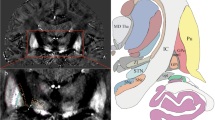
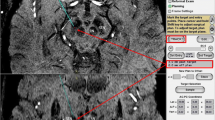
Abstract
Objectives
To image the pedunculopontine tegmental nucleus (PPN), a deep brain stimulation (DBS) target for Parkinson disease, using MRI with validated results.
Methods
This study used the MP2RAGE sequence with high resolution and enhanced grey-white matter contrast on a 7-T ultra-high-field MRI system to image the PPN as well as a diffusion spectrum imaging method on a 3-T MRI system to reconstruct the main fibre systems surrounding the PPN. The coordinates of the rostral and caudal PPN poles of both sides were measured in relation to the third and fourth ventricular landmarks on the 7-T image.
Results
The boundary of the PPN was delineated, and showed morphology consistent with previous histological works. The main fibres around the PPN were reconstructed. The pole coordinate results combined with the fibre spatial relationships validate the imaging results.
Conclusions
A practical protocol is provided to directly localise the PPN using MRI; the position and morphology of the PPN can be obtained and validated by locating its poles relative to two ventricular landmarks and by inspecting its spatial relationship with the surrounding fibre systems. This technique can be potentially used in clinics to define the boundary of the PPN before DBS surgery for treatment of Parkinson disease in a more precise and reliable manner.
Key points
• Combined information helps localise the PPN as a DBS target for PD patients
• Scan the PPN at 7 T and measure its coordinates against different ventricular landmarks
• Reconstruct the main fibres around the PPN using diffusion spectrum imaging






Similar content being viewed by others
Abbreviations
- AC:
-
Anterior commissure
- CA:
-
Cerebral aqueduct
- CTT:
-
Central tegmental tracts
- DBS:
-
Deep brain stimulation
- DIV:
-
MP2RAGE two-echo divided image
- DSCP:
-
Decussation of the superior cerebellar peduncle
- DSI:
-
Diffusion spectrum imaging
- DSI0:
-
DSI image with b value = 0
- DTI:
-
Diffusion tensor imaging
- ML:
-
Medial lemniscus
- PAG:
-
Periaqueductal grey
- PC:
-
Posterior commissures
- PD:
-
Parkinson disease
- Point B:
-
Base point of the B-F coordinate
- Point F:
-
Fastigial point of the B-F coordinate
- PPN:
-
Pedunculopontine tegmental nucleus
- QA:
-
Mean quantitative anisotropy
- REF:
-
Second echo of the MP2RAGE image
- SCP:
-
Superior cerebellar peduncle
- SNc:
-
Substantia nigra pars compacta
- STN:
-
Subthalamic nucleus
References
Olszewski J, Baxter D (1982) Cytoarchitecture of the human brain stem. Karger, Switzerland
Paxinos G, Huang XF (1995) Atlas of the human brainstem. Academic Press, San Diego
Strumpf H, Noesselt T, Schoenfeld MA et al (2016) Deep Brain Stimulation of the Pedunculopontine Tegmental Nucleus (PPN) Influences Visual Contrast Sensitivity in Human Observers. PLoS ONE 11:e0155206
Okada K, Kobayashi Y (2013) Reward prediction-related increases and decreases in tonic neuronal activity of the pedunculopontine tegmental nucleus. Front Integr Neurosci 7:36
Hong S, Hikosaka O (2014) Pedunculopontine tegmental nucleus neurons provide reward, sensorimotor, and alerting signals to midbrain dopamine neurons. Neuroscience 282:139–155
Goetz L, Piallat B, Bhattacharjee M, Mathieu H, David O, Chabardès S (2016) The primate pedunculopontine nucleus region: towards a dual role in locomotion and waking state. J Neural Transm 123:667–678
Pahapill PA, Lozano AM (2000) The pedunculopontine nucleus and Parkinson's disease. Brain 123:1767–1783
Fournier-Gosselin M-P, Lipsman N, Saint-Cyr JA, Hamani C, Lozano AM (2013) Regional anatomy of the pedunculopontine nucleus: Relevance for deep brain stimulation. Mov Disord 28:1330–1336
Karachi C, Grabli D, Bernard F et al (2010) Cholinergic mesencephalic neurons are involved in gait and postural disorders in Parkinson disease. J Clin Invest 120:2745–2754
Kojima J, Yamaji Y, Matsumura M et al (1997) Excitotoxic lesions of the pedunculopontine tegmental nucleus produce contralateral hemiparkinsonism in the monkey. Neurosci Lett 226:111–114
Nandi D, Aziz TZ, Giladi N, Winter J, Stein JF (2002) Reversal of akinesia in experimental parkinsonism by GABA antagonist microinjections in the pedunculopontine nucleus. Brain 125:2418–2430
Karachi C, André A, Bertasi E, Bardinet E, Lehéricy S, Bernard FA (2012) Functional parcellation of the lateral mesencephalus. J Neurosci 32:9396–9401
Snijders AH, Leunissen I, Bakker M et al (2011) Gait-related cerebral alterations in patients with Parkinson’s disease with freezing of gait. Brain 134:59–72
Mazzone P, Lozano A, Stanzione P et al (2005) Implantation of human pedunculopontine nucleus: a safe and clinically relevant target in Parkinson's disease. NeuroReport 16:1877–1881
Plaha P, Gill SS (2005) Bilateral deep brain stimulation of the pedunculopontine nucleus for Parkinson's disease. NeuroReport 16:1883–1887
Moro E, Hamani C, Poon YY et al (2010) Unilateral pedunculopontine stimulation improves falls in Parkinson's disease. Brain 133:215–224
Ferraye MU, Debu B, Fraix V et al (2010) Effects of pedunculopontine nucleus area stimulation on gait disorders in Parkinson's disease. Brain 133:205–214
