Abstract
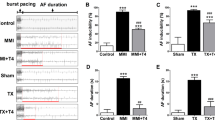
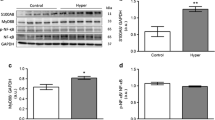
Atrial fibrillation (AF) is a common complication in hyperthyroidism. Earlier studies demonstrate that thyroid hormone decreases L-type calcium channel (LCC) current expression with resultant shortening of action potential duration (APD), providing a substrate for AF. The aim of this study was to investigate the potential mechanism underlying the regulatory effect of thyroid hormone on LCC. In a hyperthyroid rat model, thyroid hormone (triiodothyronine [T3]) administration down-regulated atrial LCC expression. In vitro, treatment of murine atrial myocytes (HL-1) with T3 decreased the expression of LCC and its current, resulting in abbreviation of APD. Furthermore, T3 inhibited the activation of cyclic AMP response element (CRE)-binding protein (CREB), including phosphorylation at Ser133 and its nuclear translocation. Transient transfection studies in HL-1 cells indicated that T3 reduced LCC promoter activity. Deletion and mutation analysis of the LCC promoter region along with chromatin immunoprecipitation using anti-CREB antibody showed that CRE was essential for T3-mediated LCC gene expression. Transfection of dominant-negative CREB (mutated Ser133) and mutant thyroid hormone receptor (TR, mutated Cys51) abolished the T3-dependent effects, suggesting an association between both transcriptional factors. Co-immunoprecipitation documented an increased binding of TR with CREB after T3 treatment. The transcriptional cross-talk 3 between TR and CREB bound to CRE mediates T3-inhibited CREB activity and LCC expression. Thyroid hormone-induced TR binding of CREB inhibits CREB activity and LCC current expression, which may contribute to AF. These findings provide an important mechanistic insight into hyperthyroidism-induced AF.








Similar content being viewed by others
References
Auer J, Scheibner P, Mische T, Langsteger W, Eber O, Eber B (2001) Subclinical hyperthyroidism as a risk factor for atrial fibrillation. Am Heart J 142:838–842. doi:10.1067/mhj.2001.119370
Bosch RF, Zeng X, Grammer JB, Popovic K, Mewis C, Kuhlkamp V (1999) Ionic mechanisms of electrical remodeling in human atrial fibrillation. Cardiovasc Res 44:121–131. doi:10.1016/S0008-6363(99)00178-9
Burstein B, Libby E, Calderone A, Nattel S (2008) Differential behaviors of atrial versus ventricular fibroblasts: a potential role for platelet-derived growth factor in atrial-ventricular remodeling differences. Circulation 117:1630–1641. doi:10.1161/CIRCULATIONAHA.107.748053
Burstein B, Nattel S (2008) Atrial fibrosis: mechanism and clinical relevance in atrial fibrillation. J Am Coll Cardiol 51:802–809. doi:10.1016/j.jacc.2007.09.064
Cameiro-Ramos MS, Diniz GP, Nadu AP, Almeida J, Vieira RLP, Santos RAS, Barreto-Chaves MLM (2010) Blockage of angiotensin II type 2 receptor prevents thyroxine-mediated cardiac hypertrophy by blocking Akt activation. Basic Res Cardiol 105:325–335. doi:10.1007/s00395-010-0089-0
Chen RN, Huang YH, Lin YC, Yeh CT, Liang Y, Chen SL, Lin KH (2008) Thyroid hormone promotes cell invasion through activation of furin expression 25 in human hepatoma cell lines. Endocrinology 149:3817–3831. doi:10.1210/en.2007-0989
Chen WJ, Lin KH, Lee YS (2000) Molecular characterization of myocardial fibrosis during hypothyroidism. Mol Cell Endocrinol 162:45–55. doi:10.1016/S0303-7207(00)00203-3
Chen WJ, Pang JHS, Lin KH, Lee DY, Hsu LA, Kuo CT (2010) Propylthiouracil, independent of its antithyroid effect, promotes VSMC differentiation via PTEN induction. Basic Res Cardiol 105:19–28. doi:10.1007/s00395-009-0045-z
Chen YC, Chen SA, Chen YJ, Chang MS, Chan P, Lin CI (2002) Effects of thyroid hormone on the arrhythmogenic activity of pulmonary vein cardiomyocytes. J Am Coll Cardiol 39:366–372. doi:10.1016/S0735-1097(01)01731-4
Claycomb WC, Lanson NA Jr, Stallworth BS, Egeland DB, Delcarpio JB, Bahinski A, Izzo NJ Jr (1998) HL-1 cells: a cardiac muscle cell line that contracts and retains phenotypic characteristics of the adult cardiomyocyte. Proc Natl Acad Sci USA 95:2979–2984
Diniz GP, Cameiro-Ramos MS, Barreto-Chaves MLM (2009) Angiotensin type 1 receptor mediates thyroid hormone-induced cardiomyocyte hypertrophy through the Akt/GSK-3β/mTOR signaling pathway. Basic Res Cardiol 104:653–667. doi:10.1007/s00395-009-0043-1
Glass CK (1994) Differential recognition of target genes by nuclear receptor monomers, dimers, and heterodimers. Endocr Rev 15:391–407. doi:10.1210/edrv-15-3-391
Harvey CB, Williams GR (2002) Mechanism of thyroid hormone action. Thyroid 12:441–446. doi:10.1089/105072502760143791
Hiroi Y, Kim HH, Ying H, Furuya F, Huang Z, Simoncini T, Noma K, Ueki K, Nguyen NH, Scanlan TS, Moskowitz MA, Cheng SY, Liao JK (2006) Rapid nongenomic actions of thyroid hormone. Proc Natl Acad Sci USA 103:14104–14109. doi:10.1073/pnas.0601600103
