Abstract
Background
Definitive and high-quality coverage of large and, in particular, massive skin defects remains a significant challenge in burn as well as plastic and reconstructive surgery because of donor site shortage. A novel and promising approach to overcome these problems is tissue engineering of skin. Clearly, before eventual clinical application, engineered skin substitutes of human origin must be grafted and then evaluated in animal models. For the various tests to be conducted it is indispensable to be able to identify human cells as such in culture and also to distinguish between graft and recipient tissue after transplantation. Here we describe a tool to identify human cells in vitro and in vivo.
Methods
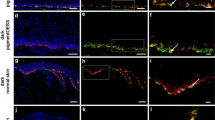
In situ hybridization allows for the detection and localization of specific DNA or RNA sequences in morphologically preserved cells in culture or tissue sections, respectively. We used digoxigenin-labeled DNA probes corresponding to human-specific Alu repeats in order to identify human keratinocytes grown in culture together with rat cells, and also to label split and full thickness skin grafts of human origin after transplantation on immuno-incompetent rats.
Results
Digoxigenin-labeled DNA probing resulted in an intensive nuclear staining of human cells, both in culture and after transplantation onto recipient animals, while recipient animal cells (rat cells) did not stain.
Conclusion
In situ hybridization using primate-specific Alu probes reliably allows distinguishing between cells of human and non-human origin both in culture as well as in histological sections. This method is an essential tool for those preclinical experiments (performed on non-primate animals) that must be conducted before novel tissue engineered skin substitutes might be introduced into clinical practice.




Similar content being viewed by others
References
Böttcher-Haberzeth S, Biedermann T, Reichmann E (2010) Tissue engineering of skin. Burns 36:450–460
Loss M, Wedler V, Kuenzi W, Meuli-Simmen C, Meyer VE (2000) Artificial skin, split-thickness autograft and cultured autologous keratinocytes combined to treat a severe burn injury of 93% of TBSA. Burns 26:644–652
Atiyeh BS, Gunn SW, Hayek SN (2005) State of the art in burn treatment. World J Surg 29:131–148
Berman B, Viera MH, Amini S, Huo R, Jones IS (2008) Prevention and management of hypertrophic scars and keloids after burns in children. J Craniofac Surg 19:989–1006
Deitch EA, Wheelahan TM, Rose MP, Clothier J, Cotter J (1983) Hypertrophic burn scars: an analysis of variables. J Trauma 23:895–898
Montano I, Schiestl C, Schneider J, Pontiggia L, Luginbühl J, Biedermann T, Böttcher S, Braziulis E, Meuli M, Reichmann E (2010) Formation of human capillaries in vitro: the engineering of prevascularized matrices. Tissue Eng Part A 16:269–282
Pontiggia L, Biedermann T, Meuli M, Widmer D, Böttcher-Haberzeth S, Schiestl C, Schneider J, Braziulis E, Montano I, Meuli-Simmen C, Reichmann E (2009) Markers to evaluate the quality and self-renewing potential of engineered human skin substitutes in vitro and after transplantation. J Invest Dermatol 129:480–490
Tharakan S, Pontiggia L, Biedermann T, Böttcher-Haberzeth S, Schiestl C, Reichmann E, Meuli M (2010) Transglutaminases, involucrin, and loricrin as markers of epidermal differentiation in skin substitutes derived from human sweat gland cells. Pediatr Surg Int 26:71–77
Biedermann T, Pontiggia L, Böttcher-Haberzeth S, Tharakan S, Braziulis E, Schiestl C, Meuli M, Reichmann E (2010) Human eccrine sweat gland cells can reconstitute a stratified epidermis. J Invest Dermatol 130:1996–2009
Just L, Timmer M, Tinius J, Stahl F, Deiwick A, Nikkhah G, Bader A (2003) Identification of human cells in brain xenografts and in neural co-cultures of rat by in situ hybridisation with Alu probe. J Neurosci Methods 126:69–77
Kariya Y, Kato K, Hayashizaki Y, Himeno S, Tarui S, Matsubara K (1987) Revision of consensus sequence of human Alu repeats–a review. Gene 53:1–10
Rowold DJ, Herrera RJ (2000) Alu elements and the human genome. Genetica 108:57–72
Worst PK, Valentine EA, Fusenig NE (1974) Formation of epidermis after reimplantation of pure primary epidermal cell cultures from perinatal mouse skin. J Natl Cancer Inst 53:1061–1064
Worst PK, Mackenzie IC, Fusenig NE (1982) Reformation of organized epidermal structure by transplantation of suspensions and cultures of epidermal and dermal cells. Cell Tissue Res 225:65–77
Kim J, Yu W, Kovalski K, Ossowski L (1998) Requirement for specific proteases in cancer cell intravasation as revealed by a novel semiquantitative PCR-based assay. Cell 94:353–362
Selden C, Hodgson H (2004) Cellular therapies for liver replacement. Transpl Immunol 12:273–288
Schieker M, Seitz S, Gülkan H, Nentwich M, Horvath G, Regauer M, Milz S, Mutschler W (2004) Tissue engineering of bone. Integration and migration of human mesenchymal stem cells in colonized contructs in a murine model. Orthopade 33:1354–1360
Hurelbrink CB, Barker RA (2005) Migration of cells from primary transplants of allo- and xenografted foetal striatal tissue in the adult rat brain. Eur J Neurosci 21:1503–1510
Terrovitis JV, Smith RR, Marbán E (2010) Assessment and optimization of cell engraftment after transplantation into the heart. Circ Res 106:479–494
Szmulewicz MN, Novick GE, Herrara RJ (1998) Effects of Alu insertion on gene function. Electrophoresis 19:1260–1264
Warncke B, Valtink M, Weichel J, Engelmann K, Schafer H (2004) Experimental rat model for therapeutic retinal pigment epithelium transplantation–unequivocal microscopic identification of human donor cells by in situ hybridisation of human-specific Alu sequences. Virchows Arch 444:74–81
Acknowledgments
This work was supported by a grant from the European Union (EuroSTEC: LSHB-CT-2006-037409) and by the University of Zurich. We are particularly grateful to the Fondation Gaydoul, and the sponsors of DonaTissue (Therese Meier and Robert Zingg), the Vontobel Foundation and the Werner Spross Foundation for their financial support and their interest in our work.
Conflict of interest
The authors declare that none of them has any conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Weber, A.D., Pontiggia, L., Biedermann, T. et al. Determining the origin of cells in tissue engineered skin substitutes: a pilot study employing in situ hybridization. Pediatr Surg Int 27, 255–261 (2011). https://doi.org/10.1007/s00383-010-2776-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-010-2776-1