Abstract
Key message
Ca2+ NPs enhanced tolerance of Triticale callus under salt stress by improving biochemical activity and confocal laser scanning analysis, conferring salt tolerance on callus cells.
Abstract
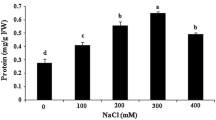
CaO NPs (Ca2+) are significant components that act as transducers in many adaptive and developmental processes in plants. In this study, effect of Ca2+ NPs on the response and regulation of the protective system in Triticale callus under short and long-salt treatments was investigated. The activation of Ca2+ NPs was induced by salt stress in callus of Triticale cultivars. MDA, H2O2, POD, and protein activities were determined in callus tissues. Concerning MDA, H2O2, protein activities, it was found that the Ca2+ NPs treatment was significant, and it demonstrated a high correlation with the tolerance levels of cultivars. Tatlıcak cultivar was detected for better MDA activities in the short time with 1.5 ppm Ca2+ NPs concentration of 50 g and 100 g NaCl. Similarly, the same cultivar responded with better H2O2 activity at 1.5 ppm Ca2+ NPs 100 g NaCl in the short time. POD activities exhibited a decreasing trend in response to the increasing concentrations of Ca2+ NPs. The best result was observed at 1.5 ppm Ca2+ NPs 100 g NaCl in the short term. Based on the protein content, treatment of short-term cultured callus cells with 1.5 ppm Ca2+ NPs inhibited stress response and it significantly promoted Ca2+ NPs signals as compared to control callus. Confocal laser scanning analysis proved that the application of Ca2+ NPs could alleviate the adverse effects of salt stress by the inhibition of stress severity in callus cells. This study demonstrated, under in vitro conditions, that the application of Ca2+ NPs can significantly suppress the adverse effects of salt stress on Triticale callus; it was also verified that the concentration of Ca2+ NPs could be important parameter to be considered in adjusting the micronutrient content in the media for this plant.








Similar content being viewed by others
References
Ali A, Zafar H, Zia M, Haq I, Phull AR, Ali JS, Hussain A (2016) Synthesis, characterization, applications, and challenges of iron oxide nanoparticles. Nanotechnol Sci Appl 9:49–67. https://doi.org/10.2147/NSA.S99986
Anantharaman A, Ramalakshmi S, Mary G (2016) Green synthesis of calcium oxide nanoparticles and its applications. J Eng Res Appl 10:27–31
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Berridge MJ, Bootman MD, Roderick HL (2003) Calcium signalling dynamics, homeostasis and remodelling. Nat Rev Mol Cell Biol 4(7):517–529. https://doi.org/10.1038/nrm1155
Bezirganoglu I (2017) Response of five Triticale genotypes to salt stress in in vitro culture. Turk J Agric For 41(5):372–380. https://doi.org/10.3906/tar-1703-103
Chance B, Maehly AC (1955) Assay of catalase and peroxidase. Methods Enzymol 2:764–775. https://doi.org/10.1002/9780470110171.ch14
Chugh A, Khurana P (2003) Regeneration via somatic embryogenesis from leaf basal segments and genetic transformation of bread and emmer wheat by particle bombardment. Plant Cell Tissue Organ Cult 74(2):151–161
Dietrich P, Anshütz U, Kugler A, Becker D (2010) Physiology and biophysics of plant ligand-gated ion channels. Plant Biol 1:80–93. https://doi.org/10.1111/j.1438-8677.2010.00362.x
