Abstract
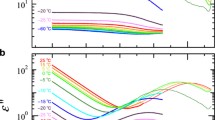
1H NMR spectroscopy was used to investigate the temperature-induced phase transitions in aqueous solutions of poly(N-isopropylmethacrylamide)/poly(N-vinylcaprolactam) (PIPMAm/PVCL) mixtures to find out if the phase transition of the given component (PIPMAm or PVCL) is affected by the presence of the second component. Our results that PVCL and PIPMAm transitions are in polymer mixtures shifted by ~2 K towards higher temperatures in comparison with neat polymers and depend on polymer concentration show that such effect exists. Spin–spin relaxation times of water (HDO) indicate that in solutions with c ≥ 1 wt% a portion of water is predominantly bound in PVCL mesoglobules even at temperatures above the LCST transition of PIPMAm component. Water is with time released from these mesoglobules without any induction period so indicating that it is mostly indirectly bound water. We assume that there is a direct connection between character of the bound water and the transition temperatures.










Similar content being viewed by others
References
Schild HG (1992) Poly(N-isopropylacrylamide): experiment, theory and application. Prog Polym Sci 17:163–249
Aseyev VO, Tenhu H, Winnik FM (2006) Temperature dependence of the colloidal stability of neutral amphiphilic polymers in water. Adv Polym Sci 196:1–85
Liu R, Fraylich M, Saunders BR (2009) Thermoresponsive copolymers: from fundamental studies to applications. Colloid Polym Sci 287:627–643
Fujishige S, Kubota K, Ando I (1989) Phase transition of aqueous solutions of poly(N-isopropylacrylamide) and poly(N-isopropylmethacrylamide). J Phys Chem 93:3311–3313
Cherkasskaya OV, Anufrieva VV, Krakoviak MG, Ananeva TD, Lushchik VB (1992) Intramolecular structure formation and migration of the energy of electron excitement groups of macromolecules in solution. Vysokomol Soedin A 34(10):90–98
Tiktopulo EI, Uversky VN, Lushchik VB, Klenin SL, Bychkova VE, Ptitsyn OB (1995) “Domain” coil- globule transition in homopolymers. Macromolecules 28:7519–7524
Netopilík M, Bohdanecký M, Chytrý V, Ulbrich K (1997) Cloud point of poly(N-isopropylmethacrylamide) solutions in water: is it really a point? Macromol Rapid Commun 18:107–111
Maeda Y, Nakamura T, Ikeda I (2001) Changes in the hydration states of poly(N-n-propylmethacrylamide) and poly(N-isopropylmethacrylamide) during their phase transitions in water observed by FTIR spectroscopy. Macromolecules 34:8246–8251
Kirsh YE, Yanul NA, Popkov YM (2002) Temperature behavior of thermo-responsive poly-N-vinylcaprolactam and poly-N-isopropylmethacrylamide in aqueous solutions involving organic solutes. Eur Polym J 38:403–406
Salmerón Sánchez M, Hanyková L, Ilavský M, Monleón Pradas M (2004) Thermal transitions of poly(N-isopropylmethacrylamide) in aqueous solutions. Polymer 45:4087–4094
Wu Y, Meersman F, Ozaki Y (2006) A novel application of hybrid two-dimensional correlation infrared spectroscopy: exploration of the reversibility of the pressure- and temperature-induced phase separation of poly(N-isopropylacrylamide) and poly(N-isopropylmethacrylamide) in aqueous solution. Macromolecules 39:1182–1188
Schmidt P, Dybal J, Trchová M (2006) Investigations of the hydrophobic and hydrophilic interactions in polymer-water systems by ATR FTIR and Raman spectroscopy. Vib Spectrosc 42:278–283
