Abstract
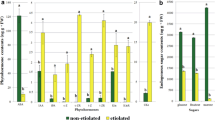
Embryogenic callus and suspension cultures of carrot (Daucus carota L., cv. Nantaise), growing on/in medium including 1 mg/l 2,4-dichlorophenoxy acetic acid (2,4-D), were transferred to medium with or without this plant growth regulator, to impair or induce, respectively, further development of somatic embryos. The endogenous hormone levels of the cultures were determined over 7 days by means of radio-immunoassay, to characterize their evolution in the initial stages of embryo development. In general, levels of indoleacetic acid (IAA) and abscisic acid (ABA) showed only short-lived differences among treatments during this time in both types of tissue analyzed (i.e., a peak of IAA in callus cultures in the absence of 2,4-D, 48 h after medium change, and higher ABA contents 144 h after subculture of suspension cultures in the presence of 2,4-D). Gibberellins (1, 3 and 20) were detected only in suspension cultures devoid of 2,4-D, starting 24 h after subculture. Concerning the evaluated cytokinins—zeatin/zeatin riboside and N6(Δ2-isopentenyl) adenine/N6(Δ2-isopentenyl) adenosine—the most remarkable observation is that high levels of the former generally coincided with low concentrations of the latter, indicating a shift from precursor to the active form, and vice versa.


Similar content being viewed by others
References
Bertling I, Bangerth F (1995) Changes in hormonal pattern of the new growth of Sclerocarya birrea after rejuvenation treatment with GA3 and ‘heading back’. Gartenbauwissenschaft 60:119–124
Centeno ML, Rodríguez R, Berros B, Rodríguez A (1997) Endogenous hormonal content and somatic embryogenic capacity of Corylus avellana L. cotyledons. Plant Cell Rep 17:139–144. DOI 10.1007/s002990050366
Chen C-M (1997) Cytokinin biosynthesis and interconversion. Physiol Plant 101:665–673. DOI 10.1034/j.1399-3054.1997.1010402.x
Dodeman VL, Ducreux G (1996) Total protein pattern expression during induction and development of carrot somatic embryos. Plant Sci 120:57–69. DOI 10.1016/S0168-9452(96)04487-1
Einset JW (1986) Zeatin biosynthesis from N6-(Δ2-isopentenyl) adenine in Actinidia and other woody plants. Proc Natl Acad Sci USA 83:972–975
Fehér A, Pasternak TP, Dudits D (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tissue Organ Cult 74:201–228. DOI 10.1023/A:1024033216561
Fujii N (1997) Pattern of DNA binding of nuclear proteins to the proximal Agrobacterium rhizogenes rolC promoter is altered during somatic embryogenesis of carrot. Gene 201:55–62. DOI 10.1016/S0378-1119(97)00427-7
Fujimura T, Komamine A (1979) Synchronization of somatic embryogenesis in a carrot suspension culture. Plant Physiol 64:162–164
Fujimura T, Komamine A (1980) Mode of action of 2,4-D and zeatin on somatic embryogenesis in a carrot cell suspension culture. Z Pflanzenphysiol 99:1–8
Giuliano G, Rosellini D, Terzi M (1983) A new method for purification of the different stages of carrot embryoids. Plant Cell Rep 2:216–218
Grieb B, Schäfer F, Imani J, Mashayekhi KN, Arnholdt-Schmitt B, Neumann KH (1997) Changes in soluble proteins and phytohormone concentrations of cultured carrot petiole explants during induction of somatic embryogenesis (Daucus carota L.). J Appl Bot 71:94–103
Guzzo F, Baldan B, Levi M, Sparvoli E, Lo Schiavo F, Terzi M, Mariani P (1995) Early cellular events during induction of carrot explants with 2,4-D. Protoplasma 185:28–36
Guzzo F, Cantamessa K, Portaluppi P, Levi M (2002) Flow cytometry and sorting of protoplasts from carrot cell cultures reveal two cell subpopulations with different morphogenetic potential. Plant Cell Rep 21:214–219. DOI 10.1007/s00299-002-0519-z
Hatzopoulos P, Fong F, Sung ZR (1990) Abscisic acid regulation of DC8, a carrot embryogenic gene. Plant Physiol 94:690–695
Jiménez VM, Bangerth F (2001) Endogenous hormone levels in explants and in embryogenic and non-embryogenic cultures of carrot. Physiol Plant 111:389–395. DOI 10.1034/j.1399-3054.2001.1110317.x
Jiménez VM, Guevara E, Herrera J, Bangerth F (2001) Endogenous hormone levels in habituated nucellar Citrus callus during the initial stages of regeneration. Plant Cell Rep 20:92–100. DOI 10.1007/s002990000280
Kamada H, Harada H (1981) Changes in the endogenous level and effects of abscisic acid during somatic embryogenesis of Daucus carota L. Plant Cell Physiol 22:1423–1429
Kitamiya E, Suzuki S, Sano T, Nagata T (2000) Isolation of two genes that were induced upon the initiation of somatic embryogenesis on carrot hypocotyls by high concentrations of 2,4-D. Plant Cell Rep 19:551–557. DOI 10.1007/s002990050772