Ditta A, Arshad M (2016) Applications and perspectives of using nanomaterials for sustainable plant nutrition. Nanotechnol Rev 5(2):209–229. https://doi.org/10.1515/ntrev-2015-0060
Dubrovina AS, Aleynova OA, Kiselev KV (2016) Influence of overexpression of the true and false alternative transcripts of calcium-dependent protein kinase CPK9 and CPK3 genes on the growth, stress tolerance, and resveratrol content in Vitis amurensis cell cultures. Acta Physiol Plant 38:78
Elmaghrabi AM, Francis D, Rogers HJ, Ochatt SJ (2019) Nuclear migration: an indicator of plant salinity tolerance in vitro. Front Plant Sci 10:783. https://doi.org/10.3389/fpls.2019.00783
El-Sharkawy MS, El-Beshsbeshy TR, Mahmoud EK, Abdelkader NI, Al-Shal RM, Missaoui AM (2017) Response of Alfalfa under salt stress to the application of potassium sulfate nanoparticles. Am J Plant Sci 8:1751–1773. https://doi.org/10.4236/ajps.2017.88120
Erdal S (2012) Androsterone-induced molecular and physiological changes in maize seedlings in response to chilling stress. Plant Physiol Biochem 57:1–7. https://doi.org/10.01016/j.plaphy.2012.04.016
Etienne H, Berger A, Carron MP (1991) Water status of callus from Hevea brasiliensis during induction of somatic embryogenesis. Physiol Plant 82:213–218. https://doi.org/10.1111/j.1399-3054.1991.tb00083.x
Farooq MA, Saqib ZA, Akhtar J (2015) Silicon mediated oxidative stress tolerance and genetic variability in rice (Oryza sativa L.) grown under combined stress of salinity and boron toxicity. Turk J Agric For 39:718–729. https://doi.org/10.3906/tar-1410-26
Finka A, Cuendet AF, Maathuis FJ, Saidi Y, Goloubinoff P (2012) Plasma membrane cyclic nucleotide gated calcium channels control land plant thermal sensing and acquired thermotolerance. Plant Cell 24:3333–3348. https://doi.org/10.1105/tpc.112.095844
Flores-López LZ, Espinoza-Gómez H, Somanathan R (2019) Silver nanoparticles: electron transfer, reactive oxygen species, oxidative stress, beneficial and toxicological effects. J Appl Toxicol 39(1):16–26. https://doi.org/10.1002/jat.3654
Foyer CH, Noctor G (2005) Redox homeostasis and antioxidant signaling: A metabolic interface between stress perception and physiological responses. Plant Cell 17:1866–1875. https://doi.org/10.1105/tpc.105.033589
Frietsch S, Wang YF, Sladek C (2007) A cyclic nucleotide-gated channel is essential for polarized tip growth of pollen. Proc Natl Acad Sci USA 104:14531–14536. https://doi.org/10.1073/pnas.0701781104
Fruk L, Crocker L (2019) Flavin conjugated polydopamine nanoparticles displaying light-driven monooxygenase activity. Front Chem 7:278. https://doi.org/10.3389/fchem.2019.00278
Gohari G, Mohammadi A, Akbari A, Panahirad S, Dadpour MR, Fotopoulos V, Kimura S (2020) Titanium dioxide nanoparticles (TiO2 NPs) promote growth and ameliorate salinity stress effects on essential oil profile and biochemical attributes of Dracocephalum moldavica. Sci Rep 10(1):1–14
González-Melendi P, Fernández-Pacheco R, Coronado MJ, Corredor E, Testillano PS, Risueno MC, Perez-de-Luque A (2008) Nanoparticles as smart treatment-delivery systems in plants: assessment of different techniques of microscopy for their visualization in plant tissues. Ann Bot 101(1):187–195. https://doi.org/10.1093/aob/mcm283
