Abstract
Objectives
To investigate the effect of klotho gene and β-glucuronidase activity on stone formation in patients with urinary tract stone disease (UTSD).
Methods
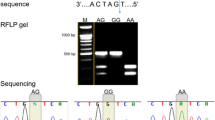
A total of 103 patients with UTSD and 102 controls with no specific urolithiasis history were enrolled into the study. G395A and C1818T polymorphisms of klotho gene were analyzed with PCR method. Serum levels of calcium and phosphorus and 24-h urine levels of β-glucuronidase activity, calcium and phosphorus levels were measured biochemically.
Results
A total of 103 of patients were male (50.2 %) and 102 were female (49.8 %) (p 0.945). Twenty-four-hour urine levels of calcium were significantly higher in UTSD group, whereas no difference was observed in phosphorus levels (p < 0.001, p 0.074, respectively). As for the G395A polymorphism, type of GG was significantly higher in the patient group compared to the controls (p = 0.02), while GA genotype was significantly higher in the controls (p = 0.001). There was no significant difference in F352V and C1818T polymorphism between the patient and control groups. β-glucuronidase activity was slightly lower in the patient group without significance (p 0.932).When patients with GG genotype and the rest were compared, there were no significant difference in all parameters.
Conclusions
Any polymorphism altering the function of klotho gene may result with stone formation. We found that there are more GG sequences of G395A gene in patients with UTSD. That may be a polymorphism of klotho gene which results with stone formation. Further studies with more patients should be accomplished which are combining the genetic and epigenetic factors associated with urolithiasis and klotho gene to enlighten the etiology of this disease.
Similar content being viewed by others
References
Sarıca K (2007) Üriner sistem taş hastalığında epidemiyoloji. Üriner Sistem Taş Hastalığı. istanbul: nobel tıp kitapevi, 9–18
Yoshida O, Okada Y (1990) Epidemiology of urolithiasis in Japan: a chronological and geographical study. Urol Int 45(2):104–111
Akinci M, Esen T, Tellaloğlu S (1991) Urinary stone disease in Turkey: an updated epidemiological study. Eur Urol 20(3):200–203
Curhan GC, Rimm EB, Willett WC, Stampfer MJ (1994) Regional variation in nephrolithiasis incidence and prevalence among United States men. J Urol 151(4):838–841
Borghi L, Ferretti PP, Elia GF, Amato F, Melloni E, Trapassi MR et al (1990) Epidemiological study of urinary tract stones in a northern Italian city. Br J Urol 65(3):231–235
Thun MJ, Schober S (1991) Urolithiasis in Tennessee: an occupational window into a regional problem. Am J Public Health 81(5):587–591
Stamatelou KK, Francis ME, Jones CA, Nyberg LM, Curhan GC (2003) Time trends in reported prevalence of kidney stones in the United States: 1976–1994. Kidney Int 63(5):1817–1823
Nabeshima Y (2008) Discovery of alpha-Klotho and FGF23 unveiled new insight into calcium and phosphate homeostasis. Clin Calcium 18(7):923–934
Civitelli R, Ziambaras K (2011) Calcium and phosphate homeostasis: concerted interplay of new regulators. J Endocrinol Invest 34(7 Suppl):3–7
Chang Q, Hoefs S, van der Kemp AW, Topala CN, Bindels RJ, Hoenderop JG (2005) The beta-glucuronidase klotho hydrolyzes and activates the TRPV5 channel. Science 310(5747):490–493
Kurosu H, Ogawa Y, Miyoshi M, Yamamoto M, Nandi A, Rosenblatt KP et al (2006) Regulation of fibroblast growth factor-23 signaling by klotho. J Biol Chem 281(10):6120–6123
Hu MC, Shi M, Zhang J, Pastor J, Nakatani T, Lanske B et al (2010) Klotho: a novel phosphaturic substance acting as an autocrine enzyme in the renal proximal tubule. FASEB J Off Publ Fed Am Soc Exp Biol 24(9):3438–3450
