Abstract
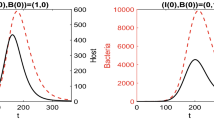
A network epidemic SIWR model for cholera and other diseases that can be transmitted via the environment is developed and analyzed. The person-to-person contacts are modeled by a random contact network, and the contagious environment is modeled by an external node that connects to every individual. The model is adapted from the Miller network SIR model, and in the homogeneous mixing limit becomes the Tien and Earn deterministic cholera model without births and deaths. The dynamics of our model shows excellent agreement with stochastic simulations. The basic reproduction number \(\mathcal {R}_0\) is computed, and on a Poisson network shown to be the sum of the basic reproduction numbers of the person-to-person and person-to-water-to-person transmission pathways. However, on other networks, \(\mathcal {R}_0\) depends nonlinearly on the transmission along the two pathways. Type reproduction numbers are computed and quantify measures to control the disease. Equations giving the final epidemic size are obtained.

Similar content being viewed by others
References
Ball F, Neal P (2008) Network epidemic models with two levels of mixing. Math Biosci 212:69–87
Bekessy A, Bekessy P, Komlos J (1972) Asymptotic enumeration of regular matrices. Stud Sci Math Hung 7:343–353
Bender EA, Canfield ER (1978) The asymptotic number of labelled graphs with given degree sequences. J Comb Theory A 24:296–307
Eames KTD, Keeling MJ (2002) Modelling dynamic and network heterogeneneities in the spread of sexually transmitted diseases. Proc Natl Acad Sci 99:13330–13335
Eisenberg MC, Shuai Z, Tien JH, van den Driessche P (2013) A cholera model in a patchy environment with water and human movement. Math Biosci 246:105–112
Gillespie DT (1976) A general method for numerically simulating the stochastic time evolution of coupled chemical reactions. J Comput Phys 22:403–434
Gillespie DT (1977) Exact stochastic simulation of coupled chemical reactions. J Phys Chem 81:2340–2361
Heesterbeek JAP, Roberts MG (2007) The type-reproduction number T in models for infectious disease control. Math Biosci 206:3–10
Kapp C (2009) Zimbabwe’s humanitarian crisis worsens. Lancet 373:447
Keeling MJ (1999) The effects of local spatial structure on epidemiological invasions. Proc Roy Soc B 266:859–867
Kiss I, Green D, Kao R (2006) The effect of contact heterogeneity and multiple routes of transmission on final epidemic size. Math Biosci 203:124–136
Koch D, Illner R, Ma J (2013) Edge removal in random contact networks and the basic reproducation number. J Math Biol 67:217–238
Li M, Illner R, Edwards R, Ma J (2014) Marketing new products: Bass models on random graphs. Commun Math Sci. (to appear)
Lindquist J, Ma J, van den Driessche P, Willeboordse FH (2010) Effective degree network disease models. J Math Biol 62:143–164
Miller JC (2011) A note on a paper by Erik Volz: SIR dynamics in random networks. J Math Biol 62:349–358
Miller JC, Volz EM (2013) Model hierarchies in edge-based compartmental modeling for infectious disease spread. J Math Biol 67:869–899
Molloy M, Reed B (1995) A critical point for random graphs with a given degree sequence. Random Struct Algorithms 6:161–179
Mugero C, Hoque A (2001) Review of cholera epidemic in South Africa with focus on KwaZulu-Natal province. Technical report, KwaZulu-Natal Department of Health, Pietermaritzburg
Newman MEJ (2002) Spread of epidemic disease on networks. Phys Rev E 66:016128
Newman MEJ, Strogatz SH, Watts DJ (2001) Random graphs with arbitrary degree distributions and their applications. Phys Rev E 64:026118
Pastor-Satorras R, Vespignani A (2002) Epidemic dynamics in finite size scale-free networks. Phys Rev E 65:035108(R)
Roberts MG, Heesterbeek JAP (2003) A new method for estimating the effort required to control an infectious disease. Proc R Soc Lond B 270:1359–1364
Sack DA, Sack RB, Chaignat C-L (2006) Getting serious about cholera. N Engl J Med 355:649–651
Taylor TJ, Kiss IZ (2014) Interdependency and hierarchy of exact and approximate epidemic models on networks. J Math Biol 69:183–211
Tien JH, Earn DJD (2010) Multiple transmission pathways and disease dynamics in a waterborne pathogen model. Bull Math Biol 72:1506–1533
Tuite AR, Tien J, Eisenberg M, Earn DJD, Ma J, Fisman DN (2010) Cholera epidemic in Haiti, 2010: using a transmission model to explain spatial spread of disease and identify optimal control interventions. Ann Intern Med 154:593–601
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–42
Volz EM (2008) SIR dynamics in random networks with heterogeneous connectivity. J Math Biol 56:293–310
World Health Organization (WHO) (2012) Cholera fact sheet. Technical report, 2012. http://www.who.int/mediacentre/factsheets/fs107/en/
Acknowledgments
Research supported by Discovery grants from the Natural Sciences and Engineering Research Council Canada (PvdD and JM), National Natural Science Foundation of China (No. 11371087) (ML) and Natural Science Foundation of Shanghai (No. 12ZR1400100) (ML). The authors thank Tom Britton and two anonymous referees for helpful comments, especially for pointing out the similarities between our results and those of the two-level mixing model of Ball and Neal (2008).
Author information
Authors and Affiliations
Corresponding author
Appendix A The homogeneous mixing limit
Appendix A The homogeneous mixing limit
Here we show that our model (14) becomes the Tien and Earn (2010) SIWR model without births and deaths in a homogeneously mixing population, i.e., on a complete graph. On such a complete graph with \(N\gg 1\) individuals, every node has degree \(N-1\). Thus
Thus using (14c),
where \(q=\beta _I(N-1)\).
For a complete graph, \(I\) should be equal to \(\phi /\theta \). This can be verified by computing \(\frac{d}{dt}\left( \frac{\phi }{\theta }\right) \) and comparing with \(dI/dt=qSI+\beta _W WS-\gamma I\).
as \(N\rightarrow \infty \). This agrees with \(dI/dt\) provided that \(\theta (t)\rightarrow 1\) for any time \(t\) as \(N\rightarrow \infty \). This can be proved from (14b):
and thus with \(\theta (0)=1\), for any \(t\), \(\theta \ge 1-qt/(N-1)\rightarrow 1\) as \(N\rightarrow \infty \). Thus, on a complete network, our model becomes model (1) as the population size \(N\rightarrow \infty \).
In fact, Miller [2011, Appendix A] showed that the limit holds for an SIR model if all individuals have the same degree and the degree approaches infinity. In addition, Miller and Volz (2013) relaxed the condition to be the average degree \(\left\langle k\right\rangle \rightarrow \infty \) while \(\left\langle k^4\right\rangle /\left\langle k\right\rangle ^4\rightarrow 1\). That is, the limit holds even if the network does not approach a complete graph. This same argument applies here.
Rights and permissions
About this article
Cite this article
Li, M., Ma, J. & van den Driessche, P. Model for disease dynamics of a waterborne pathogen on a random network. J. Math. Biol. 71, 961–977 (2015). https://doi.org/10.1007/s00285-014-0839-y
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-014-0839-y