Abstract
Purpose
Treatment for a large abdominal wall defect remains challenging. The aim of this study was to optimize tissue engineering therapy of muscle constructs using a rat model.
Methods
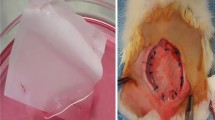
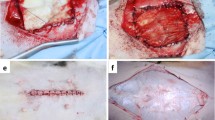
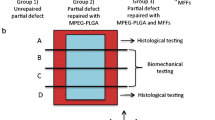
Experimental abdominal wall defects were created in Wister rats. The animal model was divided into three groups: collagen sponge (CS), hybrid scaffold (HS) and hybrid scaffold containing bone marrow liquid (HSBM). Hybrid scaffolds comprised collagen sponge and poly L-lactide (PLLA) sheets. Abdominal wall defects were covered by three kinds of sheets. Thereafter, the bone marrow liquid was spread onto the sheets. Rats were killed at 4, 8, and 16 weeks. Pathological examinations were performed using hematoxylin–eosin and desmin antibody staining.
Results
The CS group showed abdominal hernia, whereas the HS and HSBM groups did not. Vascular formation was confirmed in all groups. Muscle tissue was recognized at the marginal area of the sheet only in the HSBM group.
Conclusion
The HS and HSBM groups show a greater intensity than the CS group. Muscle tissue regeneration is solely recognized in the HSBM group. Our experimental data suggest that the triad of scaffold, cell, and growth factor is fundamental for ideal biomaterials. The HSBM may be useful for reconstruction of abdominal wall defects.




Similar content being viewed by others
References
Ramirez OM, Ruas E, Dellon AL (1990) “Components separation” method for closure of abdominal-wall defects: an anatomic and clinical study. Plast Reconstr Surg 86:519–526
de Vries Reilingh TS, van Goor H, Rosman C et al (2003) “Components separation technique” for the repair of large abdominal wall hernias. J Am Coll Surg 196:32–37
Levi DM, Tzakis AG, Kato T et al (2003) Transplantation of the abdominal wall. Lancet 361:2173–2176. doi:10.1016/S0140-6736(03)13769-5
Siemionow M (2015) Vascularized composite allotransplantation: a new concept in musculoskeletal regeneration. J Mater Sci Mater Med 26:266. doi:10.1007/s10856-015-5601-5
Rice RD, Ayubi FS, Shaub ZJ et al (2009) Comparison of Surgisis®, AlloDerm®, and Vicryl Woven Mesh® grafts for abdominal wall defect repair in an animal model. Aesth Plast Surg 34:290–296. doi:10.1007/s00266-009-9449-2
Porzionato A, Sfriso M, Pontini A et al (2015) Decellularized human skeletal muscle as biologic scaffold for reconstructive surgery. Int J Mol Sci 16:14808–14831. doi:10.3390/ijms160714808
Langer R, Vacanti JP (1993) Tissue engineering. Science 260:920–926
Butler DL, Juncosa-Melvin N, Boivin GP et al (2008) Functional tissue engineering for tendon repair: a multidisciplinary strategy using mesenchymal stem cells, bioscaffolds, and mechanical stimulation. J Orthop Res 26:1–9. doi:10.1002/jor.20456
Howard D, Buttery LD, Shakesheff KM, Roberts SJ (2008) Tissue engineering: strategies, stem cells and scaffolds. J Anat 213:66–72. doi:10.1111/j.1469-7580.2008.00878.x
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126:663–676. doi:10.1016/j.cell.2006.07.024
Caplan AI (2007) Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J Cell Physiol 213:341–347. doi:10.1002/jcp.21200
Sanganalmath SK, Bolli R (2013) Cell therapy for heart failure: a comprehensive overview of experimental and clinical studies, current challenges, and future directions. Circ Res 113:810–834. doi:10.1161/CIRCRESAHA.113.300219
Seiler MJ, Aramant RB (2012) Cell replacement and visual restoration by retinal sheet transplants. Prog Retin Eye Res 31:661–687. doi:10.1016/j.preteyeres.2012.06.003
Lai JY, Chang PY, Lin JN (2003) Body wall repair using small intestinal submucosa seeded with cells. J Pediatr Surg 38:1752–1755. doi:10.1016/S0022-3468(03)00617-1
Nowacki M et al (2015) Blood vessel matrix seeded with cells: a better alternative for abdominal wall reconstruction-a long-term study. BioMed Res Int 2015:890613. doi:10.1155/2015/890613
Inui A, Kokubu T, Makino T et al (2010) Potency of double-layered Poly L-lactic Acid scaffold in tissue engineering of tendon tissue. Int Orthop 34:1327–1332. doi:10.1007/s00264-009-0917-8
Zhang Z, Zhang T, Li J et al (2014) Preparation of poly(l-lactic acid)-modified polypropylene mesh and its antiadhesion in experimental abdominal wall defect repair. J Biomed Mater Res 102:12–21. doi:10.1002/jbm.b.32947
Hashi CK, Derugin N, Janairo RRR et al (2010) Antithrombogenic modification of small-diameter microfibrous vascular grafts. Arterioscler Thromb Vasc Biol 30:1621–1627. doi:10.1161/ATVBAHA.110.208348
Sato Y, Oba T, Natori N, Danjo K (2010) Evaluation of additives required for periodontal disease formulation using basic fibroblast growth factor. Chem Pharm Bull 58:1582–1586. doi:10.1248/cpb.58.1582
Yin H, Price F, Rudnicki MA (2013) Satellite cells and the muscle stem cell niche. Physiol Rev 93:23–67. doi:10.1152/physrev.00043.2011
Yablonka-Reuveni Z (2011) The skeletal muscle satellite cell: still young and fascinating at 50. J Histochem Cytochem 59:1041–1059. doi:10.1369/0022155411426780
Acknowledgments
This work was supported by the Institute of Laboratory Animal Sciences, Kagoshima University (Frontier Science Research Center). We thank Mr. Brian Quinn for comments and help with the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Suzuhigashi, M., Kaji, T., Nakame, K. et al. Abdominal wall regenerative medicine for a large defect using tissue engineering: an experimental study. Pediatr Surg Int 32, 959–965 (2016). https://doi.org/10.1007/s00383-016-3949-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-016-3949-3