Abstract
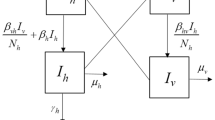
Zika virus has acquired worldwide concern after a recent outbreak in Latin America that started in Brazil, with associated neurological conditions such as microcephaly in newborns from infected mothers. The virus is transmitted mainly by Aedes aegypti mosquitoes, but direct (sexual) transmission has been documented. We formulate a reaction diffusion model that considers spatial movement of humans and vectors, with local contact transmission of Zika virus. Vaccination is introduced as a control variable, giving immunity to susceptible humans, in order to characterize an optimal vaccination strategy that minimizes the costs associated with infections and vaccines. The optimal control characterization is obtained in terms of state and adjoint equations. Parameter estimation and numerical simulations are carried out using data for the initial 2015 Zika outbreak in the state of Rio Grande do Norte in Brazil. Several scenarios are considered and analyzed in terms of number of new infections and costs, showing that the optimal control application is successful, significantly reducing these quantities.










Similar content being viewed by others
References
Abboubakar H, Kamgang JC, Nkamba LN, Tieudjo D (2017) Bifurcation thresholds and optimal control in transmission dynamics of arboviral diseases. J Math Biol 76:1–49
Agusto F, Bewick S, Fagan W (2017) Mathematical model for Zika virus dynamics with sexual transmission route. Ecol Complex 29:61–81
Althaus CL, Low N (2016) How relevant is sexual transmission of Zika virus? PLoS Med. https://doi.org/10.1371/journal.pmed.1002157
Anguelov R, Dufourd C, Dumont Y (2017) Simulations and parameter estimation of a trap-insect model using a finite element approach. Math Comput Simul 133:47–75
Asano E, Gross LJ, Lenhart S, Real LA (2008) Optimal control of vaccine distribution in a rabies metapopulation model. Math Biosci Eng 5(2):219–238
Besnard M, Lastere S, Teissier A, Cao-Lormeau V, Musso D (2014) Evidence of perinatal transmission of Zika virus, French Polynesia, December 2013 and February 2014. Eurosurveillance 19(13):20,751
Betts JT (2010) Practical methods for optimal control and estimation using nonlinear programming, vol 19. SIAM, Philadelphia
Bonyah E, Khan MA, Okosun K, Islam S (2017) A theoretical model for Zika virus transmission. PLoS ONE. https://doi.org/10.1371/journal.pone.0185540
Brockmann D, Hufnagel L, Geisel T (2006) The scaling laws of human travel. Nature 439(7075):462
De Araujo AL, Boldrini JL, Calsavara BM (2016) An analysis of a mathematical model describing the geographic spread of dengue disease. J Math Anal Appl 444(1):298–325
De Silva KR, Phan TV, Lenhart S (2017) Advection control in parabolic PDE systems for competitive populations. Discrete Contin Dyn Syst Ser B 22(3):1049–1072
Douglas J Jr, Dupont T, Ewing RE (1979) Incomplete iteration for time-stepping a Galerkin method for a quasilinear parabolic problem. SIAM J Numer Anal 16(3):503–522
D’Ortenzio E, Matheron S, de Lamballerie X, Hubert B, Piorkowski G, Maquart M, Descamps D, Damond F, Yazdanpanah Y, Leparc-Goffart I (2016) Evidence of sexual transmission of Zika virus. N Engl J Med 374(22):2195–2198
Evans LC (1998) Partial differential equations. American Mathematical Society, Providence
Fister R (1997) Optimal control of harvesting in a predator-prey parabolic system. Houst J Math 23(2):341–355
Fitzgibbon W, Morgan J, Webb G (2017) An outbreak vector-host epidemic model with spatial structure: the 2015–2016 zika outbreak in rio de janeiro. Theoretical Biology and Medical Modelling 14(1):7
