Abstract
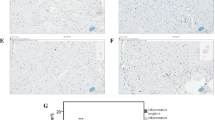
The ubiquitin (Ub)-proteasome system (UPS) is an important proteolytic mechanism for selecting and digesting cytotoxic proteins. The aim of this study is to elucidate expression and in situ localization of the UPS in the myocardium from patients with dilated cardiomyopathy (DCM) with refractory heart failure. The expression profile of the oxidative stress-induced cytotoxic proteins was also examined. Myocardium was obtained from 26 patients with DCM at the left ventriculoplasty. Ten normal autopsied hearts served as controls. Myocardial expressions of Ub and proteasomes were studied immunohistochemically. Oxidative stresses were examined in point of localization of the oxidation-induced modifier molecules (OMM). The relationship between immunohistochemical results and clinical parameters was also evaluated. Both Ub and proteasomes were stained positive in granular structures accumulating between the myofibrils and adjacent to nuclei in cardiomyocytes. The OMMs were also positive in the same Ub-positive granular structures. The area fraction of Ub, proteasomes and OMM was significantly higher in DCM hearts than in normal controls. Significant positive correlation was observed between the area fractions of Ub and plasma levels of brain natriuretic peptide (p = 0.046) in DCM hearts. In conclusion, enhanced expression of the UPS colocalized with OMM in cardiomyocytes may be involved in the pathophysiology of DCM hearts.








Similar content being viewed by others
References
Klausner RD, Sitia R (1990) Protein degradation in the endoplasmic reticulum. Cell 62:611–614
Hershko A, Ciechanover A, Varshavsky A (2000) Basic medical award. The ubiquitin system. Nat Med 6:1073–1081
Weissman AM (2001) Themes and variations on ubiquitylation. Nat Rev Mol Cell Biol 2:169–178
Ohsumi Y (2001) Molecular dissection of autophagy: two ubiquitin-like system. Nat Rev Mol Cell Biol 2:211–216
Wang CW, Klionsky DJ (2003) The molecular mechanism of autophagy. Mol Med 9:65–76
Stadtman ER (1992) Protein oxidation and aging. Science 257:1220–1224
Bucciantini M, Giannoni E, Chiti F, Broni F, Formiqli L, Zurdo J, Taddei N, Ramponi G, Dobson CM, Stefani M (2002) Inherent toxicity of aggregates implies a common mechanism for protein misfolding diseases. Nature 416:483–484
Belch JJ, Bridges AB, Scott N, Chopra M (1991) Oxygen free radicals and congestive heart failure. Br Heart J 65:245–248
Weekes J, Morrison K, Mullen A, Wait R, Barton P, Dunn MJ (2003) Hyperubiquitination of protein in dilated cardiomyopathy. Proteomics 3:208–216
Shimomura H, Terasaki F, Hayashi T, Kitaura Y, Isomura T, Suma T (2001) Autophagic degeneration as a possible mechanism of myocardial cell death in dilated cardiomyopathy. Jpn Circ J 65:965–968
Johnston JA, Ward CL, Kopito RR (1998) Aggresomes: a cellular response to misfolded proteins. J Cell Biol 143:1883–1898
Lowe J, Blanchard A, Morrell K, Lennox G, Revnolds L, Billett M, Landon M, Mayer RJ (1988) Ubiquitin is a common factor in intermediate filament inclusion bodies of diverse type in man, including those of Parkinson’s disease, Pick’s disease, and Alzheimer’s disease, as well as Rosenthal fibres in cerebellar astrocytomas, cytoplasmic bodies in muscle, and Mallory bodies in alcoholic liver disease. J Pathol 155:9–15
McNaught KS, Shashidharan P, Perl DP, Jenner P, Olanow CW (2002) Aggresome-related biogenesis of Lewy bodies. Eur J Neurosci 16:2136–2148
Kopito RR (2000) Aggresomes, inclusion bodies and protein aggregation. Trends Cell Biol 10:524–530
Tran PB, Miller RJ (1999) Aggregates in neurodegenerative disease: crows and power? Trends Neurosci 22:194–197
McMurray J, Chopra M, Abdullah I, Smith WE, Darqie HJ (1993) Evidence of oxidative stress in chronic heart failure in humans. Eur Heart J 14:1493–1498
Keith M, Geranmayegan A, Sole MJ, Kurian R, Robinson A, Omran AS, Jeejeebhoy KN (1998) Increased oxidative stress in patients with congestive heart failure. J Am Coll Cardiol 31:1352–1356
