Abstract
Bombyx mori bidensovirus (BmBDV) was previously termed as Bombyx mori densovirus type 2 and later it was reclassified in the new genus bidensovirus of the new family Bidnaviridae. The genome of BmBDV Zhenjiang isolate (BmBDV-Z) consists of two non-homologous single-stranded linear DNA molecules VD1 and VD2 which are encapsidated into separate virion. To investigate the infectivity of BmBDV DNA, recombinant plasmids pGEM-VD1 inserted with VD1 genome were transfected into the BmN cells of silkworm. Structural proteins of BmBDV were detected with Western blot and immunofluorescence assay, which indicates pGEM-VD1 replicated in the transfected BmN cells and viral proteins were also expressed. Through TEM observation, we identified about 20 nm BmBDV-like viral particles, which confirmed that BmBDV can be generated after transfection. Subsequently, a recombinant baculovirus BmBac-VD1 inserted with VD1 genome was constructed. Results of Western blot and immunofluorescence assay indicated that viral structural proteins of BmBDV were expressed in the BmBac-VD1-infected cells. Baculiform and spherical virions were also observed in infected cells by TEM, and two kinds of virions were separated. However, results of molecular biological detection revealed that infectious sequence from BmBac-VD1 was packaged within spherical virion. Therefore, we suggested that vector inserted with BmBDV genomic DNA showed infectivity, and BmBDV-like viral particles packaging recombinant DNA can be produced in the cultured BmN cells. Outcome of our current research provided not only a new method of infection to explore the gene function of BmBDV in vitro but also a protocol to facilitate development of more effective new-type pesticides.





Similar content being viewed by others
References
Adams MJ, Carstens EB (2012) Ratification vote on taxonomic proposals to the international committee on taxonomy of viruses. Arch Virol 157:1411–1422
Afanasiev BN, Carlson J (2000) Densovirinae as gene transfer vehicles. Contrib Microbiol 4:33–58
Afanasiev BN, Kozlov YV, Carlson JO, Beaty BJ (1994) Densovirus of Aedes aegypti as an expression vector in mosquito cells. Exp Parasitol 79(3):322–339
Bergoin M, Tijssen P (2000) Molecular biology of Densovirinae. Contrib Microbiol 4:12–32
Bonami JR, Mari J, Poulos BT, Lightner DV (1995) Characterization of hepatopancreatic parvo-likevirus, a second unusual parvovirus pathovirus pathogenic for penaeid shrimps. J Gen Virol 76(Pt4):813–817
Bossin H, Fournier P, Royer C, Barry P, Cérutti P, Gimenez S, Couble P, Bergoin M (2003) Junonia coenia densovirus-based vectors for stable transgene expression in Sf9 cells: influence of the densovirus sequences on genomic integration. J Virol 77(20):11060–11071
Carter BJ (1990) Parvoviruses as vectors. In: Tijssen P (ed) Handbook of parvoviruses, vol II. CRC Press, Boca Raton, pp 247–284
Fediere G (2004) Epidemiology and pathology of Densovirinae. Contrib Microbiol 4:1–11
Firth AE, Brierley I (2012) Non-canonical translation in RNA viruses. J Gen Virol 93(Pt 7):1385–1409
Fu YH, Tang SM, Tan GX, Liu T, Guo XJ (2008) Cloning of the Genome of Bombyx mori densovirus Zhenjiang strain and rescue of infectious virions from recombinant plasmids in the silkworm. Sci Seric 24(3):453–458
Fuxa JR (2003) Ecology of insect nucleopolyhedroviruses. Agric Ecosyst Environ 103:27–43
Giraud C, Devauchelle G, Bergoin M (1992) The densovirus of Junonia coenia (JcDNV) as an insect cell expression vector. Virology 186:207–218
Hu Y, Zheng J, Iizuka T, Bando H (1994) A densovirus newly isolated from the smoky-brown cockroach Periplaneta fuliginosa. Arch Virol 138:365–372
Hu L, Zhang L, Shen C, Lu J, Zhang J, Hu Y (2007) The densovirus of Periplaneta fuliginosa (PfDNV) as an insect vector for persistent foreign gene expression in vivo. Biochem Biophys Res Commun 358(4):976–982
Iwashita Y, Chun CY (1982) The development of a densonucleosis virus isolated from silkworm larvae, Bombys mori, of China. In: Akai H, King RC, Morohoshi S (eds) The ultrastructure and functioning of insect Cell, p161–164
Jourdan M, Jousset FX, Gervais M, Skory S, Bergoin M, Dumas B (1990) Cloning of the genome of a densovirus and rescue of infectious virions from recombinant plasmid in the insect host Spodoptera littoralis. Virology 179:403–409
