Abstract
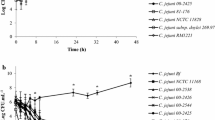
Campylobacter jejuni (C. jejuni) is a leading cause of human enteritis worldwide and the most frequently reported zoonotic agent in the European Union. Despite the fact that C. jejuni is a microaerobic bacteria, known as a fragile one, it is able to survive through adverse conditions such as oxidative stress. The purpose of this study was first to test the oxidative stress resistance in 22 C. jejuni strains of various origins, and to compare adhesive and invasive abilities of four selected strains in the intestinal cell line Ht-29. Secondly, the effect of an oxidative stress on C. jejuni adhesion to Ht-29 cells was investigated. Results show that all the tested strains were able to survive after a 24-h incubation period in broth containing 10 μM of paraquat. From 12.5 μM of paraquat, bacterial strains exhibit different behaviour, and only three strains are able to survive at 25 μM of paraquat. In addition, this study revealed that the number of bound bacteria to epithelial cells increases with augmentation of paraquat concentration, suggesting a link between oxidative stress survival of C. jejuni and virulence on Ht-29 cells.


Similar content being viewed by others
References
Allos BM (2001) Campylobacter jejuni infections: update on emerging issues and trends. Clin Infect Dis 32:1201–1206
Butzler JP (2004) Campylobacter, from obscurity to celebrity. Clin Microbiol Infect 10:868–876
Young VB, Mansfield LS (2005) Campylobacter infection-clinical context. In: Ketley JM, Konkel ME (eds) Campylobacter: cellular and molecular biology. Horizon Bioscience, Norfolk, pp 1–12
Anonymous (2009) The community report on trends and sources of zoonoses and zoonotic agents in the European Union in 2007. EFSA J 223:22–35
Fosse J, Seegers H, Magras C (2008) Foodborne zoonoses due to meat: a quantitative approach for a comparative risk assessment applied to pig slaughtering in Europe. Vet Res 39:01
Jacobs-Reitsma W (2000) Campylobacter in the food supply. In: Blaser MJ, Nachamkin I (eds) Campylobacter, 2nd edn. ASM, Washington, DC, pp 467–481
Kuhn R, Lohler J, Rennick D et al (1993) Interleukin-10-deficient mice develop chronic enterocolitis. Cell 75:263–274
Mead PS, Slutsker L, Dietz V et al (1999) Food-related illness and death in the United States. Emerg Infect Dis 5:607–625
Nachamkin I, Allos BM, Ho TW (2000) Campylobacter jejuni infection and the association with Guillain-Barre syndrome. In: Blaser MJ, Nachamkin I (eds) Campylobacter, 2nd edn. ASM Press, Washington, DC, pp 155–175
Smith MA, Finel M, Korolik V et al (2000) Characteristics of the aerobic respiratory chains of the microaerophiles Campylobacter jejuni and Helicobacter pylori. Arch Microbiol 174:1–10
Baillon ML, Van Vliet AHM, Ketley JM et al (1999) An iron-regulated alkyl hydroperoxide reductase (AhpC) confers aerotolerance and oxidative stress resistance to the microaerophilic pathogen Campylobacter jejuni. J Bacteriol 181:4798–4804
Day WA, Sajecki JL, Pitts TM et al (2000) Role of catalase in Campylobacter jejuni intracellular survival. Infect Immun 68:6337–6345
Palyada K, Yi-Qian S, Flint A et al (2009) Characterization of the oxidative stress stimulon and PerR regulon of Campylobacter jejuni. BMC Genomics 10:481
Parkhill J, Wren BW, Mungall K et al (2000) The genome sequence of the food-borne pathogen Campylobacter jejuni reveals hypervariable sequences. Nature 403:665–668
Pesci EC, Cottle DL, Pickett CL (1994) Genetic, enzymatic, and pathogenic studies of the iron superoxide dismutase of Campylobacter jejuni. Infect Immun 62:2687–2694
Van Vliet AHM, Baillon ML, Penn CW et al (1999) Campylobacter jejuni contains two fur homologs: characterization of iron-response regulation of peroxide stress defense genes by the PerR repressor. J Bacteriol 181:6371–6376
Purdy D, Cawthraw S, Dickinson JH et al (1999) Generation of a superoxide dismutase (SOD)-deficient mutant of Campylobacter coli: Evidence for the significance of SOD in Campylobacter survival and colonization. Appl Environ Microbiol 65:2540–2546
Garenaux A, Guillou S, Ermel G et al (2008) Role of the Cj1371 periplasmic protein and the Cj0355c two-component regulator in the Campylobacter jejuni NCTC 11168 response to oxidative stress caused by paraquat. Res Microbiol 159:718–726
Hofreuter D, Tsai J, Watson RO et al (2006) Unique features of a highly pathogenic Campylobacter jejuni strain. Infect Immun 74:4694–4707
Chynoweth RW, Hudson JA, Thom K (1998) Aerobic growth and survival of Campylobacter jejuni in food and stream water. Lett Appl Microbiol 27:341–344
Van Deun K, Haesebrouck F, Heyndrickx M et al (2007) Virulence properties of Campylobacter jejuni isolates of poultry and human origin. J Med Microbiol 56:1284–1289
Coote JG, Stewart-Tull DES, Owen RJ et al (2007) Comparison of virulence-associated in vitro properties of typed strains of Campylobacter jejuni from different sources. J Med Microbiol 56:722–732
Farr SB, Kogoma T (1991) Oxidative stress responses in Escherichia coli and Salmonella typhimurium. Microbiol Rev 55:561–585
Mihaljevic RR, Sikic M, Klancnikb A et al (2007) Environmental stress factors affecting survival and virulence of Campylobacter jejuni. Microb Pathog 43:120–125
Pogacar MS, Mihaljevic RR, Klancnik A et al (2009) Survival of stress exposed Campylobacrer jejuni in the murine macrophage J774 cell line. Int J Food Microbiol 129:68–73
Fields JA, Thompson SA (2008) Campylobacter jejuni CsrA mediates oxidative stress responses, biofilm formation, and host cell invasion. J Bacteriol 190:3411–3416
Augusto AC, Miguel F, Mendonca S et al (2007) Oxidative stress expression status associated to Helicobacter pylori virulence in gastric diseases. Clin Biochem 40:615–622
Hebrard M, Viala JPM, Meresse P et al (2009) Redundant hydrogen peroxide scavengers contribute to Salmonella virulence and oxidative stress resistance. J Bacteriol 191:4605–4614
Acknowledgements
We are grateful to Magalie Ritz for her scientific help. We thank the Microbiological Laboratory of Public Health of the National Institute for Public Health and the Environment (The Netherlands) and Agence Française de Sécurité Sanitaire des Aliments (AFSSA, Unité HQPAP, Ploufragan, France) for providing C. jejuni strains. This study was supported by grants from the Institut National de la Recherche Agronomique (INRA), and from the Région des Pays de la Loire.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haddad, N., Maillart, G., Garénaux, A. et al. Adhesion Ability of Campylobacter jejuni to Ht-29 Cells Increases with the Augmentation of Oxidant Agent Concentration. Curr Microbiol 61, 500–505 (2010). https://doi.org/10.1007/s00284-010-9644-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-010-9644-z