Abstract
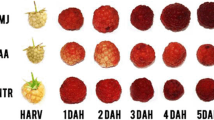
Jasmonates (JAs) play a role in the responses to environmental stress and during growth processes, including fruit/seed development. To better understand the molecular basis of the developmental control exerted by JAs in fruit and seed, methyl jasmonate (MJ, 0.80 mM) was applied to peach fruit (Prunus persica var. laevis Gray) at an early (S1) developmental stage and under field conditions. Mesocarp and seed were sampled at time intervals until ripening; at harvest, MJ-treated fruit were less ripe than controls as assessed by a nondestructive device called a DA-meter. Real-time reverse-transcription polymerase chain reaction analyses revealed that JA-related gene expression (AOS1 and JAZs) was affected early (24 h) after treatment, whereas peaks in transcript accumulation of mesocarp (CYCD3, RD22, SP, Aux/IAA) and seed (PRP, SSADH, PRU, LEA) developmental marker genes were shifted in accord with a developmental slowing down. At ripening (S4), in the mesocarp the upregulation of the ethylene biosynthetic genes ACO1 and ACS1 and of the softening-related genes PG and EXP2 was dramatically counteracted by MJ. Ethylene signaling (ETR1, ETR2) was also affected. Because JAs cross-talk with other hormones, the transcript amounts of major hormone-related genes such as GH3, IAA-AH, NCED, and GA2ox were evaluated and showed changes that further support the hypothesis of delay of the developmental program.






Similar content being viewed by others
References
Bartel B, Fink GR (1995) ILR1, an amidohydrolase that releases active indole-3-acetic acid from conjugates. Science 268:1745–1748
Bonghi C, Trainotti L, Botton A, Tadiello A, Rasori, Ziliotto F, Zaffalon V, Casadoro G, Ramina A (2011) A microarray approach to identify genes involved in seed-pericarp cross-talk and development in peach. BMC Plant Biol 11:107
Böttcher C, Keyzers RA, Boss PK, Davies C (2010) Sequestration of auxin by the indole-3-acetic acid-amido synthetase GH3-1 in grape berry (Vitis vinifera L.) and the proposed role of auxin conjugation during ripening. J Exp Bot 61:3615–3625
Chang S, Puryear J, Cairney J (1993) A simple and efficient method for isolating RNA from pine trees. Plant Mol Biol Report 11:113–116
Cipollini D (2010) Constitutive expression of methyl jasmonate-inducible responses delays reproduction and constrains fitness responses to nutrients in Arabidopsis thaliana. Evol Ecol 24:59–68
Costa G, Noferini M, Fiore G (2005) Metodo ed apparato per determinare la qualità di prodotti ortofrutticoli. Italy Patent No. MO2005000211, University of Bologna
Dardick C, Callahan AM, Chiozzotto R, Schaffer RJ, Piagnani MC, Scorza R (2010) Stone formation in peach fruit exhibits spatial coordination of the lignin and flavonoid pathways and similarity to Arabidopsis dehiscence. BMC Biol 8:13
Devoghalaere F, Doucen T, Guitton B, Keeling J, Payne W, Ling TJ, Ross JJ, Hallett IC, Gunaseelan K, Dayatilake GA, Diak R, Breen KC, Tustin DS, Costes E, Chagné D, Schaffer RJ, David KM (2012) A genomics approach to understanding the role of auxin in apple (Malus x domestica) fruit size control. BMC Plant Biol 12:7
Fan X, Mattheis JP, Fellman JK (1998) A role for jasmonate in climacteric fruit ripening. Planta 204:444–449
Feys BJF, Benedetti CE, Penfold CN, Turner JG (1994) Arabidopsis mutants selected for resistance to the phytotoxin coronatine are male-sterile, insensitive to methyl jasmonate, and resistant to a bacterial pathogen. Plant Cell 6:751–759
