Abstract
Purpose
Alterations of the brain microstructure and metabolism have been identified in patients with neurofibromatosis type 1 (NF1). In this study, we analyzed the basal ganglia of NF1 subjects without cognitive delay throughout a combined approach with magnetic resonance spectroscopy (MRS) and diffusion tensor imaging (DTI) in order to better define the metabolic and microstructural characteristics of these regions and, furthermore, to verify if metabolic and microstructural abnormalities may be present in normally developed NF1 patients.
Methods
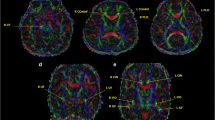
A 3-T MRI with multivoxel MRS and DTI was performed in 14 NF1 patients and eight controls. N-acetyl-aspartate (NAA), choline (Cho), creatine (Cr) values and ratios, fractional anisotropy, and apparent diffusion coefficient (ADC) were calculated, for a total of four regions of interest (ROI) for each hemisphere.
Results
NF1 patients, compared to healthy controls, showed (a) decreased NAA in all the four ROI, (b) increased Cho and decreased Cr in three of the four ROI, (c) decreased NAA/Cho ratio in three ROI, and (d) increased ADC in all the four ROI. A trend of increased ADC was present in three of the four ROI of NF1 patients with unidentified bright objects (UBOs) and younger than 18 years.
Conclusion
These data confirm the presence of neuroaxonal damage with myelin disturbances in NF1 patients. We showed that metabolic and microstructural anomalies can be present in the same time in NF1 patients without developmental delay or cognitive deficits. Relations between brain anomalies, UBOs, and cognitive functions need further studies.

Similar content being viewed by others
References
Ruggieri M, Upadhyaya M, Di Rocco C et al (2009) Neurofibromatosis type 1 and related disorders. In: Ruggieri M, Pascual-Castroviejo I, Di Rocco C (eds) Neurocutaneous disorders: phakomatoses and hamartoneoplastic syndromes. Springer, New York, pp 51–151
Castillo M, Green C, Kwock L et al (1995) Proton MR spectroscopy in patients with neurofibromatosis type 1: evaluation of hamartomas and clinical correlation. AJNR Am J Neuroradiol 16:141–147
Zamboni SL, Loenneker T, Boltshauser E, Martin E, Il'yasov KA (2007) Contribution of diffusion tensor MR imaging in detecting cerebral microstructural changes in adults with neurofibromatosis type 1. AJNR Am J Neuroradiol 28:773–776
Barbier C, Chabernaud C, Barantin L et al (2011) Proton MR spectroscopic imaging of basal ganglia and thalamus in neurofibromatosis type 1: correlation with T2 hyperintensities. Neuroradiology 53:141–148
Chabernaud C, Sirinelli D, Barbier C et al (2009) Thalamo-striatal T2-weighted hyperintensities (unidentified bright objects) correlate with cognitive impairments in neurofibromatosis type 1 during childhood. Develop Neuropsychol 34(6):736–748
Gonen O, Wang ZJ, Viswanathan AK, Molloy PT, Zimmerman RA (1999) Three-dimensional multivoxel proton MR spectroscopy of the brain in children with neurofibromatosis type 1. AJNR Am J Neuroradiol 20:1333–1341
Wilkinson ID, Griffiths PD, Wales JK (2001) Proton magnetic resonance spectroscopy of brain lesions in children with neurofibromatosis type 1. Magn Reson Im 19:1081–1089
Alkan A, Sarac K, Kutlu R et al (2003) Proton MR spectroscopy features of normal appearing white matter in neurofibromatosis type 1. Magn Reson Im 21:1049–1053
Wang PY, Kaufmann WE, Koth CW, Denckla MB, Barker PB (2000) Thalamic involvement in neurofibromatosis type 1: evaluation with proton magnetic resonance spectroscopic imaging. Ann Neurol 47:477–484