Li GH, Hu ZY, Guo XL, Li GT, Tang Q, Wang P, Chen KP, Yao Q (2013) Identification of BmBDV VD1-ORF4 reveals a novel protein associated with viral structural component. Curr Micribiol 66(6):527–534
Li G, Zhou Q, Hu Z, Wang P, Tang Q, Chen K, Yao Q (2015) Determination of the proteins encoded by BmBDV VD1-ORF4 and their interacting proteins in BmBDV-infected midguts. Curr Microbiol 70(4):623–629
Liu TX, Kang L (2005) Research on insectology-progress and prospect: research and application of insect baculovirus on pest control. Science press, Beijin, pp 76–85
Mari J, Bonami JR, Lighter DV (1993) Partial cloning of the genome of infectious hypodermal and haemopoietic necrosis virus, an unusual parvovirus pathogenic for penaeid shrimps, diagnosis of the disease using a specific probe. J Gen Virol 74:2637–2643
Maxwell IH, Terrell KL, Maxwell F, Maxwell F (2002) Autonomous parvovirus vectors. Methods 28(2):168–181
Moscardi F (1999) Assessment of the application of baculoviruses for control of Lepidoptera. Annu Rev Entomol 44:257–289
Moscardi F, Santos B (2005) Producao comercial de nucleopoliedrosis de Anticarsia gemmatalis HUBNER (Lep.: Noctuidae) em laboratorio. In: Proceedings of the IX Simposio de Controle Biologico, Recife, Brazil, p 42
Moscardi F, Morales L, Santos B (2002) The successful use of AgMNPV for the control of velvet bean caterpillar, Anticarsia gemmatalis, in soybean in Brazil. In: Proceedings of the VIII international colloquium on invertebrate pathology and microbial control and XXXV annual meeting of the Society for Invertebrate Pathology. Foz do Iguassu Brazil, pp 86–91
Summers MD, Smith GE (1987) A manual of methods for baculovirus vectors and insect cell culture procedures. Tex Agric Exp Stat Bull 1555:1–57
Tijssen P, Li Y, El-Far M, Szelei J, Letarte M, Zádori Z (2003) Organization and expression strategy of the ambisense genome of densonucleosis virus of Galleria mellonella. J Virol 77(19):10357–10365
Wang YJ, Yao Q, Chen KP, Wang Y, Lu J, Han X (2007) Characterization of the genome structure of Bombyx mori densovirus (China isolate). Virus Genes 35:103–108
Wang Y, Abd-Alla AM, Bossin H, Li Y, Bergoin M (2013) Analysis of the transcription strategy of the Junonia coenia densovirus (JcDNV) genome. Virus Res 174(1–2):101–107
Zhang XD, Zhang JM, Guo HT, Zhu LH, Hu YY (1999) Rescue of infectious virions from recombinant plasmid with the genome of Periplaneta fuliginosa densovirus. Virology 14:152–156
Acknowledgments
Authors greatly acknowledge the financial support of the National Natural Science Foundation of China (31272500), State Key Laboratory of Silkworm Genome Biology (SKLSGB201200011), the National Basic Research Program of China (973 Program, 2012CB114600), the Specialized Research Fund for the Doctoral Program of Higher Education (20113201130002), and a project funded by the Priority Academic Program of Development of Jiangsu Higher Education Institutions. Thanks to Prof. Xijie Guo for providing plasmid pGEM-VD1, and to Prof. Keping Chen and Prof. Xiaofeng Wu for providing antibodies.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors have declared that no conflict of interest exists.
Additional information
Rui Guo and Guangli Cao have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
284_2016_1102_MOESM1_ESM.docx
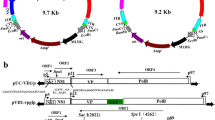
Supplementary material 1 (DOCX 40 kb) Fig. S1 Construction steps of recombinant baculovirus BmBac-VD1. VD1 genome excised from the pGEM-VD1 with restriction endonucleases SphI and EcoRI was subcloned into pFastBac™ Dual to obtain the donor plasmid pFastBac™ Dual-VD1, then the constructed plasmid was transformed into E. coli DH10 containing B. mori nucleopolyhedrovirus bacmid and helper plasmid. The transformations were screened on a LB agar medium containing kanamycin (50 µg/ml), gentamycin (7 µg/ml),tetracycline (10 µg/ml), X-Gal (100 µg/ml) and IPTG (40 µg/ml) to generate the recombinant Bmbacmid-VD1 according to the protocol of Bac-to-Bac baculovirus expression system
Rights and permissions
About this article
Cite this article
Guo, R., Cao, G., Zhu, Y. et al. Novel Infection System of Recombinant BmBDV DNA into BmN Cells of Silkworm, Bombyx mori . Curr Microbiol 73, 587–594 (2016). https://doi.org/10.1007/s00284-016-1102-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-016-1102-0