Gao D, Lou Y, He D, Porco TC, Kuang Y, Chowell G, Ruan S (2016) Prevention and control of zika as a mosquito-borne and sexually transmitted disease: a mathematical modeling analysis. Sci Rep. https://doi.org/10.1038/srep28070
Gonzalez MC, Hidalgo CA, Barabasi AL (2008) Understanding individual human mobility patterns. Nature 453(7196):779
Hackbusch W (1978) A numerical method for solving parabolic equations with opposite orientations. Computing 20(3):229–240
Health Office (2017) Rio Grande do Norte epidemiological bulletin, 2016. http://www.saude.rn.gov.br. Accessed 16 August 2017
Hinze M, Schulz V (2010) Preface of the guest-editors. GAMM-Mitteilungen 33(2):131–132
Hiriart-Urruty JB, Korytowski A, Maurer H, Szymkat M (2016) Advances in mathematical modeling. Optimization and optimal control, vol 109. Springer, Berlin
Honório NA, Castro MG, Barros FSMd, Magalhães MdAFM, Sabroza PC (2009) The spatial distribution of Aedes aegypti and Aedes albopictus in a transition zone, Rio de Janeiro, Brazil. Cadernos de Saúde Pública 25(6):1203–1214
Hughes TJ (2012) The finite element method: linear static and dynamic finite element analysis. Courier Corporation, North Chelmsford
Keeling MJ, Rohani P (2008) Modeling infectious diseases in humans and animals. Princeton University Press, Princeton
Kon CML, Labadin J, Tiga J (2016) Generic reaction-diffusion model for transmission of mosquito-borne diseases: results of simulation with actual cases. In: ECMS, pp 93–99
Lenhart S, Workman JT (2007) Optimal control applied to biological models. Crc Press, New York
LeVeque RJ (2007) Finite difference methods for ordinary and partial differential equations: steady-state and time-dependent problems, vol 98. SIAM, Philadelphia
Li X, Yong J (2012) Optimal control theory for infinite dimensional systems. Springer, Berlin
Lions JL (1971) Optimal control of systems governed by partial differential equations. Springer, Berlin
Lou Y, Zhao XQ (2011) A reaction-diffusion malaria model with incubation period in the vector population. J Math Biol 62(4):543–568
Malone RW, Homan J, Callahan MV, Glasspool-Malone J, Damodaran L, Schneider ADB, Zimler R, Talton J, Cobb RR, Ruzic I, Smith-Gagen J, Janies D, Wilson J (2016) Zika virus: medical countermeasure development challenges. PLoS Negl Trop Dis. https://doi.org/10.1371/journal.pntd.0004530
Marcondes CB, Ximenes MdFFd (2016) Zika virus in Brazil and the danger of infestation by Aedes (Stegomyia) mosquitoes. Revista da Sociedade Brasileira de Medicina Tropical 49(1):4–10
Marino S, Hogue IB, Ray CJ, Kirschner DE (2008) A methodology for performing global uncertainty and sensitivity analysis in systems biology. J Theor Biol 254(1):178–196
Miyaoka TY, Meyer JFCA, Souza JMR (2017) A general boundary condition with linear flux for advection–diffusion models. Trends Appl Comput Math 18(2):253–272
Mlakar J, Korva M, Tul N, Popović M, Poljšak-Prijatelj M, Mraz J, Kolenc M, Resman Rus K, Vesnaver Vipotnik T, Fabjan Vodušek V, Vizjak A, Jože P, Petrovec M (2016) Avšič Županc T (2016) Zika virus associated with microcephaly. New England Journal of Medicine 374:951–958
Mocenni C, Madeo D, Sparacino E (2011) Linear least squares parameter estimation of nonlinear reaction diffusion equations. Math Comput Simul 81(10):2244–2257
Motta IJ, Spencer BR, Cordeiro da Silva SG, Arruda MB, Dobbin JA, Gonzaga YB, Arcuri IP, Tavares RC, Atta EH, Fernandes RF, Costa DA, Ribeiro LJ, Limonte F, Higa LM, Voloch CM, Brindeiro RM, Tanuri A, Ferreira OC (2016) Evidence for transmission of Zika virus by platelet transfusion. N Engl J Med 375(11):1101–1103
Murray JD (2002) Mathematical biology. Vol. 2: spatial models and biomedical applications. Springer, New York
Neilan RM, Lenhart S (2011) Optimal vaccine distribution in a spatiotemporal epidemic model with an application to rabies and raccoons. J Math Anal Appl 378(2):603–619