Dybal J, Trchová M, Schmidt P (2009) The role of water in structural changes of poly(N-isopropylacrylamide) and poly(N-isopropylmethacrylamide) studied by FTIR, Raman spectroscopy and quantum chemical calculations. Vib Spectrosc 51:44–51
Kano M, Kokufuta E (2009) On the temperature-responsive polymers and gels based on N-propylacrylamides and N-propylmethacrylamides. Langmuir 25:8649–8655
Tang Y (2010) Collapse kinetics for individual poly(N-isopropylmethacrylamide) chains. Polymer 51:897–901
Zhang LP, Noda I, Wu YQ (2010) An application of concatenated 2D correlation spectroscopy: exploration of the reversibility of the temperature-induced hydration variation of poly(N-isopropylmethacrylamide) in aqueous solution. J Mol Struct 974:80–87
Djokpé E, Vogt W (2001) N-isopropylacrylamide and N-isopropylmethacrylamide: cloud points of mixtures and copolymers. Macromol Chem Phys 202:750–757
Tang YC, Ding YW, Zhang G (2008) Role of methyl in the phase transition of poly(N-isopropylmethacrylamide). J Phys Chem B 112:8447–8451
Lebedev VT, Torok G, Cser L, Kali G, Kirsh YE, Sibilev AI, Orlova DN (2001) NSE-study of poly(N-vinylcaprolactam) by coil-globule transition. Physica B 287:50–54
Maeda Y, Nakamura T, Ikeda I (2002) Hydration and phase behavior of poly(N-vinylcaprolactam) and poly(N-vinylpyrrolidone) in water. Macromolecules 35:217–222
Lebedev V, Torok G, Cser L, Treimer W, Orlova D, Sibilev A (2003) Polymer hydration and microphase decomposition in poly(N-vinylcaprolactam)water complex. J Appl Cryst 36:967–969
Van Durme K, Verbrugghe S, Du Prez FE, Van Mele B (2004) Influence of poly(ethylene oxide) grafts on kinetics of LCST behavior in aqueous poly(N-vinylcaprolactam) solutions and networks studied by modulated temperature DSC. Macromolecules 37:1054–1061
Laukkanen A, Valtola L, Winnik FM, Tenhu H (2004) Formation of colloidally stable phase separated poly(N-vinylcaprolactam) in water: a study by dynamic light scattering, microcalorimetry, and pressure perturbation calorimetry. Macromolecules 37:2268–2274
Aseyev V, Hietala S, Laukkanen A, Nuopponen M, Confortini O, Du Prez FE, Tenhu H (2005) Mesoglobules of thermoresponsive polymers in dilute aqueous solutions above the LCST. Polymer 46:7118–7131
Dubovik AS, Makhaeva EE, Grinberg VY, Khokhlov AR (2005) Energetics of cooperative transitions of N-vinylcaprolactam polymers in aqueous solutions. Macromol Chem Phys 206:915–928
Chee CK, Rimmer S, Soutar I, Swanson L (2006) Fluorescence investigations of the conformational behaviour of poly(N-vinylcaprolactam). React Funct Polym 66:1–11
Vorobev MM, Burova TV, Grinberg NV, Dubovik AS, Falcev NG, Lozinsky VI (2010) Hydration characterization of N-vinylcaprolactam polymers by absorption millimeter-wave measurements. Colloid Polym Sci 288:1457–1463
Sun S, Wu P (2011) Infrared spectroscopic insight into hydration behavior of poly(N-vinylcaprolactam) in water. J Phys Chem B 115:11609–11618
Spěváček J (2009) NMR investigations of phase transition in aqueous polymer solutions and gels. Curr Opin Colloid Interface Sci 14:184–191
Starovoytova L, Spěváček J, Hanyková L, Ilavský M (2003) 1H NMR study of phase transition of uncharged and negatively charged poly(N-isopropylmethacrylamide) in D2O solutions. Macromol Symp 203:239–246
Starovoytova L, Spěváček J, Hanyková L, Ilavský M (2004) 1H NMR study of thermotropic phase transitions in D2O solutions of poly(N-isopropylmethacrylamide)/poly(vinyl methyl ether) mixtures. Polymer 45:5905–5911