Kiyosue T, Nakajima M, Yamaguchi I, Satoh S, Kamada H, Harada H (1992) Endogenous levels of abscisic acid in embryogenic cells, non-embryogenic cells and somatic embryos of carrot (Daucus carota L.). Biochem Physiol Pflanzen 188:343–347
Kiyosue T, Satoh S, Kamada H, Harada H (1993) Somatic embryogenesis in higher plants. J Plant Res 3(Special Issue):75–82
Komamine A, Kawahara R, Matsumoto M, Sunabori S, Toya T, Fujiwara A, Tsukuhara M, Smith J, Ito M, Fukuda H, Nomura K, Fujimura T (1992) Mechanisms of somatic embryogenesis in cell cultures: physiology, biochemistry, and molecular biology. In Vitro Cell Dev Biol Plant 28P:11–14
Michalczuk L, Cooke TJ, Cohen JD (1992a) Auxin levels at different stages of carrot embryogenesis. Phytochemistry 31:1097–1103. DOI 10.1016/0031-9422(92)80241-6
Michalczuk L, Ribnicky DM, Cooke TJ, Cohen JD (1992b) Regulation of indole-3-acetic acid biosynthetic pathways in carrot cell cultures. Plant Physiol 100:1346–1353
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Nagamori E, Omote M, Honda H, Kobayashi T (2001) Enhanced and prolonged production of plantlets regenerated from carrot callus in a viscous additive-supplemented medium. J Biosci Bioeng 91:283–287. DOI 10.1016/S1389-1723(01)80135-3
Noma M, Huber J, Ernst D, Pharis RP (1982) Quantitation of gibberellins and the metabolism of [3H]gibberellin A1, during somatic embryogenesis in carrot and anise cell cultures. Planta 155:369–376
Nomura K, Komamine A (1985) Identification and isolation of single cells that produce somatic embryos at a high frequency in a carrot suspension culture. Plant Physiol 79:988–991
Nuti-Ronchi V, Giorgetti L (1995) The cell’s commitment to somatic embryogenesis. In: Bajaj YPS (ed) Somatic embryogenesis and synthetic seed. I. Biotechnology in agriculture and forestry, vol 30. Springer, Berlin Heidelberg New York, pp 3–19
Osuga K, Masuda H, Komamine A (1999) Synchronization of somatic embryogenesis at high frequency using carrot suspension cultures: model systems and application in plant development. Methods Cell Sci 21:129–140. DOI 10.1023/A:1009884806166
Ribnicky DM, Ilic N, Cohen JD, Cooke TJ (1996) The effects of exogenous auxins on endogenous indole-3-acetic acid metabolism. Plant Physiol 112:549–558
Ribnicky DM, Cohen JD, Hu W-S, Cooke TJ (2002) An auxin surge following fertilization in carrots: a mechanism for regulating plant totipotency. Planta 214:505–509. DOI 10.1007/s004250100639
Ross JJ (1998) Effects of auxin transport inhibitors on gibberellins in pea. J Plant Growth Regul 17:141–146
Sasaki K, Shimomura K, Kamada H, Harada H (1994) IAA metabolism in embryogenic and non-embryogenic carrot cells. Plant Cell Physiol 35:1159–1164
Sato-Nara K, Fukuda H (2000) The rates of deceleration of nuclear and organellar DNA syntheses differ in the progenitor cells of the apical meristems during carrot somatic embryogenesis. Planta 211:457–466. DOI 10.1007/s004250000317
Schiavone FM, Cooke TJ (1985) A geometric analysis of somatic embryo formation in carrot cell cultures. Can J Bot 63:1573–1578
Schrader S, Kaldenhoff R, Richter G (1997) Expression of novel genes during somatic embryogenesis of suspension-cultured carrot cells (Daucus carota). J Plant Physiol 150:63–68
Seitz HU, Seitz U, Alfermann W (1985) Pflanzliche Gewebekultur: ein Praktikum. Gustav Fischer, Stuttgart, p 114
Sharma AK (1999) Synchronization in plant cells—an introduction. Methods Cell Sci 21:73–78. DOI 10.1023/A:1009828419370
Stasolla C, Loukanina N, Ashihara H, Yeung EC, Thorpe TA (2003) Changes in deoxyribonucleotide biosynthesis during carrot somatic embryogenesis. Plant Physiol Biochem 41:779–785. DOI 10.1016/S0981-9428(03)00122-0
Tokuji Y, Kuriyama K (2003) Involvement of gibberellin and cytokinin in the formation of embryogenic cell clumps in carrot (Daucus carota). J Plant Physiol 160:133–141. DOI 10.1078/0176-1617-00892
Vu JCV, Niedz RP, Yelenosky G (1993) Glycerol stimulation of chlorophyll synthesis, embryogenesis and carboxylation and sucrose metabolism enzymes in nucellar callus of ‘Hamlin’ sweet orange. Plant Cell Tissue Organ Cult 33:75–80
Acknowledgements
The authors thank the German Academic Exchange Service (DAAD) for financial support in the form of short-term scholarships. The skilled work of A. Azofeifa, E. Tavares and S. Masís, conducting part of the in vitro culture, is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by D. Dudits
Rights and permissions
About this article
Cite this article
Jiménez, V.M., Guevara, E., Herrera, J. et al. Evolution of endogenous hormone concentration in embryogenic cultures of carrot during early expression of somatic embryogenesis. Plant Cell Rep 23, 567–572 (2005). https://doi.org/10.1007/s00299-004-0869-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-004-0869-9