Abstract
The nanocomposite of titanium dioxide (TiO2) combined with graphene oxide (GO) and cerium (Ce) was successfully synthesized via sol–gel method followed by calcining at 300 °C for 2 h. The composite was characterized by X-ray diffraction, ultraviolet–visible diffuse reflectance spectroscopy, scanning electron microscopy (SEM) and Brunauer–Emmett–Teller. The photocatalytic activity was evaluated by photodegradation of methylene blue (MB) under the irradiation of xenon lamp. This study demonstrated that GO and Ce co-doped in TiO2 could broaden absorption edge to the visible light and increase surface area of samples. SEM observation showed that addition of Ce could solve the problem of the agglomeration of GO under the same experimental conditions. Moreover, the MB photocatalytic degradation rate of the composite with GO doped for 0.2 % and Ce doped for 0.6 % (mass ratio) was up to 97.7 %, which was largely attributed to the synergistic effects in the GO, Ce and TiO2 system.
Similar content being viewed by others
1 Introduction
The condition of contamination is deteriorating day by day. It is becoming a burning issue. Therefore, the development of an effective and stable method to decrease the organic pollutants is an urgent need to address the issue. As far as we know, photocatalytic oxidation technology is an effective method for the degradation of pollutants. Among many photocatalytic oxidants, titanium dioxide (TiO2) is well known for its good performance: excellent physical and chemical stability, non-toxicity and inexpensiveness [1]. However, the photocatalytic decomposition of the pollutions by bare TiO2 is limited. TiO2-based materials have been the most promising candidates for photocatalytic decontamination [2]. TiO2 in pure form and doped with metallic ions or carbonaceous material is widely reported and applied in photocatalysis [3–5].
Graphene oxide (GO) is a kind of excellent performance of new carbon materials, consisting of sheet-like graphene framework with reactive oxygen functional groups and defects, which can provide a sheet as a good candidate for supporting metal or metal oxide particles [6, 7]. Up to now, many efforts have been made to prepare GO–TiO2 composite utilizing the unique properties of GO in order to increase the efficiency of photocatalysis [8–10]. Liu et al. [9] developed a water/toluene two-phase method for self-assembling TiO2 nanorods on the whole GO layer and revealed the effective absorption of MB by large GO sheets and the effective charge transfer from TiO2 nanorods to large GO sheets. Jiang et al. [10] synthesized graphene oxide/TiO2 by in situ depositing TiO2 on the graphene oxide and found that the excellent enhancing effect of graphene oxide sheets on the photocatalytic properties of TiO2 was attributed to a large surface area and much increased adsorption capacity. However, the investigations were largely performed under UV light, and the MB photocatalytic degradation of GO–TiO2 under visible light was not well. Therefore, it will be meaningful to research how to make GO–TiO2 have good catalytic ability under visible light. It is reported that Ce ion doping could broaden the absorption spectra of TiO2 to the visible light region [11, 12], because Ce can form complexes with various Lewis bases including organic acids, aldehydes, alcohols and thiols in the interaction of the functional groups with their f orbital [13]. As an inexpensive and relatively non-toxic RE ions, cerium has been utilized to degrade organic pollutants, especially in combination with TiO2 [14]. The key feature of Ce ion doping is the disparate electronic structures of Ce3+ with 4f 15d 0 and Ce4+ with 4f 05d 0, which can lead to diverse optical properties and different catalytic properties [15, 16]. Hence, Ce ion doping could improve TiO2 photocatalytic activity significantly [17, 18].
From the above, GO doping can increase the specific surface area of TiO2 and does not extend the absorption band edge of TiO2 to the visible light region. And Ce doping is just the opposite. In addition, the active oxygen functional groups and defects of GO are beneficial to combine with metal or metal oxide. And the alcohol hydroxyl group on the surface of GO and Ce salt can also constitute a REDOX system, which helps to graft with other substances [19]. These provide the basis for the idea and implementation of the new photocatalyst for the selection of the three species of TiO2, GO, Ce. Although TiO2 doped with GO or Ce has been reported to be more, GO and Ce co-doping has rarely been reported. In order to solve the problem that GO–TiO2 is not responsive to the visible light, we formed GO and Ce co-doped TiO2 composites for the first time by one-step sol–gel method with calcination at 300 °C for 2 h. This new and simple method of preparing Ce–GO–TiO2 nanocomposite could achieve stronger binding between TiO2, GO and Ce, and we also studied the photocatalytic efficiency of this new catalyst for MB degradation.
