Abstract
Key message
Using Illumina sequencing technology, we have generated the large-scale transcriptome sequencing data containing abundant information on genes involved in the metabolic pathways in R. idaeus cv. Nova fruits.
Abstract
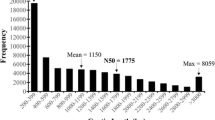
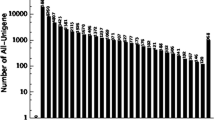
Rubus idaeus (Red raspberry) is one of the important economical crops that possess numerous nutrients, micronutrients and phytochemicals with essential health benefits to human. The molecular mechanism underlying the ripening process and phytochemical biosynthesis in red raspberry is attributed to the changes in gene expression, but very limited transcriptomic and genomic information in public databases is available. To address this issue, we generated more than 51 million sequencing reads from R. idaeus cv. Nova fruit using Illumina RNA-Seq technology. After de novo assembly, we obtained 42,604 unigenes with an average length of 812 bp. At the protein level, Nova fruit transcriptome showed 77 and 68 % sequence similarities with Rubus coreanus and Fragaria versa, respectively, indicating the evolutionary relationship between them. In addition, 69 % of assembled unigenes were annotated using public databases including NCBI non-redundant, Cluster of Orthologous Groups and Gene ontology database, suggesting that our transcriptome dataset provides a valuable resource for investigating metabolic processes in red raspberry. To analyze the relationship between several novel transcripts and the amounts of metabolites such as γ-aminobutyric acid and anthocyanins, real-time PCR and target metabolite analysis were performed on two different ripening stages of Nova. This is the first attempt using Illumina sequencing platform for RNA sequencing and de novo assembly of Nova fruit without reference genome. Our data provide the most comprehensive transcriptome resource available for Rubus fruits, and will be useful for understanding the ripening process and for breeding R. idaeus cultivars with improved fruit quality.



Similar content being viewed by others
References
Aanes H, Winata CL, Lin CH, Chen JP, Srinivasan KG, Lee SG, Lim AY, Hajan HS, Collas P, Bourque G, Gong Z, Korzh V, Mathavan S (2011) Zebrafish mRNA sequencing deciphers novelties in transcriptome dynamics during maternal to zygotic transition. Genome Res 21:1328–1338
Abdou AM, Higashiguchi S, Horie K, Kim M, Hatta H, Yokogoshi H (2006) Relaxation and immunity enhancement effects of gamma-aminobutyric acid (GABA) administration in humans. BioFactors 26:201–208
Ageta-Ishihara N, Takemoto-Kimura S, Nonaka M, Adachi-Morishima A, Suzuki K, Kamijo S, Fujii H, Mano T, Blaeser F, Chatila TA, Mizuno H, Hirano T, Tagawa Y, Okuno H, Bito H (2009) Control of cortical axon elongation by a GABA-driven Ca2+/calmodulin-dependent protein kinase cascade. J Neurosci 29:13720–13729
Akihiro T, Koike S, Tani R, Tominaga T, Watanabe S, Iijima Y, Aoki K, Shibata D, Ashihara H, Matsukura C, Akama K, Fujimura T, Ezura H (2008) Biochemical mechanism on GABA accumulation during fruit development in tomato. Plant Cell Physiol 49:1378–1389
Alexander L, Grierson D (2002) Ethylene biosynthesis and action in tomato: a model for climacteric fruit ripening. J Exp Bot 53:2039–2055
Anttonen MJ, Karjalainen RO (2005) Environmental and genetic variation of phenolic compounds in red raspberry. J Food Compost Anal 18:759–769
Beekwilder J, Jonker H, Meesters P, Hall RD, van der Meer IM, de Vos RCH (2005) Antioxidants in raspberry: on-line analysis links antioxidant activity to a diversity of individual metabolites. J Agric Food Chem 53:3313–3320
Beuve N, Rispail N, Laine P, Cliquet JB, Ourry A, Le Deunff E (2004) Putative role of γ-aminobutyric acid (GABA) as a long-distance signal in up-regulation of nitrate uptake in Brassica napus L. Plant Cell Environ 27:1035–1046
Bouche N, Fromm H (2004) GABA in plants: just a metabolite? Trends Plant Sci 9:110–115
Bradish CM, Perkins-Veazie P, Fernandez GE, Xie G, Jia W (2012) Comparison of flavonoid composition of red raspberries (Rubus idaeus L.) grown in the Southern United States. J Agric Food Chem 60:5779–5786
Cercós M, Soler G, Iglesias DJ, Gadea J, Forment J, Talón M (2006) Global analysis of gene expression during development and ripening of citrus fruit flesh. A proposed mechanism for citric acid utilization. Plant Mol Biol 62:513–527
de Ancos B, Gonzales EM, Cano MP (2000) Ellagic acid, vitamin C, and total phenolic contents and radical scavenging capacity affected by freezing and frozen storage in raspberry fruit. J Agric Food Chem 48:4565–4570