Starovoytova L, Spěváček J (2006) Effect of time on the hydration and temperature-induced phase separation in aqueous polymer solutions. 1H NMR study. Polymer 47:7329–7334
Starovoytova L, Spěváček J, Trchová M (2007) 1H NMR and IR study of temperature-induced phase transition of negatively charged poly(N-isopropylmethacrylamide-co-sodium methacrylate) copolymers in aqueous solutions. Eur Polym J 43:5001–5009
Spěváček J, Starovoytova L, Hanyková L, Kouřilová H (2008) Polymer-solvent interactions in solutions of thermoresponsive polymers studied by NMR and IR spectroscopy. Macromol Symp 273:17–24
Kouřilová H, Hanyková L, Spěváček J (2009) NMR study of phase separation in D2O/ethanol solutions of poly(N-isopropylmethacrylamide) induced by solvent composition and temperature. Eur Polym J 45:2935–2941
Spěváček J, Hanyková L, Starovoytova L (2004) 1H NMR relaxation study of thermotropic phase transition in poly(vinyl methyl ether)/D2O solutions. Macromolecules 37:7710–7718
Starovoytova L, Spěváček J, Ilavský M (2005) 1H NMR study of temperature-induced phase transitions in D2O solutions of poly(N-isopropylmethacrylamide)/poly(N-isopropylacrylamide) mixtures and random copolymers. Polymer 46:677–683
Kouřilová H, Šťastná J, Hanyková L, Sedláková Z, Spěváček J (2010) 1H NMR study of temperature- induced phase separation in solutions of poly(N-isopropylmethacrylamide-co-acrylamide) copolymers. Eur Polym J 46:1299–1306
Spěváček J (2011) NMR investigations of temperature-induced phase transition in aqueous polymer solutions. Macromol Symp 305:18–25
Spěváček J, Dybal J, Starovoytova L, Zhigunov A, Sedláková Z (2012) Temperature-induced phase separation and hydration in poly(N-vinylcaprolactam) aqueous solutions: a study by NMR and IR spectroscopy, SAXS, and quantum chemical calculations. Soft Matter 8:6110–6119
Farrar TC, Becker ED (1971) Pulse and Fourier transform NMR. Academic Press, New York, p 27
Tokuhiro T, Amiya T, Mamada A, Tanaka T (1991) NMR study of poly(N-isopropylacrylamide) gels near phase transition. Macromolecules 24:2936–2943
Spěváček J, Geschke D, Ilavský M (2001) 1H NMR study of temperature collapse of linear and crosslinked poly(N,N-diethylacrylamide) in D2O. Polymer 42:463–468
Spěváček J, Hanyková L, Ilavský M (2001) Phase separation in poly(N,N-diethylacrylamide)/D2O solutions and physical gels as studied by 1H NMR spectroscopy. Macromol Chem Phys 202:1122–1129
Díez-Peña E, Quijada-Garrido I, Barrales-Rienda JM, Wilhelm M, Spiess HW (2002) NMR studies of the structure and dynamics of polymer gels based on N-isopropylacrylamide (NiPAAm) and methacrylic acid (MAA). Macromol Chem Phys 203:491–502
Rusu M, Wohlrab S, Kuckling D, Möhwald H, Schönhoff M (2006) Coil-to-globule transition of PNIPAM graft copolymers with charged side chains: a 1H and 2H NMR and spin-relaxation study. Macromolecules 39:7358–7363
Acknowledgments
This work was supported by the Czech Science Foundation (Project 202/09/1281), Ministry of Education of CR (Project MSM0021620835) and AS CR (Project AVOZ 40500505).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kouřilová, H., Spěváček, J. & Hanyková, L. 1H NMR study of temperature-induced phase transitions in aqueous solutions of poly(N-isopropylmethacrylamide)/poly(N-vinylcaprolactam) mixtures. Polym. Bull. 70, 221–235 (2013). https://doi.org/10.1007/s00289-012-0831-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-012-0831-x