Abstract
Objectives
To investigate the potential of DCE-MRI to discriminate responders from non-responders after neoadjuvant chemo-radiotherapy (CRT) for locally advanced rectal cancer (LARC). We investigated several shape parameters for the time-intensity curve (TIC) in order to identify the best combination of parameters between two linear parameter classifiers.
Methods
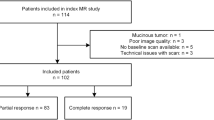
Seventy-four consecutive patients with LARC were enrolled in a prospective study approved by our ethics committee. Each patient gave written informed consent. After surgery, pathological TNM and tumour regression grade (TRG) were estimated. DCE-MRI semi-quantitative analysis (sqMRI) was performed to identify the best parameter or parameter combination to discriminate responders from non-responders in response monitoring to CRT. Percentage changes of TIC shape descriptors from the baseline to the presurgical scan were assessed and correlated with TRG. Receiver operating characteristic analysis and linear classifier were applied.
Results
Forty-six patients (62.2 %) were classified as responders, while 28 subjects (37.8 %) were considered as non-responders. sqMRI reached a sensitivity of 93.5 % and a specificity of 82.1 % combining the percentage change in Maximum Signal Difference (ΔMSD) and Wash-out Slope (ΔWOS), the Standardized Index of Shape (SIS).
Conclusions
SIS obtains the best result in discriminating responders from non-responders after CRT in LARC, with a cut-off value of −3.0 %.
Key Points
• DCE-MRI shape descriptors are investigated to assess preoperative CRT response in LARC.
• Identification of the best TIC shape descriptors combination through a linear classifier.
• Identification of a single MRI index to predict neoadjuvant treatment response.






Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A (2012) Cancer statistics, 2012. CA Cancer J Clin 62:10–29
Avallone A, Delrio P, Guida C et al (2006) Biweekly oxaliplatin, raltitrexed, 5-fluorouracil and folinic acid combination chemotherapy during preoperative radiation therapy for locally advanced rectal cancer: a phase I-II study. Br J Cancer 94:1809–1815
Janjan NA, Crane C, Feig BW et al (2001) Improved overall survival among responders to preoperative chemoradiation for locally advanced rectal cancer. Am J Clin Oncol 24:107–112
Beets-Tan RG, Beets GL (2004) Rectal cancer: review with emphasis on MR imaging. Radiology 232:335–346
Leach MO, Brindle KM, Evelhoch JL et al (2005) The assessment of antiangiogenic and antivascular therapies in early-stage clinical trials using magnetic resonance imaging: issues and recommendations. Br J Cancer 92:1599–1610
Salmo E, El-Dhuwaib Y, Haboubi NY (2010) Histological grading of tumour regression and radiation colitis in locally advanced rectal cancer following neo-adjuvant therapy; a critical appraisal. Color Dis. doi:10.1111/j.1463-1318.2010.02412.x
Daniel BL, Yen YF, Glover GH et al (1998) Breast disease: dynamic spiral MR imaging. Radiology 209:499–509
Gollub MJ, Gultekin DH, Akin O, Do RK, Fuqua JL 3rd, Gonen M et al (2012) Dynamic contrast enhanced-MRI for the detection of pathological complete response to neoadjuvant chemotherapy for locally advanced rectal cancer. Eur Radiol 22:821–831
Petrillo A, Fusco R, Petrillo M, Granata V, Filice S, Delrio P et al (2014) Dynamic contrast enhanced-MRI in locally advanced rectal cancer: value of time intensity curve visual inspection to assess neo-adjuvant therapy response. J Physiol Health 110:255–267
Hong HS, Kim SH, Park HJ, Park MS, Kim KW, Kim WH et al (2013) Correlations of dynamic contrast-enhanced magnetic resonance imaging with morphologic, angiogenic, and molecular prognostic factors in rectal cancer. Yonsei Med J 54:123–130
