Abstract
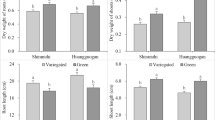
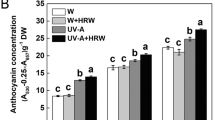
Chinese fir (Cunninghamia lanceolata) cotyledons are green in darkness due to the coexistence of functional dark-operative protochlorophyllide reductase (DPOR) and light-dependent protochlorophyllide reductase (LPOR). In this study, we found that the POR gene for LPOR and the ChlB, ChlL, and ChlN genes for three DPOR subunits were strongly downregulated in dark-grown new sprouts in which chlorophyll biosynthesis was inhibited. We extracted, sequenced, and annotated the total RNA from etiolated (dark-grown) and green (light-grown) sprouts. A total of 70,499 unigenes were found, of which 47,410 were annotated. Among them, 3263 differentially expressed genes were identified between dark- and light-grown samples. According to the Kyoto Encyclopedia of Genes and Genomes databases, the five most affected metabolic pathways were flavonoid biosynthesis, photosynthesis-antenna proteins, photosynthesis, cutin, suberine and wax biosynthesis and stilbenoid, diarylheptanoid, and gingerol biosynthesis. Based on the acquired cDNA sequences, we applied quantitative PCR to further analyze the relative expression levels of 18 nuclear genes and 11 chloroplast genes related to photophosphorylation. Both transcriptomic and quantitative PCR results indicated that dark treatment can significantly block the expression of all of the identified genes related to the protein complexes of photosynthetic electron flow in new sprouts. Dark treatment of new sprouts of Chinese fir caused etiolation via blockage of the expression of DPOR- and LPOR-encoding genes. Both nuclear and plastid genomic genes related to photosynthesis were significantly downregulated in etiolated sprouts. The results provide new clues to understanding the different mechanisms of chloroplast development in different photosynthetic organs.





Similar content being viewed by others
References
Armstrong GA (1998) Greening in the dark: light-independent chlorophyll biosynthesis from anoxygenic photosynthetic bacteria to gymnosperms. J Photoch Photobio B 43(2):87–100
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24(1):1–14
Brzezowski P, Richter AS, Grimm B (2015) Regulation and function of tetrapyrrole biosynthesis in plants and algae. Biochim Biophys Acta Bioenerg 1847(9):968–985
Bujaldon S, Kodama N, Rappaport F, Subramanyam R, deVitry C, Takahashi Y, Wollman FA (2017) Functional accumulation of antenna proteins in chlorophyll b-less mutants of Chlamydomonas reinhardtii. Mol Plant 10(1):115–130
Chi W, Sun X, Zhang L (2013) Intracellular signaling from plastid to nucleus. Annu Rev Plant Biol 64(1):559–582
Demko V, Pavlovic A, Valkova D, Grimm B, Hudak J (2009) A novel insight into the regulation of light-independent chlorophyll biosynthesis in Larix decidua and Picea abies seedlings. Planta 230(1):165–176
Fujita Y, Bauer CE (2000) Reconstitution of light-independent protochlorophyllide reductase from purified BchL and BchN-BchB subunits. In vitro confirmation of nitrogenase-like features of a bacteriochlorophyll biosynthesis enzyme. J Biol Chem 275(31):23583–23588
Goslings D, Meskauskiene R, Kim C, Lee KP, Nater M, Apel K (2004) Concurrent interactions of heme and FLU with Glu-tRNA reductase (HEMA1), the target of metabolic feedback inhibition of tetrapyrrole biosynthesis, in dark- and light-grown Arabidopsis plants. Plant J 40(6):957–967
Josse E, Halliday K (2008) Skotomorphogenesis: the dark side of light signalling. Curr Biol 18(24):R1144–R1146
Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M et al. (2008) KEGG for linking genomes to life and the environment. Nucleic Acids Res 36:D480–D484
Kreft S, Knapp M, Kreft I (1999) Extraction of rutin from buckwheat (Fagopyrum esculentum Moench) seeds and determination by capillary electrophoresis. J Anim Sci 47(11):4649–4652
Larmor J, Stokes SGG (1907) Memoir and scientific correspondence of the late Sir George Gabriel Stokes. Cambridge University Press, Cambridge
Margna U, Margna E, Paluteder A (1990) Localization and distribution of favonoids in buckwheat seedling cotyledons. J Plant Physiol 136(2):166–171
Mohanty S, Baishna BG, Tripathy C (2006) Light and dark modulation of chlorophyll biosynthetic genes in response to temperature. Planta 224(3):692–699
Morren E (1858) Dissertation sur les feuilles vertes et coloré es envisagénes spécialement au point de vue des rapports de la chlorophylle et de l’ Erythrophylle. Annoot-Braeckman, Gand