Van Engelen SJ, Krab LC, Moll HA et al (2008) Quantitative differentiation between healthy and disordered brain matter in patients with neurofibromatosis type I using diffusion tensor imaging. AJNR Am J Neuroradiol 29:816–822
Eastwood JD, Fiorella DJ, MacFall JF, Delong DM, Provenzale JM, Greenwood RS (2001) Increased brain apparent diffusion coefficient in children with neurofibromatosis type 1. Radiology 219:354–358
Alkan A, Sigirci A, Kutlu R et al (2005) Neurofibromatosis type 1: diffusion weighted imaging findings of brain. Eur J Radiol 56:229–234
Tognini G, Ferrozzi F, Garlaschi G et al (2005) Brain apparent diffusion coefficient evaluation in pediatric patients with neurofibromatosis type 1. J Comput Assist Tomogr 29:298–304
Ferraz-Filho JRL, da Rocha AJ, Pontes Muniz M, Souza AS, Goloni-Bertollo EM, Pavarino-Bertelli EC (2012) Unidentified bright objects in neurofibromatosis type 1: conventional MRI in the follow-up and correlation of microstructural lesions on diffusion tensor images. Eur J Ped Neurol 16:42–47
Ferraz-Filho JRL, da Rocha AJ, Pontes Muniz M, Souza AS, Goloni-Bertollo EM, Pavarino-Bertelli EC (2012) Diffusion tensor MR imaging in neurofibromatosis type 1: expanding the knowledge of microstructural brain abnormalities. Pediatr Radiol 42:449–454
Mori H, Abe O, Okubo T et al (2001) Diffusion property in a hamartomatous lesion of neurofibromatosis type 1. J Comput Assist Tomogr 25:537–539
National Institutes of Health Consensus Development Conference (1988) Neurofibromatosis: conference statement. Arch Neurol 45:575–578
Sevick RJ, Barkovich AJ, Edwards MS, Koch T, Berg B, Lempert T (1992) Evolution of white matter lesions in neurofibromatosis type 1: MR findings. AJR Am J Roentgenol 159:171–175
Kraut MA, Gerring JP, Cooper KL, Thompson RE, Denckla MB, Kaufmann WE (2004) Longitudinal evolution of unidentified bright objects in children with neurofibromatosis-1. Am J Med Genet 129A:113–119
DiPaolo DP, Zimmerman RA, Rorke LB, Zackai EH, Bilaniuk LT, Yachnis AT (1995) Neurofibromatosis type 1: pathologic substrate of high signal intensity foci in the brain. Radiology 195:721–724
Moore BD, Slopis JM, SChomer D, Jackson EF, Levy BM (1996) Neuropsychological significance of areas of high signal intensity on brain MRIs of children with neurofibromatosis. Neurology 46:1660–1668
Goh WH, Khong PL, Leung CS, Wong VC (2004) T2-weighted hyperintensities (unidentified bright objects) in children with neurofibromatosis 1: their impact on cognitive function. J Child Neurol 19:853–858
Hyman SL, Gill DS, Shores EA, Steinberg A, North KN (2007) T2 hyperintensities in children with neurofibromatosis type 1 and their relationship with cognitive functioning. J Neurol Neurosurg Psychiatry 78:1088–1091
Denckla MB, Hofman K, Mazzocco MMM et al (1996) Relationship between T2-weighted hyperintensities (unidentified bright objects) and lower IQs in children with neurofibromatosis type 1. Am J Med Gen 67:98–102
Spalice A, Nicita F, Papetti L et al (2010) Usefulness of diffusion tensor imaging and fiber tractography in neurological and neurosurgical pediatric diseases. Childs Nerv Syst 26:995–1002
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nicita, F., Di Biasi, C., Sollaku, S. et al. Evaluation of the basal ganglia in neurofibromatosis type 1. Childs Nerv Syst 30, 319–325 (2014). https://doi.org/10.1007/s00381-013-2236-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-013-2236-z