Bajpai J, Gamanagatti S, Sharma MC, Kumar R, Vishnubhatla S, Khan SA et al (2010) Noninvasive imaging surrogate of angiogenesis in osteosarcoma. Pediatr Blood Cancer 54:526–531
Zhang XM, Yu D, Zhang HL et al (2008) 3D dynamic contrast-enhanced MRI of rectal carcinoma at 3T: correlation with microvascular density and vascular endothelial growth factor markers of tumor angiogenesis. J Magn Reson Imaging 27:1309–1316
Knopp MV, Weiss E, Sinn HP et al (1999) Pathophysiologic basis of contrast enhancement in breast tumors. J Magn Reson Imaging 10:260–266
Pham CD, Roberts TP, van Bruggen N et al (1998) Magnetic resonance imaging detects suppression of tumor vascular permeability after administration of antibody to vascular endothelial growth factor. Cancer Investig 16:225–230
Fusco R, Sansone M, Maffei S, Petrillo A (2012) Dynamic contrast-enhanced MRI in breast cancer: a comparison between distributed and compartmental tracer kinetic models. J Biomed Graph Comput 2:23–36
Evelhoch JL (1999) Key factors in the acquisition of contrast kinetic data for oncology. J Magn Reson Imaging 10:254–259
Fusco R, Petrillo A, Petrillo M, Sansone M (2013) Use of tracer kinetic models for selection of semi-quantitative features for DCE-MRI data classification. Appl Magn Reson 44:1311–1324
Guo JY, Reddick WE (2009) DCE-MRI pixel-by-pixel quantitative curve pattern analysis and its application to osteosarcoma. J Magn Reson Imaging 30:177–184
Avallone A, Delrio P, Pecori B et al (2011) Oxaliplatin plus dual inhibition of thymidilate synthase during preoperative pelvic radiotherapy for locally advanced rectal carcinoma: long-term outcome. Int J Radiat Oncol Biol Phys 79:670–676
Petrillo A, Catalano O, Delrio P et al (2007) Post-treatment fistulas in patients with rectal cancer: MRI with rectal superparamagnetic contrast agent. Abdom Imaging 32:328–331
Dresen RC, Beets GL, Rutten HJ et al (2009) Locally advanced rectal cancer: MR imaging for restaging after neoadjuvant radiation therapy with concomitant chemotherapy. Part I. Are we able to predict tumor confined to the rectal wall? Radiology 252:81–91
Andreola S, Leo E, Belli F et al (2001) Adenocarcinoma of the lower third of the rectum surgically treated with a <10-mm distal clearance: preliminary results in 35 N0 patients. Ann Surg Oncol 8:611–615
Mandard AM, Dalibard F, Mandard JC, Marnay J, Henry-Amar M, Petiot JF et al (1994) Pathologic assessment of tumor regression after preoperative chemoradiotherapy of esophageal carcinoma. Clinicopathologic correlations. Cancer 73:2680–2686
Obuchowski NA (2005) ROC analysis. AJR Am J Roentgenol 184:364–372
De Long ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837–845
Schisterman EF, Perkins NJ, Liu A, Bondell H (2005) Optimal cut-point and its corresponding Youden Index to discriminate individuals using pooled blood samples. Epidemiology 16:73–81
Schabel MC, Morrell GR, Oh KY, Walczak CA, Barlow RB, Neumayer LA (2010) Pharmacokinetic mapping for lesion classification in dynamic breast MRI. J Magn Reson Imaging 31:1371–1378
Lavini C, de Jonge MC, van de Sande MG, Tak PP, Nederveen AJ, Maas M (2007) Pixel-by-pixel analysis of DCE MRI curve patterns and an illustration of its application to the imaging of the musculoskeletal system. Magn Reson Imaging 25:604–612
Tuncbilek N, Karakas HM, Altaner S (2004) Dynamic MrI in indirect estimation of microvessel density, histologic grade, and prognosis in colorectal adenocarcinomas. Abdom Imaging 29:166–172
Blomqvist L, Fransson P, Hindmarsh T (1998) The pelvis after surgery and radio-chemotherapy for rectal cancer studied with Gd-DTPA-enhanced fast dynamic MR imaging. Eur Radiol 8:781–787
Kim SH, Lee JM, Gupta SN, Han JK, Choi BI (2014) Dynamic contrast-enhanced MRI to evaluate the therapeutic response to neoadjuvant chemoradiation therapy in locally advanced rectal cancer. J Magn Reson Imaging 40:730–737