Deighton N, Brennan R, Finn C, Davies HV (2000) Antioxidant properties of domesticated and wild Rubus species. J Sci Food Agric 80:1307–1313
Dincheva I, Badjakov I, Kondakova V, Batchvarova R (2013) Metabolic profiling of red raspberry (Rubus idaeus) during fruit development and ripening. Int J Agric Sci Res 3:81–88
Dobson P, Graham J, Stewart D, Brennan R, Hackett CA, McDougall GJ (2012) Over-seasons analysis of quantitative trait loci affecting phenolic content and antioxidant capacity in raspberry. J Agric Food Chem 60:5360–5366
Fortes AM, Agudelo-Romero P, Silva MS, Ali K, Sousa L, Maltese F, Choi YH, Grimplet J, Martinez-Zapater JM, Verpoorte R, Pais MS (2011) Transcript and metabolite analysis in Trincadeira cultivar reveals novel information regarding the dynamics of grape ripening. BMC Plant Biol 11:149
Grabherr MG, Haas BJ, Yassour M, Levin JZ, Thompson DA, Amit I, Adiconis X, Fan L, Raychowdhury R, Zeng Q, Chen Z, Mauceli E, Hacohen N, Gnirke A, Rhind N, di Palma F, Birren BW, Nusbaum C, Lindblad-Toh K, Friedman N, Regev A (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644–652
Grotewold E (2006) The genetics and biochemistry of floral pigments. Annu Rev Plant Physiol 57:761–780
Hanson E, Berkheimer S, Schilder A, Isaacs R, Kravchenko S (2005) Raspberry variety performance in Southern Michigan. HortTechnology 15:716–721
Hyun TK, Rim Y, Jang HJ, Kim CH, Park J, Kumar R, Lee S, Kim BC, Bhak J, Nguyen-Quoc B, Kim SW, Lee SY, Kim JY (2012) De novo transcriptome sequencing of Momordica cochinchinensis to identify genes involved in the carotenoid biosynthesis. Plant Mol Biol 79:413–427
Hyun TK, Eom SH, Jeun YC, Han SH, Kim J-S (2013a) Identification of glutamate decarboxylases as a γ-aminobutyric acid (GABA) biosynthetic enzyme in soybean. Ind Crops Prod 49:864–870
Hyun TK, Kumar D, Cho YY, Hyun HN, Kim JS (2013b) Computational identification and phylogenetic analysis of the oil-body structural proteins, oleosin and caleosin, in castor bean and flax. Gene 515:454–460
Hyun TK, Lee S, Rim Y, Kumar R, Han X, Lee SY, Lee CH, Kim JY (2014) De novo RNA sequencing and metabolite profiling to identify genes involved in anthocyanin biosynthesis in Korean black raspberry (Rubus coreanus Miquel). PLoS ONE 9:e88292
Jaakola L, Määttä K, Pirttilä AM, Törrönen R, Kärenlampi S, Hohtola A (2002) Expression of genes involved in anthocyanin biosynthesis in relation to anthocyanin, proanthocyanidin, and flavonol levels during bilberry fruit development. Plant Physiol 130:729–739
Jia HF, Chai YM, Li CL, Lu D, Luo JJ, Qin L, Shen YY (2011) Abscisic acid plays an important role in the regulation of strawberry fruit ripening. Plant Physiol 157:88–199
Kahkonen MP, Hopia AI, Heinonen M (2001) Berry phenolics and their antioxidant activity. J Agric Food Chem 49:4076–4082
Kassim A, Poette J, Paterson A, Zait D, McCallum S, Woodhead M, Smith K, Hackett C, Graham J (2009) Environmental and seasonal influences on red raspberry anthocyanin antioxidant contents and identification of quantitative traits loci (QTL). Mol Nutr Food Res 53:625–634
Kim JY, Lee MY, Ji GE, Lee YS, Hwang KT (2009) Production of gamma-aminobutyric acid in black raspberry juice during fermentation by Lactobacillus brevis GABA100. Int J Food Microbiol 130:12–16
Lancien M, Roberts MR (2006) Regulation of Arabidopsis thaliana 14-3-3 gene expression by γ-aminobutyric acid. Plant Cell Environ 29:1430–1436
Li L, Ban ZJ, Li XH, Wu MY, Wang AL, Jiang YQ, Jiang YH (2012) Differential expression of anthocyanin biosynthetic genes and transcription factor PcMYB10 in pears (Pyrus communis L.). PLoS ONE 7:e46070
Lijavetzky D, Carbonell-Bejerano P, Grimplet J, Bravo G, Flores P, Fenoll J, Hellín P, Oliveros JC, Martínez-Zapater JM (2012) Berry flesh and skin ripening features in Vitis vinifera as assessed by transcriptional profiling. PLoS ONE 7:e39547
Lin-Wang K, Bolitho K, Grafton K, Kortstee A, Karunairetnam S, McGhie TK, Espley RV, Hellens RP, Allan AC (2010) An R2R3 MYB transcription factor associated with regulation of the anthocyanin biosynthetic pathway in Rosaceae. BMC Plant Biol 10:50
Logacheva MD, Kasianov AS, Vinogrado DV, Samigullin TH, Gelfand MS, Makeev VJ, Penin AA (2011) De novo sequencing and characterization of floral transcriptome in two species of buckwheat (Fagopyrum). BMC Genom 12:30