Finkelstein RR (2004) The role of hormones during seed development and germination. In: Davies PJ (ed) Plant hormones: biosynthesis, signal transduction, action!. Kluwer Academic, Dordrecht, pp 513–537
Ghiani A, Onelli E, Aina R, Cocucci M, Citterio S (2011) A comparative study of melting and non-melting flesh peach cultivars reveals that during fruit ripening endopolygalacturonase (endo-PG) is mainly involved in pericarp textural changes, not in firmness reduction. J Exp Bot 62:4043–4054
Goetz S, Hellwege A, Stenzel I, Kutter C, Hauptmann V, Forner S, McCaig B, Hause G, Miersch O, Wasternack C, Hause B (2012) Role of cis-12-oxo-phytodienoic acid in tomato embryo development. Plant Physiol 158:1715–1727
Goossens A, Hakkinen ST, Laakso I, Seppanen-Laakso T, Biondi S, De Sutter V, Lammertyn F, Nuutila AM, Soderlund H, Zabeau M, Inze D, Oksman-Caldentey K-M (2003) A functional genomics approach toward the understanding of secondary metabolism in plant cells. Proc Natl Acad Sci USA 100:8595–8600
Han YY, Li AX, Li F, Zhao MR, Wang W (2012) Characterization of a wheat (Triticum aestivum L.) expansin gene, TaEXPB23, involved in the abiotic stress response and phytohormone regulation. Plant Physiol Biochem 54:49–58
Hayama H, Ito A, Moriguchi T, Kashimura Y (2003) Identification of a new expansin gene closely associated with peach fruit softening. Postharvest Biol Technol 2:1–10
Heil M, Ton J (2008) Long-distance signalling in plant defence. Trends Plant Sci 13:264–272
Howe GA, Lee GI, Itoh A, Li L, De Rocher AE (2000) Cytochrome P450-dependent metabolism of oxylipins in tomato. Cloning and expression of allene oxide synthase and fatty acid hydroperoxide lyase. Plant Physiol 123:711–724
Ismail A, Riemann M, Nick P (2012) The jasmonate pathway mediates salt tolerance in grapevines. J Exp Bot 63:2127–2139
Kanno Y, Jikumaru Y, Hanada A, Nambara E, Abrams SR, Kamiya Y, Seo M (2010) Comprehensive hormone profiling in developing Arabidopsis seeds: examination of the site of ABA biosynthesis, ABA transport and hormone interactions. Plant Cell Physiol 51:1988–2001
Kaush KD, Sobolev AP, Goyal RK, Fatima T, Laila-Beevi R, Saftner RA, Handa AK, Mattoo AK (2012) Methyl jasmonate deficiency alters cellular metabolome, including the aminome of tomato (Solanum lycopersicum L.) fruit. Amino Acids 42:843–856
Kazan K, Manners JM (2011) JAZ repressors and the orchestration of phytohormone crosstalk. Trends Plant Sci 17:22–31
Kondo S, Tomyiama A, Seto H (2000) Changes of endogenous jasmonic acid and methyl jasmonate in apples and sweet cherries during fruit development. J Am Soc Hortic Sci 125:282–287
Kondo S, Yamada H, Setha S (2007) Effects of jasmonates differed at fruit ripening stages on 1-aminocyclopropane-1-carboxylate (ACC) synthase and ACC oxidase gene expression in pears. J Am Soc Hortic Sci 132:120–125
Kubigsteltig I, Laudert D, Weiler EW (1999) Structure and regulation of the Arabidopsis thaliana allene oxide synthase gene. Planta 208:463–471
Kumar R, Agarwal P, Tyagi AK, Sharma AK (2012) Genome-wide investigation and expression analysis suggest diverse roles of auxin-responsive GH3 genes during development and response to different stimuli in tomato (Solanum lycopersicum). Mol Genet Genomics 287:221–235
Linkies A, Leubner-Metzger G (2012) Beyond gibberellins and abscisic acid: how ethylene and jasmonates control seed germination. Plant Cell Rep 31:253–270