Neitzel I, Tröltzsch F (2008) On convergence of regularization methods for nonlinear parabolic optimal control problems with control and state constraints. Control Cybernet 37(4):1013–1043
NIAID (2017) National Institute of Allergy and Infectious Diseases. https://www.niaid.nih.gov/news-events/nih-begins-testing-investigational-zika-vaccine-humans. Accessed 26 Oct 2017
Oehler E, Watrin L, Larre P, Leparc-Goffart I, Lastere S, Valour F, Baudouin L, Mallet H, Musso D, Ghawche F (2014) Zika virus infection complicated by Guillain-Barre syndrome–case report, French Polynesia, December 2013. Eurosurveillance. https://doi.org/10.2807/1560-7917.ES2014.19.9.20720
Otero M, Schweigmann N, Solari HG (2008) A stochastic spatial dynamical model for Aedes aegypti. Bull Math Biol 70(5):1297
PAHO (2017) Pan American Health Organization. https://www.paho.org/zika. Accessed 26 Oct 2017
Powell MJ (2009) The BOBYQA algorithm for bound constrained optimization without derivatives. Cambridge NA Report NA2009/06, University of Cambridge, Cambridge
Raimundo SM, Yang HM, Massad E (2016) Modeling vaccine preventable vector-borne infections: yellow fever as a case study. J Biol Syst 24(02n03):193–216
Rodrigues HS, Monteiro MTT, Torres DF (2014) Vaccination models and optimal control strategies to dengue. Math Biosci 247:1–12
Soubeyrand S, Roques L (2014) Parameter estimation for reaction–diffusion models of biological invasions. Popul Ecol 56(2):427–434
Valega-Mackenzie W, Ríos-Soto KR (2018) Can vaccination save a Zika virus epidemic? Bull Math Biol 80(3):598–625
Wang L, Zhao H, Oliva SM, Zhu H (2017) Modeling the transmission and control of Zika in brazil. Sci Rep. https://doi.org/10.1038/s41598-017-07264-y
WHO (2017) World Health Organization. https://www.who.int/topics/zika. Accessed 26 Oct 2017
Zheng B, Tang M, Yu J, Qiu J (2017) Wolbachia spreading dynamics in mosquitoes with imperfect maternal transmission. J Math Biol 76:1–29
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This work was supported by CAPES (Process Number 88881.134100/2016-01) and by the National Institute for Mathematical and Biological Synthesis, an Institute sponsored by the National Science Foundation through NSF Award #DBI-1300426, with additional support from The University of Tennessee, Knoxville.
Appendix: Derivation of optimal control
Appendix: Derivation of optimal control
We seek to find an optimal control \(u^* \in U\) satisfying (4), where U is the set of admissible controls defined in (3). This optimization problem is subject to the state system (1), with state functions in \(L^2(0,T;H^1(\varOmega ))\) and corresponding time derivatives in \(L^2(0,T;H^1(\varOmega )^*)\). In order to find \(u^*\), we have to solve an optimality system, which consists of the state PDEs, adjoint PDEs, and the optimal control characterization. To obtain the sensitivity system, we must differentiate the control-to-state map \(u \rightarrow \left( S,I,R,S_v,I_v \right) (u)\). We use that system and the objective functional to find the adjoint system. Then we use the sensitivity and adjoint system to simplify the derivative of the map \(u \rightarrow J(u)\), finally obtaining the desired optimal control characterization. The derivatives of both of these maps are computed as Fréchet derivatives (derivatives in dircetion \(\ell \)). In the following, we provide an overview of the derivation of the sensitivity and adjoint equations, and the control characterization. Details about optimal control theory in PDEs can be found in De Silva et al. (2017), Fister (1997), Lenhart and Workman (2007), Li and Yong (2012), Lions (1971).