2 Experimental
2.1 Materials and reagents
Expanded graphite (AF99, 325 mesh, 99 % carbon content) was obtained from the QingdaoLihaofeng Graphite Co., Ltd. Tetrabutyl titanate was used as a Ti-precursor and supplied by Tianjin Guangfu Reagent Co., Ltd. with a purity of 98 %. Absolute ethyl alcohol and acetic acid glacial (analytical reagent) were purchased from Tianjin Fuyu Reagent Co., Ltd. Cerium nitrate hexahydrate (analytical reagent) and methylene blue were obtained from the Tianjin Cameo Reagent Co., Ltd. All other reagents, sulfuric acid (95–98 %), KMnO4, sodium nitrate and H2O2 (30 %) were all in analytical grade, and ultrapure water was prepared in the laboratory.
2.2 Preparation of GO and Ce–GO–TiO2 samples
2.2.1 Synthesis of graphene oxide (GO)
GO was prepared from expanded graphite powder on the basis of the modified Hummers method [20, 21]. In brief, 1.0 g of graphite powder and 0.5 g of NaNO3 were added into 23 mL of cooled (0 °C) concentrated H2SO4, ultrasonic for 1 h. Then, 3 g of KMnO4 was added gradually with continuous stirring, and the temperature of the mixture was maintained below 10 °C. After 2 h, the mixture was moved to water bath and turned on the ultrasonic; the temperature of the water was kept at 35 °C for 30 min. One hundred milliliters of ultrapure water was added slowly to the mixture solution and maintained at a water bath of 98 °C for 15 min with modest mechanical stirring. The reaction was terminated by addition of 60 mL of ultrapure water followed by 25 mL of 30 % H2O2 aqueous solution. The solid product was separated by centrifugation and washed repeatedly with 5 % HCl solution and ultrapure water until sulfate anion could not be detected with BaCl2. The resultant solid was dried in vacuum at 50 °C.
2.2.2 Preparation of Ce–GO–TiO2 samples
Typically, GO (10 mg) was loaded in a 500-mL beaker with 30 mL anhydrous ethanol and subjected to ultrasonic treatment for 1 h, obtaining a homogeneous yellow-brown dispersion, and stirred with the magnetic stirrer for 1 h. Glacial acetic acid (4.00 mL) was then added in and stirred for a while. And then tetrabutyl titanate (5.00 mL) was slowly added into the above suspension and stirred. After stirring for 1 h, another 7 mL mixed solution (including anhydrous alcohol, glacial acetic acid, ultrapure water and different amounts of cerium nitrate) was dropwise added into the reaction solution. The suspension was stirred for 5 h to get sol and aged for 2 days to get gel and dried at 50 °C. The solid was grinded and then annealed in a muffle furnace at 300 °C from ambient temperature, maintained for 2 h and then naturally cooled down to room temperature.
GO–TiO2 and Ce–TiO2 samples were also prepared by the similar method as described above. The dried compound was annealed under 300 °C for 2 h.
2.3 Characterization of materials
The crystalline structure of samples was analyzed by powder X-ray diffraction (XRD) using a Siemens D5000 X-ray diffractometer with Cu Ka radiation (k = 1.54185) operated at 40 kV and 30 mA, respectively. Ultraviolet–visible diffuse reflectance spectroscopy (UV–Vis DRS) of samples was recorded on a Perkin-Elmer Lambda 35 spectrophotometer with a 560 mm integrating sphere and BaSO4 as a reference material. Field emission scanning electron microscopy (FE-SEM), performed on a Zeiss Neon 40EsB, was used to evaluate the morphology, size and texture information of the samples. Brunauer–Emmett–Teller (BET) surface area was determined by nitrogen adsorption/desorption at 77 K, using a Autosorb-1C/TCD Automatic Chemisorption and Physisorption Analyzer manufactured by Quantachrome.