Lorenzo O, Piqueras R, Sánchez-Serrano JJ, Solano R (2003) ETHYLENE RESPONSE FACTOR1 integrates signals from ethylene and jasmonate pathways in plant defense. Plant Cell 15:165–178
McConn M, Browse J (1996) The critical requirement for linolenic acid is pollen development, not photosynthesis, in an Arabidopsis mutant. Plant Cell 8:403–416
Memelink J (2009) Regulation of gene expression by jasmonate hormones. Phytochemistry 70:1560–1570
Muller PY, Janovjak H, Miserez AR, Dobbie Z (2002) Processing of gene expression data generated by quantitative real-time RT-PCR. Biotechniques 32:1372–1379
Nilo RR, Campos-Vargas R, Orellana A (2012) Assessment of Prunus persica fruit softening using a proteomic approach. J Proteomics 75:1618–1638
Ognjanov V, Vujanic-Varga D, Misic PD, Veresbaranji I, Macet K, Tesovic Z, Krstic M, Petrovic N (1995) Anatomical and biochemical studies of fruit development in peach. Sci Hortic 64:33–48
Oh Y, Baldwin IT, Gális I (2012) NaJAZh regulates a subset of defense responses against herbivores and spontaneous leaf necrosis in Nicotiana attenuata plants. Plant Physiol 159:769–788
Overmyer K, Brosché M, Kangasjärvi J (2003) Reactive oxygen species and hormonal control of cell death. Trends Plant Sci 8:335–342
Pauwels L, Inzé D, Goossens A (2009) Jasmonate-inducible gene: what does it mean? Trends Plant Sci 14:87–91
Payasi A, Sanwal GG (2010) Ripening of climacteric fruits and their control. J Food Biochem 34:679–710
Rohwer CL, Erwin GE (2008) Horticultural application of jasmonates. J Hortic Sci Biotechnol 83:283–304
Soto A, Ruiz KB, Ziosi V, Costa G, Torrigiani P (2012) Ethylene and auxin metabolism and signaling are impaired by methyl jasmonate leading to a transient slowing down of ripening in peach fruit. J Plant Physiol 169:1858–1865
Srivastava A, Handa AK (2005) Hormonal regulation of tomato fruit development: a molecular perspective. J Plant Growth Regul 24:67–82
Staswick P, Serban B, Rowe M, Tiryaki I, Maldonado MT, Maldonado MC, Suza W (2005) Characterization of an Arabidopsis enzyme family that conjugates amino acids to indole-3-acetic acid. Plant Cell 17:616–627
Thines B, Katsir L, Melotto M, Niu Y, Mandaokar A, Liu G, Nomura K, He SH, Howe GA, Browse J (2007) JAZ repressor proteins are targets of the SCFCOI1 complex during jasmonate signalling. Nature 448:661–665
Tonutti P, Bonghi C, Ruperti B, Tornielli GB, Ramina A (1997) Ethylene evolution and 1-aminocyclopropane-1-carboxylate oxidase gene expression during early development and ripening of peach fruit. J Am Soc Hortic Sci 122:642–647
Torrigiani P, Fregola F, Ziosi V, Ruiz KB, Kondo S, Costa G (2012a) Differential expression of allene oxide synthase (AOS), and jasmonate relationship with ethylene biosynthesis in seed and mesocarp of developing peach fruit. Postharvest Biol Technol 63:67–73
Torrigiani P, Bressanin D, Ruiz Carrasco K, Tadiello A, Trainotti L, Bonghi C, Ziosi V, Costa G (2012b) Spermidine application to young developing peach fruits leads to a slowing down of ripening by impairing ripening-related ethylene and auxin metabolism and signaling. Physiol Plant 146:86–98
Trainotti L, Zanin D, Casadoro G (2003) A cell wall-oriented genomic approach reveals a new and unexpected complexity of the softening in peaches. J Exp Bot 54:1821–1832
Trainotti L, Bonghi C, Ziliotto F, Zanin D, Rasori A, Casadoro G, Ramina R, Tonutti P (2006) The use of microarray μPeach1.0 to investigate transcriptome changes during transition from pre-climacteric to climacteric phase in peach fruit. Plant Sci 170:606–614