Razzaque MS, Lanske B (2007) The emerging role of the fibroblast growth factor-23-klotho axis in renal regulation of phosphate homeostasis. J Endocrinol 194(1):1–10
Kuro-o M (2009) Klotho and aging. Biochim Biophys Acta 1790(10):1049–1058
Davalos M, Konno S, Eshghi M, Choudhury M (2010) Oxidative renal cell injury induced by calcium oxalate crystal and renoprotection with antioxidants: a possible role of oxidative stress in nephrolithiasis. J Endourol Endourol Soc 24(3):339–345
Tungsanga K, Sriboonlue P, Futrakul P, Yachantha C, Tosukhowong P (2005) Renal tubular cell damage and oxidative stress in renal stone patients and the effect of potassium citrate treatment. Urol Res 33(1):65–69
Huang H-S, Ma M-C, Chen C-F, Chen J (2003) Lipid peroxidation and its correlations with urinary levels of oxalate, citric acid, and osteopontin in patients with renal calcium oxalate stones. Urology 62(6):1123–1128
Shimoyama Y, Nishio K, Hamajima N, Niwa T (2009) KLOTHO gene polymorphisms G-395A and C1818T are associated with lipid and glucose metabolism, bone mineral density and systolic blood pressure in Japanese healthy subjects. Clin Chim Acta Int J Clin Chem 406(1–2):134–138
Low AF, O’Donnell CJ, Kathiresan S, Everett B, Chae CU, Shaw SY et al (2005) Aging syndrome genes and premature coronary artery disease. BMC Med Genet 31(6):38
Cobben NA, Drent M, De Vries J, Wouters EF, Van Dieijen-Visser MP, Henderson RF (1999) Serum beta-glucuronidase activity in a population of ex-coalminers. Clin Biochem 32(8):659–664
Kuro-o M, Matsumura Y, Aizawa H, Kawaguchi H, Suga T, Utsugi T et al (1997) Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature 390(6655):45–51
Imura A, Iwano A, Tohyama O, Tsuji Y, Nozaki K, Hashimoto N et al (2004) Secreted Klotho protein in sera and CSF: implication for post-translational cleavage in release of Klotho protein from cell membrane. FEBS Lett 565(1–3):143–147
Imura A, Tsuji Y, Murata M, Maeda R, Kubota K, Iwano A et al (2007) alpha-Klotho as a regulator of calcium homeostasis. Science 316(5831):1615–1618
Xu C, Song R, Yang J, Jiang B, Wang X, Wu W et al (2013) Klotho gene polymorphism of rs3752472 is associated with the risk of urinary calculi in the population of Han nationality in Eastern China. Gene 526(2):494–497
Telci D, Dogan AU, Ozbek E, Polat EC, Simsek A, Cakir SS et al (2011) KLOTHO gene polymorphism of G395A is associated with kidney stones. Am J Nephrol 33(4):337–343
Li S-A, Watanabe M, Yamada H, Nagai A, Kinuta M, Takei K (2004) Immunohistochemical localization of Klotho protein in brain, kidney, and reproductive organs of mice. Cell Struct Funct 29(4):91–99
Martin A, David V, Quarles LD (2012) Regulation and function of the FGF23/klotho endocrine pathways. Physiol Rev 92(1):131–155
Hu MC, Kuro-o M, Moe OW (2013) Klotho and chronic kidney disease. Contrib Nephrol 180:47–63
Author’s contribution
Gürel contributed to data collection or management, data analysis and manuscript writing; Üre contributed to data analysis and manuscript writing; Temel and Aslan developed the protocol/project; Çilingir developed the protocol/project and analyzed data; Uslu and Celayir contributed to data collection or management; and Başeskioğlu developed the protocol/project and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gürel, A., Üre, İ., Temel, H.E. et al. The impact of klotho gene polymorphisms on urinary tract stone disease. World J Urol 34, 1045–1050 (2016). https://doi.org/10.1007/s00345-015-1732-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-015-1732-z