1.1 Sensitivity system
From the control-to-state map, we define \((S^\epsilon ,I^\epsilon ,R^\epsilon ,S_v^\epsilon ,I_v^\epsilon ) = (S,I,R,S_v,I_v)(u + \epsilon \ell )\). We then define the sensitivity functions, which are Gateaux derivatives of the control-to-state map:
We also have:
The sensitivity functions \(\varvec{\psi }=(\psi _1,\psi _2,\psi _3,\psi _4,\psi _5)\) satisfy a system corresponding to a linearized version of the state equations. See works by De Silva et al. (2017), Li and Yong (2012), and Lions (1971) to show justification of such convergence of these quotients and the existence of the sensitivity functions and their PDEs. These equations are used to derive PDEs for the adjoint \(\varvec{\lambda }=(\lambda _1,\lambda _2,\lambda _3,\lambda _4,\lambda _5)\), where each adjoint variable is associated with a state variable.
We now calculate equations for the sensitivity functions. We calculate an equation for the quotient \(\dfrac{S^\epsilon - S}{\epsilon }\):
Letting \(\epsilon \rightarrow 0\), passing to the limit gives:
In the same way, letting \(\epsilon \rightarrow 0\) in
gives:
Note that the term with external source f vanishes because it depends only on (x, t). Similarly:
gives:
For \(S_v\), we obtain:
and:
Similarly, for \(I_v\) we have:
and:
In a similar way, we obtain initial conditions:
and boundary conditions:
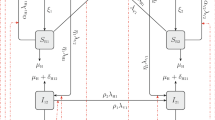
We can write the sensitivity system in vector form as following:
where:
and the matrix \(\varvec{M}^T\) is given by (6).
1.2 Adjoint system
For the adjoint variables \(\varvec{\lambda }=(\lambda _1,\ldots , \lambda _5)\), the adjoint operator is obtained from the relation:
in the appropriate weak \(L^2(Q)\) sense. The right hand side of the adjoint system is given by the derivatives of the integrand of the objective functional, with respect to the states:
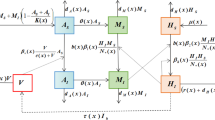
We use the expression (14) in order to derive the operators in the adjoint system. This is done using integration by parts on \(\langle \varvec{\lambda } , \mathscr {L} \varvec{\psi } \rangle \) and the boundary conditions of the sensitivity equations. The adjoint system obtained this way is:
where:
and:
with final time conditions given by (7) and boundary conditions by (8).
1.3 Optimal control characterization
We now derive an expression for an optimal control \(u^*\) as a function of the state and adjoint variables. The derivative of the map \(u \rightarrow J(u)\) at \(u^*\) in the direction \(\ell \) is:
with:
Using the sensitivity and adjoint systems we find a characterization for \(u^*\):
On the set where \(u_{\min }< u^*(x, t) < u_{\max }\), the variation \(\ell \) can be an \(L^{\infty }\) function with arbitrary sign, and thus on this set, we obtain:
Due to the boundedness \(u_{\min } \le u \le u_{\max }\), and taking variations \(\ell \) with appropriate signs when \(u^*\) is at the lower or upper bounds, we can obtain the following optimal control characterization:
Rights and permissions
About this article
Cite this article
Miyaoka, T.Y., Lenhart, S. & Meyer, J.F.C.A. Optimal control of vaccination in a vector-borne reaction–diffusion model applied to Zika virus. J. Math. Biol. 79, 1077–1104 (2019). https://doi.org/10.1007/s00285-019-01390-z
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-019-01390-z