Schaltenbrand G, Wahren W (1977) Atlas for stereotaxy of the human brain. Thieme, Stuttgart, New York
Hariz MI, Krack P, Melvill R et al (2003) A quick and universal method for stereotactic visualization of the subthalamic nucleus before and after implantation of deep brain stimulation electrodes. Stereotact Funct Neurosurg 80:96–101
Weinberger M, Hamani C, Hutchison WD, Moro E, Lozano AM, Dostrovsky JO (2008) Pedunculopontine nucleus microelectrode recordings in movement disorder patients. Exp Brain Res 188:165–174
Shimamoto SA, Larson PS, Ostrem JL, Glass GA, Turner RS, Starr PA (2010) Physiological identification of the human pedunculopontine nucleus. J Neurol Neurosurg Psychiatry 81:80–86
Zrinzo L, Zrinzo LV, Tisch S et al (2008) Stereotactic localization of the human pedunculopontine nucleus: atlas-based coordinates and validation of a magnetic resonance imaging protocol for direct localization. Brain 131:1588–1598
Zrinzo L, Zrinzo L, Massey L et al (2011) Targeting of the pedunculopontine nucleus by an MRI-guided approach: a cadaver study. J Neural Transm 118:1487–1495
Alho AT, Hamani C, Alho EJ et al (2017) Magnetic resonance diffusion tensor imaging for the pedunculopontine nucleus: proof of concept and histological correlation. Brain Struct Funct. https://doi.org/10.1007/s00429-016-1356-0:1-12
Marques JP, Kober T, Krueger G, van der Zwaag W, Van de Moortele P-F, Gruetter R (2010) MP2RAGE, a self bias-field corrected sequence for improved segmentation and T1-mapping at high field. Neuroimage 49:1271–1281
Wedeen VJ, Hagmann P, Tseng W-YI, Reese TG, Weisskoff RM (2005) Mapping complex tissue architecture with diffusion spectrum magnetic resonance imaging. Magn Reson Med 54:1377–1386
Feinberg DA, Moeller S, Smith SM et al (2010) Multiplexed Echo Planar Imaging for Sub-Second Whole Brain FMRI and Fast Diffusion Imaging. PLoS ONE 5:e15710
Setsompop K, Cohen-Adad J, Gagoski BA et al (2012) Improving diffusion MRI using simultaneous multi-slice echo planar imaging. Neuroimage 63:569–580
Rosset A, Spadola L, Ratib O (2004) OsiriX: An Open-Source Software for Navigating in Multidimensional DICOM Images. J Digit Imaging 17:205–216
Afshar F, Watkins ES, Yap JC (1978) Stereotaxic atlas of the human brainstem and cerebellar nuclei: a variability study. Raven Press, New York
Lay DC (2012) Linear Algebra and Its Applications, 4th edn. Addison Wesley, Boston
Yeh FC, Wedeen VJ, Tseng WY (2010) Generalized q-sampling imaging. IEEE Trans Med Imaging 29:1626–1635
Fedorov A, Beichel R, Kalpathy-Cramer J et al (2012) 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magn Reson Imaging 30:1323–1341
Wedeen VJ, Wang RP, Schmahmann JD et al (2008) Diffusion spectrum magnetic resonance imaging (DSI) tractography of crossing fibers. Neuroimage 41:1267–1277
Fernandez-Miranda JC, Pathak S, Engh J et al (2012) High-definition fiber tractography of the human brain: neuroanatomical validation and neurosurgical applications. Neurosurgery 71:430–453
Stefani A, Lozano AM, Peppe A et al (2007) Bilateral deep brain stimulation of the pedunculopontine and subthalamic nuclei in severe Parkinson's disease. Brain 130:1596–1607
Jenkinson N, Brittain JS, Hicks SL, Kennard C, Aziz TZ (2012) On the Origin of Oscillopsia during Pedunculopontine Stimulation. Stereotact Funct Neurosurg 90:124–129
Gut NK, Winn P (2015) Deep brain stimulation of different pedunculopontine targets in a novel rodent model of parkinsonism. J Neurosci 35:4792–4803
Thevathasan W, Coyne TJ, Hyam JA et al (2011) Pedunculopontine nucleus stimulation improves gait freezing in Parkinson disease. Neurosurgery 69:1248–1254
Thevathasan W, Silburn PA, Brooker H et al (2010) The impact of low-frequency stimulation of the pedunculopontine nucleus region on reaction time in parkinsonism. J Neurol Neurosurg Psychiatry 81:1099–1104
Welter M-L, Demain A, Ewenczyk C et al (2015) PPNa-DBS for gait and balance disorders in Parkinson’s disease: a double-blind, randomised study. J Neurol 262:1515–1525
Acknowledgements
We thank Mr. Jing Luo and Ms. Hong Xu for their assistance with the experimental conditions and Dr. Penghu Wei and Dr. Xu Yan for their technical advice.
Funding
This study has received funding from the Chinese MOST (Ministry of Science and Technology of China) “973” grant (2015CB351701), NSFC (National Natural Science Foundation of China) grants (31730039, 81601060), and Beijing Municipal Commission of Science and Technology grant (Z161100000116059).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is Yan Zhuo.
Conflict of interest
The author Jing An is an employee of Siemens responsible for customer research support. The other authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• experimental
• performed at one institution
Additional information
Yu-Qing Zhang and Yan Zhuo are joint corresponding authors.
Rights and permissions
About this article
Cite this article
Cong, F., Wang, JW., Wang, B. et al. Direct localisation of the human pedunculopontine nucleus using MRI: a coordinate and fibre-tracking study. Eur Radiol 28, 3882–3892 (2018). https://doi.org/10.1007/s00330-017-5299-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-5299-5