Abstract
Key message
We found that ApGGPPS1, ApGGPPS2, and ApGGPPS3 can convert IPP and DMAPP to GGPP. ApGGPPS2 is probably involved in andrographolide biosynthesis. ApGGPPS3 may be responsible for the synthesis of the cytosolic GGPP.
Abstract
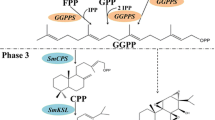
Andrographis paniculata is a traditional herb for the treatment of sore throat, flu, upper respiratory tract infections and other disorders. In A. paniculata, GGPP is not only the precursor of andrographolide and its primary bioactive compounds, but also the precursor of chlorophylls, carotenoids, gibberellins, and abscisic acid, which are the biomolecules of photosynthesis, growth regulation and other physiological and ecological processes. In this study, four cDNAs (named ApGGPPS1, ApGGPPS2, ApGGPPS3 and ApGGPPS4) encoding geranylgeranyl pyrophosphate synthases from A. paniculata were putatively isolated. Bioinformatic and phylogenetic analyses suggested that these ApGGPPS are highly similar to the geranylgeranyl pyrophosphate synthases in other plants. Prokaryotic expression showed that ApGGPPS1, ApGGPPS2 and ApGGPPS3 could convert IPP and DMAPP to GGPP, although ApGGPPS4 lacks a similar function. The expression of ApGGPPS2 was similar as ApCPS2 under MeJA treatment, ApCPS2 involved in the biosynthesis pathway of andrographolide (Shen et al., Biotechnol Lett 38:131–137, 2016a), has been proven through Virus-induced Gene Siliencing (VIGS) (Shen et al., Acta Bot Boreal 36:17–22, 2016b), and the subcellular localization of ApGGPPS2 was shown to localize in the plastid, suggested that ApGGPPS2 could be the key synthase in the biosynthesis pathway of andrographolide. In addition, ApGGPPS3 was shown to localize in the cytoplasm, suggested that ApGGPPS3 may be responsible for the synthesis of cytosolic GGPP, which may participate in the synthesis of cytosolic oligoprenols as side chains to produce ubiquinone, dolichols or other isoprenoids, in the synthesis of polyisoprenoids, and in protein prenylation.







Similar content being viewed by others
References
Ashby MN, Kutsunai SY, Ackerman S et al (1992) COQ2 is a candidate for the structural gene encoding para-hydroxybenzoate: polyprenyltransferase. J Biol Chem 267:4128–4136
Beck G, Coman D, Herren E, Ruiz-Sola MA, Rodriguez-Concepcion M, Gruissem W, Vranova E (2013) Characterization of the GGPP synthase gene family in Arabidopsis thaliana. Plant Mol Biol 82:393–416
Bick JA, Lange BM (2003) Metabolic cross talk between cytosolic and plastidial pathways of isoprenoid biosynthesis: unidirectional transport of intermediates across the chloroplast envelope membrane. Arch Biochem Biophys 415:146–154
Bouvier F, Rahier A, Camara B (2005) Biogenesis, molecular regulation and function of plant isoprenoids. Prog Lipid Res 44:357–429
Chua LS (2014) Review on liver inflammation and antiinflammatory activity of Andrographis paniculata for hepatoprotection. Phytother Res: PTR 28:1589–1598
Cyr A, Wilderman PR, Determan M, Peters RJ (2007) A modular approach for facile biosynthesis of labdane-related diterpenes. J Am Chem Soc 129:6684–6685
Garg A, Agrawal L, Misra RC, Sharma S, Ghosh S (2015) Andrographis paniculata transcriptome provides molecular insights into tissue-specific accumulation of medicinal diterpenes. BMC Genom 16:659
Gershenzon J, Dudareva N (2007) The function of terpene natural products in the natural world. Nat Chem Biol 3:408–414
Hossain MS, Urbi Z (2014) Andrographis paniculata (Burm. f.) Wall. ex Nees: a review of ethnobotany, phytochemistry, and pharmacology. Sci World J 2014:274905
Huynh TN, Ren X (2017) Producing GST-Cbx7 Fusion Proteins from Escherichia coli. BioProtoc 7:2333
Lin HX, Wang J, Qi MD, Guo J, Rong QX, Tang JF, Wu Y, Ma XH, Huang LQ (2017) Molecular cloning and functional characterization of multiple NADPH-cytochrome P450 reductases from Andrographis paniculata. Int J Biol Macromol 102:208–217
Liu C, Sun Z, Shen S, Lin L, Li T, Tian B, Hua Y (2014) Identification and characterization of the geranylgeranyl diphosphate synthase in Deinococcus radiodurans. Lett Appl Microbiol 58:219–224
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Misra RC, Garg A, Roy S, Chanotiya CS, Vasudev PG, Ghosh S (2015) Involvement of an ent-copalyl diphosphate synthase in tissue-specific accumulation of specialized diterpenes in Andrographis paniculata. Plant Sci 240:50–64
Noike M, Katagiri T, Nakayama T, Koyama T, Nishino T, Hemmi H (2008) The product chain length determination mechanism of type II geranylgeranyl diphosphate synthase requires subunit interaction. FEBS J 275:3921–3933