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplast I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 25:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Hepler PK (2005) Calcium A: central regulator of plant growth and development. Plant Cell 17(8):2142–2155. https://doi.org/10.1105/tpc.105.032508
Hills MJ, Hall LM, Messenger DF, Graf RJ, Beres BL, Eudes F (2007) Evaluation of crossability between triticale (X Triticosecale Wittmack) and common wheat, durum wheat and rye. Environ Biosaf Res 6(4):249–257. https://doi.org/10.1051/ebr:2007046
Hussain S, Khan F, Hussain HA, Nie L (2016) Physiological and biochemical mechanisms of seed priming-induced chilling tolerance in rice cultivars. Front Plant Sci 9(7):116. https://doi.org/10.3389/fpls.2016.00116
Hwang KE, Kim HW, Choi YS, Lee SYE, Yeo J, Ham YKS, Choi M, Lee MA, Kim CJ (2013) Evaluationof the antioxidant effect of ganghwayakssuk (Artemisia princeps Pamp.) extract alone and in combination with ascorbic acid inraw chicken patties. Europe PMC 95(3):593–602. https://doi.org/10.3382/ps.2013-03274
Jaleel CA, Sankar B, Sridharan R, Panneerselvam R (2007) Soil salinity alters growth, chlorophyll content, and secondary metabolite accumulation in Catharanthus roseus. Turk J Biol 32:79–83
Karaduman I, Nevin D, Acar S (2017) Green synthesis of γ-Fe2O3 nanoparticles for methane gas sensing. J Mater Sci 28:16094–16105
Khan MN, Siddiqui MH, Mohammad F, Naeem M (2012) Interactive role of nitric oxide and calcium chloride in enhancing tolerance to salt stress. Nitric Oxide 127(4):210–218. https://doi.org/10.1016/j.niox.2012.07.005
Kudla J, Batistic O, Hashimoto K (2010) Calcium signals: the lead currency of plant information processing. Plant Cell 22(3):541–563. https://doi.org/10.1105/tpc.109.072686
Kugler A, Köhler B, Palme K, Wolff P, Dietrich P (2009) Salt-dependent regulation of a CNG channel subfamily in Arabidopsis. BMC Plant Biol 27(9):140. https://doi.org/10.1186//1471-2229-9-140
Lee CW, Mahendra S, Zodrow K, Li D, Tsai YC, Braam J, Alvarez PJJ (2010) Developmental phytotoxicity of metal oxide nanoparticles to Arabidopsis thaliana. Environ Toxicol Chem 29(3):669–675. https://doi.org/10.1002/etc.58
Liang Y, Zhang W, Chen Q, Ding R (2005) Effects of silicon on H+-ATPase and H+-PPase activity, fatty acid composition and fluidity of tonoplast vesicles from roots of salt-stressed barley (Hordeum vulgare L.). Environ Exp Bot 53(1):29–37. https://doi.org/10.1016/j.envexpbot.2004.02.010
Lin SJ, Reppert J, Hu Q, Hudson JS, Reid ML, Ratnikova TA, Rao AM, Luo H, Ke PC (2009) Uptake, translocation, and transmission of carbon nanomate- rials in rice plants. Small 5:1128–1132. https://doi.org/10.1002/smll.200801556
Manchanda G, Garg N (2008) Salinity and its effects on the functional biology of legumes. Acta Physiol Plant 30:595–618. https://doi.org/10.1007/s11738-008-0173-3
Marmiroli M, Pagano L, Savo Sardaro ML, Villani M, Marmiroli N (2014) Genome-wide approach in Arabidopsis thaliana to assess the toxicity of cadmium sulfide quantum dots. Environ Sci Technol 48(10):5902–5909. https://doi.org/10.1021/es404958r
Minta A, Kao JP, Tsien RY (1989) Fluorescent indicators for cytosolic calcium based on rhodamine and fluorescein chromophores. Biol Chem 15(14):8171–8178
Montoro P, Etienne H, Michaux-Ferriere N, Carron MP (1994) Maintainable somatic embryogenesis in Hevea brasiliensis. Rev Cytol Biol Veget Bot 17(1):113–119. https://doi.org/10.1007/BF00233313