2.4 Photocatalytic tests
Photocatalytic experiments were carried out in winter in the south of China (Changsha); photocatalytic activity of some catalysts was measured by the photodegradation of methylene blue (MB) under simulated sunlight in the low-temperature condition. In a typical process, aqueous solution of MB (10 mg/L, 100 mL) and the photocatalysts (50 mg) were put into a 250-mL container under constant stirring. A 200 W xenon lamp was positioned 10 cm away from the reactor. Before illumination, the suspensions were mildly stirred for about 50 min in the dark in order to achieve adsorption–desorption equilibrium of MB on the catalyst. The photocatalytic reaction was started by turning on the lamp. At given time intervals, the suspension solution was sampled and centrifuged for analysis by recording the MB absorption spectra by a UNIC UV-2000 spectrophotometer at a fixed wavelength of 664 nm. The changes in maximum absorption versus irradiation time (C/C o vs. t) were obtained; these reflected the decrease in the MB concentration.
3 Results and discussion
3.1 XRD analysis
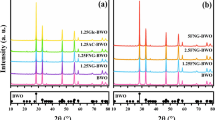
XRD is used to analyze the crystal phase and estimate the crystallite size of different samples. Figure 1 shows XRD patterns of prepared TiO2, GO–TiO2, Ce–TiO2 and Ce–GO–TiO2 photocatalysts. The XRD patterns of TiO2 and GO–TiO2 present no significant difference with the typical diffraction peak (101), (004), (200), (211), etc., of anatase phase and a rutile peak (110), illustrating the existence of grapheme oxide does not influence the crystallization of TiO2. For synthesized Ce–GO–TiO2 and Ce–TiO2, XRD peak at 25.4°, 37.8°, 48.0°, 54.3° and 62.7° is also found, corresponding to the crystal planes (101), (004), (200), (211), (204) of anatase, respectively [22], but rutile peak(110) is disappearing, suggesting that Ce doped in samples can prevent phase transition from anatase to rutile. In addition, the XRD diffraction peak positions of TiO2-doped Ce and the bare TiO2 are basically consistent, indicating that the Ce4+ is not into the lattice of TiO2 and replaces Ti4+, since the radius of Ce4+ (0.102 nm) is much bigger than Ti4+ (0.064 nm). And there is no new phase appearing at the same time, it explains Ce4+ in the form of small clusters of CeO2 evenly dispersed in TiO2 nanoparticles, so that may easily create the bonding of Ti–O–Ce around the anatase crystallite in the process of heat treatment, and these factors suppress the nucleation and growing up of rutile phase [23].
3.2 Scanning electron microscopy analysis
The morphology of GO–TiO2, Ce–GO–TiO2 and GO is shown in Fig. 2. From Fig. 2d, it is certified that the GO sample has a wrinkled paper-like structure. By reason of the different morphology of GO–TiO2 and Ce–GO–TiO2, it is very easy to distinguish them in the SEM. We can see that the Ce–GO–TiO2 has an obvious lamellar structure compared with GO–TiO2 from Fig. 2a, b, which may be due to the addition of Ce to prevent the agglomeration of GO layers; in Fig. 2b, c, the SEM observation demonstrates that the TiO2 nanoparticles are well anchored onto graphene oxide sheets. No Ce particles can be observed directly in the SEM picture, and this indicates that Ce particles are well dispersed in the doped samples, which is consistent with the XRD result (Fig. 1). The average particle size of Ce–GO–TiO2 is approximate to 5 nm in diameter from the SEM images, which is in agreement with the results obtained from BET. This means that Ce ion doping can efficiently restrain the growth of particle size.
3.3 UV–Vis spectra analysis
Figure 3 shows the UV–Vis absorption spectra of TiO2, GO–TiO2, Ce–TiO2 and Ce–GO–TiO2. The absorption edge of prepared TiO2 is limited only to ultraviolet light region; nevertheless, the absorption threshold values of the Ce–TiO2 and Ce–GO–TiO2 are extended up to the visible light range, which demonstrates Ce can make the catalyst that is not responsive to the visible light become one that can utilize visible light. Light absorption ability of GO–TiO2 is significantly improved compared to TiO2 owing to the existence of GO. The energy gap (E g) is determined by the formula [24], E g = 1239.8/λ, where λ (nm) is the wavelength of the absorption edge in the spectrum. The energy gaps of TiO2 doped with Ce, GO and Ce/GO, respectively, are corresponding to 2.52, 2.75 and 2.41 eV (see Table 1). The E g value changed from 3.2 to 2.41 eV is attributed to the incorporation of GO and Ce, which reveals that the co-doping of Ce and GO has obvious synergistic effects.