Trainotti L, Tadiello A, Casadoro G (2007) The involvement of auxin in the ripening of climacteric fruits comes of age: the hormone plays a role of its own and has an intense interplay with ethylene in ripening peaches. J Exp Bot 58:3299–3308
Wasternack C (2007) Jasmonates, an update on biosynthesis, signal transduction and action in plant stress response, growth and development. Ann Bot 100:681–697
Wasternack C, Forner S, Strnad M, Hause B (2013) Jasmonates in flower and seed development. Biochimie 95:79–85
Yang DL, Yao J, Mei CS, Tong XH, Zeng LH, Li Q, Xiao LT, Sun TP, Li J, Deng XW, Lee CM, Thomashow MF, Yang YN, He ZH, Yang, He SY (2012) Plant hormone jasmonate prioritizes defense over growth by interfering with gibberellin signaling cascade. Proc Natl Acad Sci USA 109:E1192–E1200
Yu M, Shen L, Fan B, Zhao D, Zhen Y, Sheng J (2009) The effect of MeJA on ethylene biosynthesis and induced disease resistance to Botrytis cinerea in tomato. Postharvest Biol Technol 54:153–158
Zanchin A, Bonghi C, Casadoro G, Ramina A, Rascio N (1994) Cell enlargement and cell separation during peach fruit development. Int J Plant Sci 155:49–56
Zhang M, Ping L, Zhang G, Li X (2009) Cloning and functional analysis of 9-cis-epoxycarotenoid dioxygenase (NCED) genes encoding a key enzyme during abscisic acid biosynthesis from peach and grape fruits. J Plant Physiol 166:1241–1252
Zhu Z, An F, Feng Y, Li P, Xue L, M A, Jiang Z, Kim JM, To TK, Li W, Zhang X, Yu Q, Dong Z, Chen WQ, Seki M, Zhou JM, Guo H (2011) Derepression of ethylene-stabilized transcription factors (EIN3/EIL1) mediates jasmonate and ethylene signaling synergy in Arabidopsis. Proc Natl Acad Sci USA 108:12539–12544
Ziegler J, Stenzel I, Hause B, Maucher H, Hamberg M, Grimm R, Ganal M, Wasternack C (2010) Molecular cloning of allene oxide cyclase: the enzyme establishing the stereochemistry of octadecanoids and jasmonates. J Biol Chem 594:19132–19138
Ziliotto F, Begheldo M, Rasori A, Bonghi C, Tonutti P (2008) Transcriptome profiling of ripening nectarine (Prunus persica L. Batsch) fruit treated with 1-MCP. J Exp Bot 59:2781–2791
Ziosi V, Bonghi C, Bregoli AM, Trainotti L, Biondi S, Setha S, Kondo S, Costa G, Torrigiani P (2008a) Jasmonate-induced transcriptional changes suggest a negative interference with the ripening syndrome in peach fruit. J Exp Bot 59:563–573
Ziosi V, Noferini M, Fiori G, Tadiello A, Trainotti L, Casadoro G, Costa G (2008b) A new index based on Vis spectroscopy to characterize the progression of ripening in peach fruit. Postharvest Biol Technol 49:319–329
Ziosi V, Bregoli AM, Fregola F, Costa G, Torrigiani P (2009) Jasmonate-induced ripening delay is associated with up-regulation of polyamine levels in peach fruit. J Plant Physiol 166:938–946
Acknowledgments
This research was supported by funds PRIN 2007 (project 20074AX5CA_003: Seed and fruit development in peach, phenol and jasmonate metabolism, expressed genes and related markers) from the Italian MIUR to PT. The authors thank Daniela Bressanin for collaboration and Stefania Biondi for critical reading of the manuscript and helpful criticism.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ruiz, K.B., Trainotti, L., Bonghi, C. et al. Early Methyl Jasmonate Application to Peach Delays Fruit/Seed Development by Altering the Expression of Multiple Hormone-Related Genes. J Plant Growth Regul 32, 852–864 (2013). https://doi.org/10.1007/s00344-013-9351-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-013-9351-7