Mozafari A, Havas F, Ghaderi N (2018) Application of iron nanoparticles and salicylic acid in in vitro culture of strawberries (Fragaria × ananassa Duch.) to cope with drought stress. Plant Cell Tissue Org Cult 33:1–13
Munns R, Tester M (2008) Mechanisms of Salinity Tolerance. Annu Rev Plant Biol 59:651–681
Murashige T, Skoog FA (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Plant Physiol 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Nadaroğlu H, Alaylı Güngör A, Ince S (2017) Synthesis of nanoparticles by green synthesis method. Int J Innov Res Rev 1(1):6–9
Nalci OB, Nadaroglu H, Pour AH, Gungor AA, Haliloglu K (2019) Effects of ZnO, CuO and γ-Fe3O4 nanoparticles on mature embryo culture of wheat (Triticum aestivum L.). Plant Cell Tissue and Organ Culture (PCTOC) 136(2):269–277. https://doi.org/10.1007/s11240-018-1512-8
Nawaz F, Ahmad R, Ashraf MY, Waraich EA, Khan SZ (2015) Effect of selenium foliar spray on physiological and biochemical processes and chemical constituents of wheat under drought stress. Ecotoxicol Environ Saf 113:191–200. https://doi.org/10.1016/j.ecoenv.2014.12.003
Noctor G, Veljovic-Jovanovic S, Driscoll S, Novitskaya L, Foyer CH (2002) Drought and oxidative load in the leaves of C3 plants: a predominant role for photorespiration. Ann Bot 89(7):841–850. https://doi.org/10.1093/aob/mcf096
Nomura H, Komori T, Uemura S, Kanda Y, Shimotani K, Nakai K, Furuichi T, Takebayashi K, Sugimoto T, Sano S (2013) Chloroplast-mediated activation of plant immune signalling in Arabidopsis. Nat Commun 26(3):926. https://doi.org/10.1038/ncomms19226
Palocci C, Valletta A, Chronopoulou L, Donati L, Bramosanti M, Brasili E, Baldan B, Pasqua G (2017) Endocytic pathways involved in PLGA nanoparticle uptake by grapevine cells and role of cell wall and membrane in size selection. Plant Cell Rep 36(12):1917–1928. https://doi.org/10.1007/s00299-017-2206-0
Parida AK, Das AB (2005) Salt tolerance and salinity effects on plants. Ecotoxicol Environ Saf 60(3):324–349. https://doi.org/10.1016/j.ecoenv.2004.06.010
Parihar P, Singh S, Singh R, Singh VP, Prasad SM (2015) Effect of salinity stress on plants and its tolerance strategies. Environ Sci Pollut Res Int 22(6):4056–4075. https://doi.org/10.1007/s11356-014-3739-1
Patnaik D, Vishnudasan D, Khurana P (2006) Agrobacterium mediated transformation of mature embryos of Triticum aestivum and Triticum durum. Curr Sci 91(3):307–317
Rico CM, Majumdar S, Duarte-Gardea M, Peralta-Videa JR, Gardea-Torresdey JL (2011) Interaction of nanoparticles with edible plants and their possible implications in the food chain. J Agric Food Chem 59(8):3485–3498. https://doi.org/10.1021/jf104517j
Roberts A, Haigler W (1990) Tracheary-element differentiation in suspension cultured cells of Zinnia requires uptake of extracellular Ca2+. Planta 180:502–509
Rolls GO, Farmer NJ, Hall JB (2012) Melbourne (Australia): Leica Biosystems Pty LTD. Artefacts in histological and cytological preparations, p 106. https://doi.org/10.13005/bpj/1014
Ruttkay-Nedecky B, Krystofova O, Nejdi L, Adam V (2017) Nanoparticles based on essential metals and their phytotoxicity. J Nanobiotechnol 15(1):33
Sadeghi R, Rodriguez RJ, Yao Y, Kokini JL (2017) Advances in nanotechnology as they pertain to food and agriculture: benefits and risks. Annu Rev Food Sci Technol 28(8):467–492. https://doi.org/10.1146/annurev-food-041715-033338