For the GO–TiO2 sample, the absorbance in the visible light part is significantly strengthened. However, the narrowing of band gap of carbon-doped titania is not very distinct due to the similarity between the 2p orbital energy level of C and the 2p orbital energy level of O [25]. For Ce–TiO2 sample, yellow cerium oxide is formed in TiO2 during the calcination process, which can absorb visible light [26]. In addition, the 4f orbital energy level of Ce below the conduction band of TiO2, the electrons of the valence band of Ce–TiO2 and the ground state of Ce2O3 can be excited into Ce 4f energy level under visible light irradiation, leading to light absorption wavelength red shift [27]. And the synergistic effects of the GO and Ce co-doped samples make the red shift more evident, the absorption spectra of Ce–GO–TiO2 shift from 400 to 550 nm in comparison with prepared TiO2, and red shift of absorption wavelength reaches 150 nm.
3.4 Brunauer–Emmett–Teller analysis
Figure 4 shows the nitrogen adsorption–desorption isotherm and pore size distribution curve of Ce–GO–TiO2. The sample exhibits a type IV adsorption isotherm, which is typical of a well-defined mesoporous structure. The pore size distribution is estimated from the desorption branch of the nitrogen isotherm by the Barrett–Joyner–Halenda (BJH) method [28]. For the typical Ce–GO–TiO2, the mean pore size and BET surface area were 4.54 nm and 237.6 m2 g−1, respectively. Specific surface area of TiO2, Ce–TiO2 and GO–TiO2 was also tested, as shown in Table 1. Compared with TiO2, the larger surface area will make the Ce(0.6 %)–GO–TiO2 adsorb more contaminant and incident illumination.
3.5 Photocatalytic measurements
The photocatalytic activity of TiO2, GO–TiO2, Ce–TiO2 and Ce–GO–TiO2 nanocomposites is assessed by decomposing MB under simulated sunlight irradiation (Fig. 5). The blank test result is also shown in Fig. 5, from which we can know that the MB is hardly degraded under simulated sunlight without photocatalysts, and its degradation percentage is <6 % after 5 h of exposure. After 5-h irradiation in the presence of pure TiO2, about 35.2 % of MB are found to be decomposed. When TiO2 is assembled on graphene oxide, GO–TiO2 exhibits higher photocatalytic activity than bare TiO2 nanoparticle. Ce–TiO2 is also a valid photocatalyst and shows a degradation rate of 53.7 %. In comparison with TiO2, GO–TiO2, Ce(6 %)–TiO2, Ce(1 %)–GO–TiO2 and Ce(6 %)–GO–TiO2, Ce(0.6 %)–GO–TiO2 nanocomposite reveals the highest photocatalytic efficiency, whose photocatalytic degradation rate on MB is up to 97.7 % in the same experimental condition.
To quantify the effects of GO and Ce co-doping on photoactivity, the kinetics of MB photodegradation were fitted using a first-order reaction kinetics equation of ln (C/C 0) = −kt, where C and C 0 are the contaminant concentrations during and at the beginning of an experiment, respectively, k is the reaction rate constant, and t is the time. Figure 5b summarizes the effects of TiO2, GO–TiO2, Ce–TiO2 and Ce–GO–TiO2 on the MB photodegradation kinetics under simulated sunlight, respectively. Under simulated sunlight, the combination of GO, Ce and TiO2 apparently generated a significant synergistic effect: The MB photodegradation kinetics for the Ce(0.6 %)–GO–TiO2 were 13 times than that of the prepared TiO2.
The photocatalytic experiments clearly confirm that the doping of GO and Ce to TiO2 can produce an efficient photocatalyst. The enhanced photocatalytic activity is usually attributed to the increased adsorption ability for organic pollutants on account of the extraordinary high surface area of GO. And Ce ion doping makes the doped samples have smaller average crystal size and absorb most visible light to generate more electron–hole pairs, f orbital of Cerium can make Ce4+/Ce3+ interact directly with the organic molecules, and the electrons trapped by Ce and GO are efficiently transferred to the adsorbed O2 to generate superoxide anion radicals O -2 ·, which is beneficial to react with the organic contaminants.