Hötker AM, Schmidtmann I, Oberholzer K, Düber C (2014) Dynamic contrast enhanced-MRI in rectal cancer: inter- and intraobserver reproducibility and the effect of slice selection on pharmacokinetic analysis. J Magn Reson Imaging 40:715–722
Intven M, Reerink O, Philippens ME (2014) Dynamic contrast enhanced MR imaging for rectal cancer response assessment after neo-adjuvant chemoradiation. J Magn Reson Imaging Aug 14
Lim JS, Kim D, Baek SE, Myoung S, Choi J, Shin SJ et al (2012) Perfusion MRI for the prediction of treatment response after preoperative chemoradiotherapy in locally advanced rectal cancer. Eur Radiol 22:1693–1700
Park MJ, Kim SH, Lee SJ, Jang KM, Rhim H (2011) Locally advanced rectal cancer: added value of diffusion-weighted MR imaging for predicting tumor clearance of the mesorectal fascia after neoadjuvant chemotherapy and radiation therapy. Radiology 260:771–780
Curvo-Semedo L, Lambregts DM, Maas M, Thywissen T, Mehsen RT, Lammering G et al (2011) Rectal cancer: assessment of complete response to preoperative combined radiation therapy with chemotherapy–conventional MR volumetry versus diffusion-weighted MR imaging. Radiology 260:734–743
DeVries AF, Kremser C, Hein PA et al (2003) Tumor microcirculation and diffusion predict therapy outcome for primary rectal carcinoma. Int J Radiat Oncol Biol Phys 56:958–965
Cai PQ, Wu YP, An X, Qiu X, Kong LH, Liu GC et al (2014) Simple measurements on diffusion-weighted MR imaging for assessment of complete response to neoadjuvant chemoradiotherapy in locally advanced rectal cancer. Eur Radiol 24:2962–2970
Genovesi D, Filippone A, Ausili Cèfaro G, Trignani M, Vinciguerra A, Augurio A et al (2013) Diffusion-weighted magnetic resonance for prediction of response after neoadjuvant chemoradiation therapy for locally advanced rectal cancer: preliminary results of a monoinstitutional prospective study. Eur J Surg Oncol 39:1071–1078
Wu LM, Zhu J, Hu J, Yin Y, Gu HY, Hua J et al (2013) Is there a benefit in using magnetic resonance imaging in the prediction of preoperative neoadjuvant therapy response in locally advanced rectal cancer? Int J Colorectal Dis 28:1225–1238
Song I, Kim SH, Lee SJ, Choi JY, Kim MJ, Rhim H (2012) Value of diffusion-weighted imaging in the detection of viable tumour after neoadjuvant chemoradiation therapy in patients with locally advanced rectal cancer: comparison with T2 weighted and PET/CT imaging. Br J Radiol 85:577–586
Cascini GL, Avallone A, Delrio P, Guida C, Tatangelo F, Marone P et al (2006) 18F-FDG PET is an early predictor of pathologic tumor response to preoperative radiochemotherapy in locally advanced rectal cancer. J Nucl Med 47:1241–1248
Veronesi G, Landoni C, Pelosi G et al (2002) Fluoro-deoxi-glucose uptake and angiogenesis are independent biological features in lung metastases. Br J Cancer 86:1391–1395
Acknowledgments
The scientific guarantor of this publication is Dr. Antonella Petrillo. The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article. The authors state that this work has not received any funding. One of the authors has significant statistical expertise. Institutional Review Board approval was obtained. Written informed consent was obtained from all subjects (patients) in this study. Approval from the institutional animal care committee was not required because there are no animal subjects in this study. No study subjects or cohorts have been previously reported. Methodology: Prospective, diagnostic study, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Petrillo, A., Fusco, R., Petrillo, M. et al. Standardized Index of Shape (SIS): a quantitative DCE-MRI parameter to discriminate responders by non-responders after neoadjuvant therapy in LARC. Eur Radiol 25, 1935–1945 (2015). https://doi.org/10.1007/s00330-014-3581-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-014-3581-3