Sahu G, Saha S, Datta S, Chavan P, Naık S (2017) Methanolysis of Jatropha curcas oil using K2CO3/CaO as a solid base catalyst. Turk J Chem 41(6):845–861
Sergiev I, Alxieva V, Karanov E (1997) Effect of spermone, atrazine and combination between them on some endogenous protective systems and stress markers in plants. Comp Rend Acad Bulg Sc 51:121–124
Sharma P, Jha AB, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot 26:10–1155. https://doi.org/10.1155/2012/217037
Soundararajan P, Manivannan A, Cho YS, Jeong BR (2017) Exogenous supplementation of silicon improved the recovery of hyperhydric shoots in Dianthus caryophyllus L. by stabilizing the physiology and protein expression. Front Plant Sci 8:8–738. https://doi.org/10.3389/fpls.2017.00738
Sun D, Hussain HI, Yi Z, Siegele R, Cresswell T, Kong L, Cahill DM (2014) Uptake and cellular distribution, in four plant species, of fluorescently labeled mesoporous silica nanoparticles. Plant Cell Rep 33(8):1389–1402. https://doi.org/10.1007/s00299-014-1624-5
Tang H, Krishnakuma V, Bidwel S, Rosen BD, Krishnakumar V (2014) An improved genome release for the model legume Medicago truncatula. BMC Genomics 15(1):312
Toljamo A, Blonde D, Kärenlampi S, Kokko H (2016) Reprogramming of strawberry (Fragaria vesca) root transcriptome in response to Phytophthora cactorum. PLoS ONE 11:e0161078. https://doi.org/10.1371/journal.pone.0161078
Tuteja N, Mahajan S (2007) Calcium signaling network in plants. Plant Sig Behav 2(2):79–85. https://doi.org/10.4161/psb.2.2.4176
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidantsystems in acid rain-treated bean plants protective role of exogenous polyamines. Plant Sci 151:59–66. https://doi.org/10.1016/S0168-9452(99)00197-1
Xu J (2010) AtCPK6, a functionally redundant and positive regulator involved in salt/drought stress tolerance in Arabidopsis. Planta 231(6):1251–1260. https://doi.org/10.4161/psb.20351
Yee Y, Tam NFY, Wong YS, Lu CY (2003) Growth and physiological responses of two mangrove species (Bruguiera gymnorrhiza and Kandelia candel) to water logging. Environ Exp Bot 49(3):209–221. https://doi.org/10.1016/S0098-8472(02)00071-0
Yu Y, Wang J, Zhu MI, Wei ZM (2008) Optimization of mature embryo-based high frequency callus induction and plant regeneration from elite wheat cultivars grown in China. Plant Breed 17:249–255. https://doi.org/10.1111/j.1439-0523.2007.01461.x
Zhu JK (2001) Plant salt tolerance. Trends Plant Sci 6(2):66–71. https://doi.org/10.1016/s1360-1385(00)01838-0
Zhu JK (2016) Abiotic stress signaling and responses in plants. Cell 167(2):313–324. https://doi.org/10.1016/j.cell.2016.08.029
Author information
Authors and Affiliations
Contributions
Concept—IB; Design—IB and BY; Supervision—IB; Resource—IB; Materials—BY, FB, HN and AA; Data Collection and/or Processing—BY and FB; Analysis and/or Interpretation—BY, FB and SG; Literature Search—BY and FB; Writing—IB; Critical Reviews—HN and AA.
Corresponding author
Ethics declarations
Confict of interest
The authors have declared that no conflict of interests exists.
Additional information
Communicated by Günther Hahne.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yazıcılar, B., Böke, F., Alaylı, A. et al. In vitro effects of CaO nanoparticles on Triticale callus exposed to short and long-term salt stress. Plant Cell Rep 40, 29–42 (2021). https://doi.org/10.1007/s00299-020-02613-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-020-02613-0