Figure 6 shows the effect of calcination temperature on the activity of Ce(0.6 %)–GO–TiO2 for MB degradation. The GO and Ce co-doped TiO2 photocatalyst calcined at 300 °C for 2 h exhibits the highest photocatalytic activity under simulated sunlight irradiation of 5 h. The cause of this is owing to the formation of crystalline phase of anatase and the existence of GO, Ce and TiO2 simultaneously. When the calcination temperature is low, such as 100 and 200 °C, the anatase crystallization of Ce(0.6 %)–GO–TiO2 photocatalyst is not complete and the photocatalytic activity of the sample is not very well. On the contrary, when the calcination temperature is high, such as 400 °C, GO is burned and just Ce remains in the sample.
4 Conclusion
To sum up, a hybrid Ce–GO–TiO2 photocatalyst is successfully synthesized using a simple one-step sol–gel method. The addition of Ce can inhibit the agglomeration of GO sheets and make GO–TiO2 have good catalytic ability under visible light. The photocatalytic measurement results demonstrate that the MB photocatalytic degradation rate of the Ce(0.6 %)–GO–TiO2 is up to 97.7 % under simulated sunlight for 5 h. The distinct strength in photoactivity can be put down to the synergistic effects in the GO, Ce and TiO2 system. This work indicates the potential application of Ce–GO–TiO2 in the field of organic pollutant degradation.
References
X. Chen, S.S. Mao, Titanium dioxide nanomaterials: synthesis, properties, modifications, and applications. Chem. Rev. 107, 2891–2959 (2007)
M. Anpo, P.V. Kamat, Environmentally Benign Photocatalysts: Applications of Titanium Oxide-Based Materials (Springer, Berlin, 2010)
M. Mohammadi, D. Fray, Nanostructured TiO2–CeO2 mixed oxides by an aqueous sol–gel process: effect of Ce:Ti molar ratio on physical and sensing properties. Sens. Actuators B Chem. 150, 631–640 (2010)
J. Xie, J. Li, L. Zhao, X. Zhang, B. Yu, R. Wu, R. Wang, J.-H. Liu, F. Xue, S.-T. Yang, Fabrication of TiO2–graphene oxide aerogel for the adsorption of copper ions. Nanosci. Nanotechnol. Lett. 6, 1018–1023 (2014)
G. Jiang, Z. Lin, L. Zhu, Y. Ding, H. Tang, Preparation and photoelectrocatalytic properties of titania/carbon nanotube composite films. Carbon 48, 3369–3375 (2010)
M. Zhu, P. Chen, M. Liu, Graphene oxide enwrapped Ag/AgX (X = Br, Cl) nanocomposite as a highly efficient visible-light plasmonic photocatalyst. ACS Nano 5, 4529–4536 (2011)
B. Li, T. Liu, Y. Wang, Z. Wang, ZnO/graphene-oxide nanocomposite with remarkably enhanced visible-light-driven photocatalytic performance. J. Colloid Interface Sci. 377, 114–121 (2012)
X. Pu, D. Zhang, Y. Gao, X. Shao, G. Ding, S. Li, S. Zhao, One-pot microwave-assisted combustion synthesis of graphene oxide–TiO2 hybrids for photodegradation of methyl orange. J. Alloys Compd. 551, 382–388 (2013)
J. Liu, H. Bai, Y. Wang, Z. Liu, X. Zhang, D.D. Sun, Self-assembling TiO2 nanorods on large graphene oxide sheets at a two-phase interface and their anti-recombination in photocatalytic applications. Adv. Funct. Mater. 20, 4175–4181 (2010)
G. Jiang, Z. Lin, C. Chen, L. Zhu, Q. Chang, N. Wang, W. Wei, H. Tang, TiO2 nanoparticles assembled on graphene oxide nanosheets with high photocatalytic activity for removal of pollutants. Carbon 49, 2693–2701 (2011)
C. Koepke, K. Wisniewski, L. Sikorski, D. Piatkowski, K. Kowalska, M. Naftaly, Upconverted luminescence under 800 nm laser diode excitation in Nd3+-activated fluoroaluminate glass. Opt. Mater. 28, 129–136 (2006)