Neff MM, Fankhauser C, Chory J (2000) Light: an indicator of time and place. Gene Dev 14(3):257–271
Pavlovič A, Stolárik T, Nosek L, Kouřil R, Ilík P (2016) Light-induced gradual activation of photosystem II in dark-grown Norway spruce seedlings. BBA-Bioenerg 1857(6):799–809
Ravanfar SA, Karimi E, Mehrabanjoubani P, Ebrahimi M (2018) Enhancement of phenolic and flavonoids compounds, antioxidant and cytotoxic effects in regenerated red cabbage by application of Zeatin. Nat Prod Res 2018:1–5
Reinbothe C, Bakkouri ME, Buhr F, Muraki N, Nomata J, Kurisu G, Fujita Y, Reinbothe S (2010) Chlorophyll biosynthesis: spotlight on protochlorophyllide reduction. Trends Plant Sci 15(11):614–624
Shi H, Lyu M, Luo Y, Liu S, Li Y, He H, Wei N, Deng XW, Zhong S (2018) Genome-wide regulation of light-controlled seedling morphogenesis by three families of transcription factors. Proc Natl Acad Sci USA 115(25):6482–6487
Shinohara K, Murakami A, Fujita Y (1992) Biochemical characteristics of thylakoid membranes in chloroplasts of dark-grown pine cotyledons. Plant Physiol 98(1):39–43
Shinozaki K, Ohme M, Tanaka M, Wakasugi T, Hayashida N, Matsubayashi T, Zaita N, Chunwongse J, Obokata J, Yamaguchi-Shinozaki K (1986) The complete nucleotide sequence of the tobacco chloroplast genome: its gene organization and expression. EMBO J 5(9):2043
Skinner JS, Timko MP (1999) Differential expression of genes encoding the light-dependent and light-independent enzymes for protochlorophyllide reduction during development in loblolly pine. Plant Mol Biol 39(3):577–592
Solymosi K, Schoefs B (2010) Etioplast and etio-chloroplast formation under natural conditions: the dark side of chlorophyll biosynthesis in angiosperms. Photosynth Res 105(2):143–166
Stolárik T, Nožková V, Nosek L, Pavlovič A (2017a) Dark chlorophyll synthesis may provide a potential for shade tolerance as shown by a comparative study with seedlings of European larch (Larix decidua) and Norway spruce (Picea abies). Trees 32(4):951–965
Stolárik T, Hedtke B, Šantrůček J, Ilík P, Grimm B, Pavlovič A (2017b) Transcriptional and post-translational control of chlorophyll biosynthesis by dark-operative protochlorophyllide oxidoreductase in Norway spruce. Photosynth Res 132(2):165–179
Von Arnim A, Deng XW (1996) Light control of seedling development. Annu Rev Plant Physiol Plant Mol Biol 47(1):215–243
Von Wettstein D, Gough S, Kannangara CG (1995) Chlorophyll biosynthesis. Plant Cell 7(7):1039–1057
Vothknecht UC, Westhoff P (2001) Biogenesis and origin of thylakoid membranes. BBA-Mol Cell Res 1541(1):91–101
Walles B, Hudák J (1975) A comparative study of chloroplast morphogenesis in seedlings of some conifers (Larix decidua, Pinus sylvestris and Picea abies). Stud For Suec 127:5–22
Winkel-Shirley B (2001) Flavonoid biosynthesis: a colorful model for genetics, biochemistry, cell biology and biotechnology. Plant Physiol 126(2):485–493
Woodward RB (1961) The total synthesis of chlorophyll. Pure Appl Chem 2(3–4):383–404
Wu L, Zhou ZY, Zhang CG, Chai J, Zhou Q, Wang L, Hirnerova E, Mrvkova M, Novak O, Guo GQ (2015) Functional roles of three cutin biosynthetic acyltransferases in cytokinin responses and skotomorphogenesis. PLoS ONE 10(3):e0121943
Xue X, Wang Q, Qu Y, Wu H, Dong F, Cao H, Wang H, Xiao J, Shen Y, Wan Y (2017) Development of the photosynthetic apparatus of Cunninghamia lanceolata in light and darkness. New Phytol 213(1):300–313
Yamamoto H, Kusumi J, Yamakawa H, Fujita Y (2017) The effect of two amino acid residue substitutions via RNA editing on darkoperative protochlorophyllide oxidoreductase in the black pine chloroplasts. Sci Rep 7(1):2377
Acknowledgements
We would like to thank Dr Jinling Feng (Fujian Agriculture and Forestry University) who collected and provided the Chinese fir trees. We thank LetPub (www.letpub.com) for its linguistic assistance during the preparation of this manuscript. This work is supported by the National Natural Science Foundation of China (31671489, 31271433, 31871424) and the Fundamental Research Funds for the Central Universities (2018ZY37).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All the authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Q., Liu, S., Li, X. et al. Expression of Genes in New Sprouts of Cunninghamia lanceolata Grown Under Dark and Light Conditions. J Plant Growth Regul 39, 481–491 (2020). https://doi.org/10.1007/s00344-019-09996-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-019-09996-9