Johannessen M, Delghandi MP, Moens U (2004) What turns CREB on? Cell Signal 16:1211–1227. doi:10.1016/j.cellsig.2004.05.001
Kahaly GJ, Dillmann WH (2005) Thyroid hormone action in the heart. Endocr Rev 26:704–728. doi:10.1210/er.2003-0033
Klein I, Ojamaa K (2001) Thyroid hormone and the cardiovascular system. N Engl J Med 344:501–509
Komiya N, Isomoto S, Nakao K, Hayano M, Yano K (2002) Electrophysiological abnormalities of the atrial muscle in patients with paroxysmal atrial fibrillation associated with hyperthyroidism. Clin Endocrinol 56:39–44. doi:27101046/j0300-0664200101459x
Laflamme L, Hamann G, Messier N, Maltais S, Langlois MF (2002) RXR acts as a coregulator in the regulation of genes of the hypothalamo-pituitary axis by thyroid hormone receptors. J Mol Endocrinol 29:61–72. doi:10.1677/jme.0.0290061
Maia AL, Harney JW, Larsen PR (1996) Is there a negative TRE in the luciferase reporter cDNA? Thyroid 6:325–328. doi:10.1089/thy.1996.6.325
Mendez-Pertuz M, Sanchez-Pacheco A, Aranda A (2003) The thyroid hormone receptor antagonizes CREB-mediated transcription. EMBO J 22:3102–3112. doi:10.1093/emboj/cdg295
Muller FU, Lewin G, Baba HA, Boknik P, Fabritz L, Kirchhefer U, Kirchhof P, Loser K, Matus M, Neumann J, Riemann B, Schmitz W (2005) Heart-directed expression of a human cardiac isoform of cAMP-response element modulator in transgenic mice. J Biol Chem 280:6906–6914. doi:10.1074/jbc.M407864200
Nattel S (2002) New ideas about atrial fibrillation 50 years on. Nature 415:219–226. doi:10.1038/415219a
Qi YX, Yeh YH, Xiao L, Burstein B, Maguy A, Chartier D, Villeneuve LR, Brundel BJJM, Dobrev D, Nattel S (2008) Cellular signaling underlying atrial tachycardia remodeling of L-type calcium current. Circ Res 103:845–854. doi:10.1161/CIRCRESAHA.108.175463
Shaywitz AJ, Greenberg ME (1999) CREB: a stimulus-induced transcription factor activated by a diverse array of extracellular signals. Annu Rev Biochem 68:821–861
Sawin CT, Geller A, Wolf PA, Belanger AJ, Baker E, Bacharach P, Wilson PM, Benjamin EJ, D’Agostino RB (1994) Low serum thyrotropin concentrations as a risk factor for atrial fibrillation in older persons. N Engl J Med 311:1249–1252
Tsai CT, Wang DL, Chen WP, Hwang JJ, Hsieh CS, Hsu KL, Tseng CD, Lai LP, Tseng YZ, Chiang FT, Lin JL (2007) Angiotensin II increases expression of α1C subunit of L-type calcium channel through a reactive oxygen species and cAMP response element-binding protein-dependent pathway in HL-1 myocytes. Circ Res 100:1476–1485. doi:10.1161/01.RES.0000268497.93085.e1
Van Wagoner DR, Nerbonne JM (2000) Molecular basis of electrical remodeling in atrial fibrillation. J Mol Cell Cardiol 32:1101–1117. doi:10.1006/jmcc.2000.1147
Watanabe H, Ma M, Washizuka T, Komura S, Yoshida T, Hosaka Y, Hatada K, Chinushi M, Yamamoto T, Watanabe K, Aizawa Y (2003) Thyroid hormone regulates mRNA expression and currents of ion channels in rat atrium. Biochem Biophys Res Commun 308:439–444. doi:10.1016/S0006-291X(03)01420-7
Wikstrom L, Johansson C, Salto C, Barlow C, Barros AC, Baas F, Forrest D, Thoren P, Vennstrom B (1998) Abnormal heart rate and body temperature in mice lacking thyroid hormone receptor α1. EMBO J 17:455–461. doi:10.1093/emboj/17.2.455
Yue L, Feng J, Gaspo R, Li GR, Wang Z, Nattel S (1997) Ionic remodeling underlying action potential changes in a canine model of atrial fibrillation. Circ Res 81:512–525
Yue L, Melnyk P, Gaspo R, Wang Z, Nattel S (1999) Molecular mechanisms underlying ionic remodeling in a dog model of atrial fibrillation. Circ Res 84:776–784
Yu WC, Lee SH, Tai CT, Tsai CF, Hsieh MH, Chen CC, Ding YA, Chang MS, Chen SA (1999) Reversal of atrial electrical remodeling following cardioversion of long-standing atrial fibrillation in man. Cardiovasc Res 42:470–476. doi:10.1016/S0008-6363(99)00030-9
Zhang J, Lazar MA (2000) The mechanism of action of thyroid hormones. Annu Rev Physiol 62:439–466
Acknowledgments
This work was supported by grants from the Chang Gung Research Grant Foundation [G32102 and G331023] and National Science Council Grants [NSC-94-2314-B-182A-198]. We thank Mr. Chih-Chun Chen for his technical assistance in confocal microscope.
Author information
Authors and Affiliations
Corresponding author
Additional information
W.-J. Chen and Y.-H. Yeh contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, WJ., Yeh, YH., Lin, KH. et al. Molecular characterization of thyroid hormone-inhibited atrial L-type calcium channel expression: implication for atrial fibrillation in hyperthyroidism. Basic Res Cardiol 106, 163–174 (2011). https://doi.org/10.1007/s00395-010-0149-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00395-010-0149-5