Ohnuma S, Hirooka K, Hemmi H, Ishida C, Ohto C, Nishino T (1996) Conversion of product specificity of archaebacterial geranylgeranyl-diphosphate synthase. Identification of essential amino acid residues for chain length determination of prenyltransferase reaction. J Biol Chem 271:18831–18837
Okada K, Saito T, Nakagawa T, Kawamukai M, Kamiya Y (2000) Five geranylgeranyl diphosphate synthases expressed in different organs are localized into three subcellular compartments in Arabidopsis. Plant Physiol 122:1045–1056
Ruiz-Sola MA, Coman D, Beck G, Barja MV, Colinas M, Graf A, Welsch R, Rutimann P, Buhlmann P, Bigler L, Gruissem W, Rodriguez-Concepcion M, Vranova E (2016) Arabidopsis GERANYLGERANYL DIPHOSPHATE SYNTHASE 11 is a hub isozyme required for the production of most photosynthesis-related isoprenoids. New Phytol 209:252–264
Santner A, Calderon-Villalobos LIA, Estelle M (2009) Plant hormones are versatile chemical regulators of plant growth. Nat Chem Biol 5:301–307
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3:1101–1108
Shen QQ, Li L, Jiang Y, Wang Q (2016a) Functional characterization of ent-copalyl diphosphate synthase from Andrographis paniculata with putative involvement in andrographolides biosynthesis. Biotechnol Lett 38: 131–137
Shen QQ, Liu Q, Li CC, Fu YP, Wang Q (2016b) Functional characterization of ApCPS Involved in andrographolides biosynthesis by virus-induced gene silencing. Acta Bot Boreal 36:17–22
Song L, Poulter CD (1994) Yeast farnesyl-diphosphate synthase: site directed mutagenesis of residues in highly conserved prenyltransferase domains I and II. Proc Natl Acad Sci USA 91:3044–3048
Sparkes IA, Runions J, Kearns A et al (2006) Rapid, transient expression of fluorescent fusion proteins in tobacco plants and generation of stably transformed plants. Nat Protoc 1:2019–2025
Subramanian R, Zaini Asmawi M, Sadikun A (2012) A bitter plant with a sweet future? A comprehensive review of an oriental medicinal plant: Andrographis paniculata. Phytochem Rev 11:39–75
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucl Acids Res 22:4673–4680
Vandermoten S, Haubruge E, Cusson M (2009) New insights into short-chain prenyltransferases: structural features, evolutionary history and potential for selective inhibition. Cell Mol Life Sci 66:3685–3695
Vickers CE, Bongers M, Liu Q, Delatte T, Bouwmeester H (2014) Metabolic engineering of volatile isoprenoids in plants and microbes. Plant Cell Environ 37:1753–1775
Vranova E, Hirsch-Hoffmann M, Gruissem W (2011) AtIPD: a curated database of Arabidopsis isoprenoid pathway models and genes for isoprenoid network analysis. Plant Physiol 156:1655–1660
Wang K, Ohnuma S (1999) Chain-length determination mechanism of isoprenyl diphosphate synthases and implications for molecular evolution. Trends Biochem Sci 24:445–451
Wang C, Chen Q, Fan D, Li J, Wang G, Zhang P (2016) Structural analyses of short-chain prenyltransferases identify an evolutionarily conserved GFPPS Clade in brassicaceae plants. Mol Plant 9:195–204
Zhang M, Su P, Zhou YJ et al (2015) Identification of geranylgeranyl diphosphate synthase genes from Tripterygium wilfordii. Plant Cell Rep 34:2179–2188
Zhu XF, Suzuki K, Okada K, Tanaka K, Nakagawa T, Kawamukai M, Matsuda H (1997) Cloning and functional expression of a novel geranylgeranyl pyrophosphate synthase gene from Arabidopsis thaliana in Escherichia coli. Plant Cell Physiol 38:357–361
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant Nos. 81673547; 81773830), High-level Teachers in Beijing Municipal Universities in the Period of 13th Five-year Plan (Grant No. CIT&TCD20170324), the key project at central government level: The ability establishment of sustainable use for valuable Chinese medicine resources (Grant No. 2060302) and National Program for Special Support of Eminent Professionals.
Author information
Authors and Affiliations
Contributions
L.H and W.G designed the project; J.W performed the research, analyzed the data and wrote the paper; P.S, H.L, T.C and J.G, participated in the research. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Communicated by Salim Al-Babili.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, J., Lin, HX., Su, P. et al. Molecular cloning and functional characterization of multiple geranylgeranyl pyrophosphate synthases (ApGGPPS) from Andrographis paniculata. Plant Cell Rep 38, 117–128 (2019). https://doi.org/10.1007/s00299-018-2353-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-018-2353-y