McDougall GJ, Martinussen I, Stewart D (2008) Towards fruitful metabolomics: high throughput analyses of polyphenol composition in berries using direct infusion mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 871:362–369
Mullen W, McGinn J, Lean MEJ, MacLean MR, Gardner P, Duthie GG, Yakota T, Crozier A (2002) Ellagitannins, flavonoids, and other phenolics in red raspberries and their contribution to antioxidant capacity and vasorelaxation properties. J Agric Food Chem 50:5191–5196
Niu SS, Xu CJ, Zhang WS, Zhang B, Li X, Lin-Wang K, Ferguson IB, Allan AC, Chen KS (2010) Coordinated regulation of anthocyanin biosynthesis in Chinese bayberry (Myrica rubra) fruit by a R2R3 MYB transcription factor. Planta 231:887–899
Oláh S, Füle M, Komlósi G, Varga C, Báldi R, Barzó P, Tamás G (2009) Regulation of cortical microcircuits by unitary GABA-mediated volume transmission. Nature 461:1278–1281
Palapol Y, Ketsa S, Lin-Wang K, Ferguson IB, Allan AC (2009) A MYB transcription factor regulates anthocyanin biosynthesis in mangosteen (Garcinia mangostana L.) fruit during ripening. Planta 229:1323–1334
Potter D, Eriksson T, Evans RC, Oh S, Smedmark JEE, Morgan DR, Kerr M, Robertson KR, Arsenault M, Dickinson TA, Campbell CS (2007) Phylogeny and classification of Rosaceae. Plant Syst Evol 266:5–43
Rolin D, Baldet P, Just D, Chevalier C, Biran M, Raymond P (2000) NMR study of low subcellular pH during the development of cherry tomato fruit. Aust J Plant Physiol 27:61–69
Rosel TD, Hung LH, Medenbach J, Donde K, Starke S, Benes V, Ratsch G, Bindereif A (2011) RNA-seq analysis in mutant zebrafish reveals role of U1Cprotein in alternative splicing regulation. EMBO J 30:1965–1976
Ross HA, McDougall GJ, Stewart D (2007) Antiproliferative activity is pedominantly associated with ellagitannins in raspberry extracts. Phytochemistry 68:218–228
Shelp BJ, Bown AW, McLean MD (1999) Metabolism and functions of gamma-aminobutyric acid. Trends Plant Sci 4:446–452
Shelp BJ, Bozzo GG, Trobacher CP, Zarei A, Deyman KL, Brikis CJ (2012) Hypothesis/review: contribution of putrescine to 4-aminobutyrate (GABA) production in response to abiotic stress. Plant Sci 193–194:130–135
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary and genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Vesterlund L, Jiao H, Unneberg P, Hovatta O, Kere J (2011) The zebrafish transcriptome during early development. BMC Dev Biol 11:30
Ward JA, Weber CA (2012) Comparative RNA-Seq for the investigation of resistance to Phytophthora root rot in the red raspberry Latham. Acta Hortic 946:67–72
Yousefi G, Yousefi S, Emam-Djomeh Z (2013) A comparative study on different concentration methods of extracts obtained from two raspberries (Rubus idaeus L.) cultivars: evaluation of anthocyanins and phenolics contents and antioxidant activity. Int J Food Sci Technol 48:1179–1186
Zifkin M, Jin A, Ozga JA, Zaharia LI, Schernthaner JP, Gesell A, Abrams SR, Kennedy JA, Constabel CP (2012) Gene expression and metabolite profiling of developing highbush blueberry fruit indicates transcriptional regulation of flavonoid metabolism and activation of abscisic acid metabolism. Plant Physiol 158:200–224
Acknowledgments
We thank Dr. Pill-Jae Park (Gochang Black Raspberry Research Institute) for supporting plant materials. This work was supported by Science and Technology and a Grant from the Next-Generation BioGreen 21 Program (SSAC Grant PJ009495; PJ00952004), Rural Development Administration, Republic of Korea.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by Prakash Lakshmanan.
T. K. Hyun and S. Lee have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
299_2014_1641_MOESM2_ESM.tif
Gene ontology classification of assembled unigenes. The results are summarized in three main categories: Biological process, Cellular component and Molecular function (TIFF 1240 kb)
299_2014_1641_MOESM3_ESM.tif
Histogram presentation of clusters of orthologous groups (COG) classification. All unigenes were aligned to COG database to predict and classify possible functions. (TIFF 1941 kb)
299_2014_1641_MOESM5_ESM.xls
Top BLAST hits from NCBI nr database. BLAST results against the NCBI nr database for all the distinct sequences (XLS 8580 kb)
Rights and permissions
About this article
Cite this article
Hyun, T.K., Lee, S., Kumar, D. et al. RNA-seq analysis of Rubus idaeus cv. Nova: transcriptome sequencing and de novo assembly for subsequent functional genomics approaches. Plant Cell Rep 33, 1617–1628 (2014). https://doi.org/10.1007/s00299-014-1641-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-014-1641-4