained comprehensive data clarified the applicability of two-step adsorption and four-region models to describe the adsorption isotherms of SDS onto α-Al2O3 beads. Also, proton adsorption isotherms upon SDS uptake onto α-Al2O3 can be fitted by two-step adsorption model with almost the same parameters of surfactant adsorption for 0.001 and 0.01 M NaCl. Our finding indicates that proton adsorption takes place onto the adsorbed SDS. After passing through CIP, proton adsorption onto α-Al2O3 increases at low ionic strength while the amount of proton adsorption does not change for 0.1 M NaCl. Adsorption amount of proton as a function of SDS adsorption shows that only hemimicelles are plausible for case of low ionic strength. The increase in the proton adsorption is not significant at high ionic strength, suggesting the presence of admicelles on the surface of α-Al2O3 beads.






Similar content being viewed by others
References
Atkin R, Craig VSJ, Wanless EJ, Biggs S (2003) Adv Colloid Interf Sci 103:219–304
Esumi K, Yamanaka Y (1995) J Colloid Interface Sci 172:116–120
Goloub TP, Koopal LK (1997) Langmuir 13:673–681
Lee EM, Koopal LK (1996) J Colloid Interface Sci 177:478–489
Paria S (2008) Adv Colloid Interf Sci 138:24–58
Paria S, Khilar KC (2004) Adv Colloid Interf Sci 110:75–95
Zhang R, Somasundaran P (2006) Adv Colloid Interf Sci 123–126:213–229
Kobayashi M, Nanaumi H, Muto Y (2009) Colloids Surf A Physicochem Eng Asp 347:2–7
Tulpar A, Ducker WA (2004) J Phys Chem B 108:1667–1676
Gaudin AMF, Fuerstenau DW (1955) Trans AIME 202:958–962
Fuerstenau DW (1956) J Phys Chem 60:981–985
Fuerstenau DW, Modi HJ (1959) J Electrochem Soc 106:336–341
Fuerstenau DW, Wakamatsu T (1975) Faraday Discuss Chem Soc 59:157–168
Wakamatsu T, Fuerstenau DW (1968) Advances in chemistry. Am Chem Soc 79:161–172
Somasundaran P, Fuerstenau DW (1966) J Phys Chem 70:90–96
Somasundaran P, Healy TW, Fuerstenau DW (1964) J Phys Chem 68:3562–3566
Harwell JH, Hoskins JC, Schechter RS, Wade WH (1985) Langmuir 1:251–262
Bitting D, Harwell JH (1987) Langmuir 3:500–511
Yeskie MA, Harwell JH (1988) J Phys Chem 92:2346–2352
Zhu B-Y, Gu T (1991) Adv Colloid Interf Sci 37:1–32
Fan A, Somasundaran P, Turro NJ (1997) Langmuir 13:506–510
Hankins NP, O’Have JH, Harwell JH (1996) Ind Eng Chem Res 35:2844–2855
Gu T, Zhu B-Y (1990) Colloids Surf 44:81–87
Sabatini DA, Knox RC (1992) Transport and remediation of subsurface contaminants, ACS symposium series 491. American Chemical Society, Washington DC
Rosen MJ, Kunjappu JT (2012) Surfactants and interfacial phenomena, 4th edn. Wiley, USA
Alila S, Aloulou F, Beneventi D, Boufi S (2007) Langmuir 23:3723–3731
Alila S, Boufi S, Belgacem MN, Beneventi D (2005) Langmuir 21:8106–8113
Aloulou F, Boufi S, Beneventi D (2004) J Colloid Interface Sci 280:350–358
Chandar P, Somasundaran P, Turro NJ (1987) J Colloid Interface Sci 117:31–46
Esumi K, Nagahama T, Meguro K (1991) Colloids Surf 57:149–160
Esumi K, Otsuka H, Meguro K (1991) J Colloid Interface Sci 142:582–588
Bohmer MR, Koopal LK (1992) Langmuir 8:2649–2659
Bohmer MR, Koopal LK (1992) Langmuir 8:2660–2665
Bohmer MR, Koopal LK (1992) Langmuir 8:1594–1602
Koopal LK, Lee EM, Böhmer MR (1995) J Colloid Interface Sci 170:85–97
Fuerstenau DW, Jang HM (1991) Langmuir 7:3138–3143
Goloub TP, Koopal LK, Bijsterbosch BH, Sidorova MP (1996) Langmuir 12:3188–3194
Attaphong C, Asnachinda E, Charoensaeng A, Sabatini DA, Khaodhiar S (2010) J Colloid Interface Sci 344:126–131
Lopata JJ, Werts KM, Scamehorn JF, Harwell JH, Grady BP (2010) J Colloid Interface Sci 342:415–426
Somasundaran P, Kunjappu JT (1989) Colloids Surf 37:245–268
Partyka S, Rudzinski W, Brun B, Clint JH (1989) Langmuir 5:297–304
Mączka E, Luetzenkirchen J, Kosmulski M (2013) J Colloid Interface Sci 393:228–233
Pham TD, Kobayashi M, Adachi Y (2013) Colloids Surf A Physicochem Eng Asp 436:148–157
Hayashi K (1975) Anal Biochem 67:503–506
Mukerjee P (1956) Anal Chem 28:870–873
Hummel DO (2000) Handbook of surfactant analysis. Wiley, New York
Koopal LK, Goloub TP, Davis TA (2004) J Colloid Interface Sci 275:360–367
Nizri G, Lagerge S, Kamyshny A, Major DT, Magdassi S (2008) J Colloid Interface Sci 320:74–81
Thongngam M, McClements DJ (2004) Langmuir 21:79–86
Zhu B-Y, Gu T (1989) J Chem Soc Faraday Trans 1 Phys Chem Condens Phases 85:3813–3817
Adak A, Pal A, Bandyopadhyay M (2006) Colloids Surf A Physicochem Eng Asp 277:63–68
Hu K, Bard AJ (1997) Langmuir 13:5418–5425
Ayame A, Uchida Y, Ono H, Miyamoto M, Sato T, Hayasaka H (2003) Appl Catal A Gen 244:59–70
Barrett EP, Joyner LG, Halenda PP (1951) J Am Chem Soc 73:373–380
Franks GV, Gan Y (2007) J Am Ceram Soc 90:3373–3388
Song S-H, Koelsch P, Weidner T, Wagner MS, Castner DG (2013) Langmuir 29:12710–12719
Acknowledgments
We are grateful for the financial support from JSPS KAKENHI (22248025, 23688027). Tien Duc Pham would like to thank the financial support from Vietnamese government scholarship (approved by VIED-MOET) for the doctoral study abroad. The authors thank the useful comments from two anonymous reviewers to improve the original manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 132 kb)
Rights and permissions
About this article
Cite this article
Pham, T.D., Kobayashi, M. & Adachi, Y. Adsorption of anionic surfactant sodium dodecyl sulfate onto alpha alumina with small surface area. Colloid Polym Sci 293, 217–227 (2015). https://doi.org/10.1007/s00396-014-3409-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-014-3409-3