Mallat Z, Philip I, Lebret M, Chatel D, Maclouf J, Tedqui A (1998) Elevated levels of 8-iso-prostaglandin F2α in pericardial fluid of patients with heart failure: a potential role for in vivo oxidant stress in ventricular dilatation and progression to heart failure. Circulation 97:1536–1539
Okada K, Wangpoengtrakul C, Osawa T, Toyokuni S, Tanaka K, Uchida K (1999) 4-Hydroxy-2-nonenal-mediated impairment of intracellular proteolysis during oxidative stress. Identification of proteasomes as target molecules. J Biol Chem 274:23787–23793
Bulteau AL, Lundberg KC, Humphries KM, Sadek HA, Szweda PA, Friquet B, Szweda LI (2001) Oxidative modification and inactivation of the proteasome during coronary occlusion/reperfusion. J Biol Chem 276:30057–30063
McNaught KS, Mytilineou C, Jnobaptiste R, Yabut J, Shashidharan P, Jennert P, Olanow CW (2002) Impairment of the ubiquitin–proteasome system causes dopaminergic cell death and inclusion body formation in ventral mesencephalic cultures. J Neurochem 81:301–306
Majno G, Joris I (1995) Apoptosis, oncosis and necrosis. An overview of cell death. Am J Pathol 146:3–15
Narula J, Haider N, Virmani R, DiSalvo TG, Kolodqie FD, Hajjar RJ, Schmidt U, Semigran MJ, Dec GW, Khaw BA (1996) Apoptosis in myocytes in end-stage heart failure. N Engl J Med 335:1182–1189
Mallat Z, Tedgui A, Fontaliran F, Frank R, Duriqon M, Fontaine G (1996) Evidence of apoptosis in arrhythmogenic right ventricular dysplasia. N Engl J Med 335:1190–1196
McDonald ER 3rd, El-Deiry WS (2004) Suppression of caspase-8- and -10-associated RING proteins results in sensitization to death ligands and inhibition of tumor cell growth. Proc Natl Acad Sci USA 101:6170–6175
Powell SR, Wang P, Divald A, Teichberg S, Haridas V, McCloskey TW, Davies KJA, Katzeff H (2005) Aggregates of oxidized proteins (lipofuscin) induce apoptosis through proteasome inhibition and dysregulation of proapoptotic proteins. Free Radic Biol Med 38:1093–1101
Tannous P, Zhu H, Nemchenko A, Berry JM, Johnstone JL, Shelton JM, Miller FJ Jr, Rothermel BA, Hill JA (2008) Intracellular protein aggregation is a proximal trigger of cardiomyocyte autophagy. Circulation 117:3070–3078
Jung T, Bader N, Grune T (2007) Lipofuscin. Formation, distribution, and metabolic consequences. Ann N Y Acad Sci 1119:97–111
Hasegawa K, Fujiwara H, Doyama K, Mukoyama M, Nakao K, Fujiwara T, Imura H, Kawai C (1993) Ventricular expression of atrial and brain natriuretic peptides in dilated cardiomyopathy. An immunohistochemical study of the endomyocardial biopsy specimens using specific monoclonal antibodies. Am J Pathol 142:107–116
McDonagh TA, Robb SD, Murdoch DR, Morton JJ, Ford I, Morrison CE, Tunstall-Pedoe H, McMurray JJ, Dargie HJ (1998) Biochemical detection of left-ventricular systolic dysfunction. Lancet 351:9–13
Yamamoto K, Burnett JC Jr, Jougasaki M, Nishimura RA, Bailey KR, Saito Y, Nakao K, Redfield MM (1996) Superiority of brain natriuretic peptide as a hormonal maker of ventricular hypertrophy. Hypertension 28:988–994
Tsutamoto T, Wada A, Maeda K, Hisanaga T, Maeda Y, Fukai D, Ohnishi M, Sugimoto Y, Kinoshita M (1997) Attenuation of compensation of endogenous cardiac natriuretic peptide concentration in patients with chronic symptomatic left ventricular dysfunction. Circulation 96:509–516
Acknowledgments
This study was supported in part by grants from the Ministry of Health, Labor and Welfare of Japan. The authors wish to express deep gratitude to Dr. Takafumi Ogawa and Mr. Masahiro Jo for their valuable technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Otsuka, K., Terasaki, F., Shimomura, H. et al. Enhanced expression of the ubiquitin-proteasome system in the myocardium from patients with dilated cardiomyopathy referred for left ventriculoplasty: an immunohistochemical study with special reference to oxidative stress. Heart Vessels 25, 474–484 (2010). https://doi.org/10.1007/s00380-010-0006-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-010-0006-3