Y. Wang, H. Cheng, Y. Hao, J. Ma, W. Li, S. Cai, Photoelectrochemical properties of metal-ion-doped TiO2 nanocrystalline electrodes. Thin Solid Films 349, 120–125 (1999)
K. Ranjit, I. Willner, S. Bossmann, A. Braun, Lanthanide oxide doped titanium dioxide photocatalysts: effective photocatalysts for the enhanced degradation of salicylic acid and t-cinnamic acid. J. Catal. 204, 305–313 (2001)
Y. Chai, H. Ding, Z. Zhang, Y. Xian, Z. Pan, L. Jin, Study on photocatalytic oxidation for determination of the low chemical oxygen demand using a nano-TiO2–Ce(SO4)2 coexisted system. Talanta 68, 610–615 (2006)
H. Xia, H. Zhuang, D. Xiao, T. Zhang, Photocatalytic activity of La3+/S/TiO2 photocatalyst under visible light. J. Alloys Compd. 465, 328–332 (2008)
B. Elidrissi, M. Addou, M. Regragui, C. Monty, A. Bougrine, A. Kachouane, Structural and optical properties of CeO2 thin films prepared by spray pyrolysis. Thin Solid Films 379, 23–27 (2000)
Z. Liu, B. Guo, L. Hong, H. Jiang, Preparation and characterization of cerium oxide doped TiO2 nanoparticles. J. Phys. Chem. Solids 66, 161–167 (2005)
A. Worayingyong, S. Sang-urai, M. Smith, S. Maensiri, S. Seraphin, Effects of cerium dopant concentration on structural properties and photocatalytic activity of electrospun Ce-doped TiO2 nanofibers. Appl. Phys. A 117, 1191–1201 (2014)
C. Yagci, U. Yildiz, Redox polymerization of methyl methacrylate with allyl alcohol 1,2-butoxylate-block-ethoxylate initiated by Ce(IV)/HNO3 redox system. Eur. Polym. J. 41, 177–184 (2005)
N.I. Kovtyukhova, P.J. Ollivier, B.R. Martin, T.E. Mallouk, S.A. Chizhik, E.V. Buzaneva, A.D. Gorchinskiy, Layer-by-layer assembly of ultrathin composite films from micron-sized graphite oxide sheets and polycations. Chem. Mater. 11, 771–778 (1999)
J.D. Kim, T. Palani, M.R. Kumar, S. Lee, H.C. Choi, Preparation of reusable Ag-decorated graphene oxide catalysts for decarboxylative cycloaddition. J. Mater. Chem. 22, 20665–20670 (2012)
G. Jiang, X. Zheng, Y. Wang, T. Li, X. Sun, Photo-degradation of methylene blue by multi-walled carbon nanotubes/TiO2 composites. Powder Technol. 207, 465–469 (2011)
J. Lin, C.Y. Jimmy, An investigation on photocatalytic activities of mixed TiO2-rare earth oxides for the oxidation of acetone in air. J. Photochem. Photobiol., A 116, 63–67 (1998)
B. O’regan, M. Grätzel, A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 353, 737–740 (1991)
J.-Y. Lee, J. Park, J.-H. Cho, Electronic properties of N-and C-doped TiO2. Appl. Phys. Lett. 87, 1904 (2005)
M. Sainz, A. Duran, J.F. Navarro, UV highly absorbent coatings with CeO2 and TiO2. J. Non-Cryst. Solids 121, 315–318 (1990)
F. Li, X. Li, M. Hou, K. Cheah, W. Choy, Enhanced photocatalytic activity of Ce3+–TiO2 for 2-mercaptobenzothiazole degradation in aqueous suspension for odour control. Appl. Catal. A 285, 181–189 (2005)
P. Sun, L. Liu, S.-C. Cui, J.-G. Liu, Synthesis, characterization of Ce-doped TiO2 nanotubes with high visible light photocatalytic activity. Catal. Lett. 144, 2107–2113 (2014)
Acknowledgments
The present study was supported by Hunan Provincial Natural Science Foundation of China (2015JJ6025) and International Cooperation Project (2015DFA61170).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, J., Zhang, Q., Zeng, L. et al. Synthesis, characterization and photocatalytic study of graphene oxide and cerium co-doped in TiO2 . Appl. Phys. A 122, 51 (2016). https://doi.org/10.1007/s00339-015-9580-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-015-9580-7