Abstract
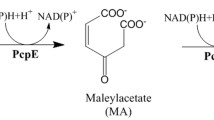
The final enzyme in the pentachlorophenol (PCP) biodegradation pathway in Sphingobium chlorophenolicum is maleylacetate reductase (PcpE), which catalyzes the reductive dehalogenation of 2-chloromaleylacetate to maleylacetate and the subsequent reduction of malyelacetate to 3-oxoadipate. In this study, the pcpE gene was cloned from S. chlorophenolicum strain ATCC 53874 and overexpressed in Escherichia coli BL21-AI cells. The recombinant PcpE, purified to higher than 95% purity using affinity chromatography, exhibited optimal activity at pH 7.0. The kinetic parameters k cat and K m were 1.2 ± 0.3 s−1 and 0.09 ± 0.04 mM, respectively, against maleylacetate under the optimal pH. In addition, the purified PcpE was able to restore PCP-degrading capability to S. chlorophenolicum strain ATCC 39723, implicating that there was no functional PcpE in the ATCC 39723 strain.





Similar content being viewed by others
References
Cai M, Xun L (2002) Organization and regulation of pentachlorophenol-degrading genes in Sphingobium chlorophenolicum ATCC 39723. J Bacteriol 184:4672–4680
Chen L, Yang J (2008) Biochemical characterization of the tetrachlorobenzoquinone reductase involved in the biodegradation of pentachlorophenol. Int J Mol Sci 9:198–212
Copley SD (2000) Evolution of a metabolic pathway for degradation of a toxic xenobiotic: the patchwork approach. Trends Biochem Sci 25:261–265
Crawford RL (1987) Biodegradation of pentachlorophenol. US Patent No. 4713340
Dai M, Rogers JB, Warner JR, Copley SD (2003) A previously unrecognized step in pentachlorophenol degradation in Sphingobium chlorophenolicum is catalyzed by tetrachlorobenzoquinone reductase (PcpD). J Bacteriol 185:302–310
Daniel V, Huber W, Bauer K, Suesal C, Mytilineos J, Melk A, Conradt C, Opelz G (2001) Association of elevated blood levels of pentachlorophenol (PCP) with cellular and humoral immunodeficiencies. Arch Environ Health 56:77–83
Frank R, Braun HE, Ripley BD, Clegg BS (1990) Contamination of rural ponds with pesticides, 1971–85, Ontario, Canada. Bull Environ Contam Toxicol 44:401–409
Ingham CF, Massy-Westropp RA, Reynolds GD, Thorpe WD (1975) A synthesis of enol-lactones. Aust J Chem 28:2499–2510
Ingram L, Tarlton K (2005) Effect of physical properties of pentachlorophenol and creosote components on vaporization from treated wood: review of prior data. Forest Prod J 55:86–89
Kaschabek SR, Reineke W (1995) Maleylacetate reductase of Pseudomonas sp. strain B13: specificity of substrate conversion and halide elimination. J Bacteriol 177:320–325
Khurana V, Barkin JS (2001) Pancreatitis induced by environmental toxins. Pancreas 22:102–105
Massy-Westropp RA, Price MF (1980) The synthesis of 5-oxo-2,5-dihydrofuran-2-ylideneacetic acids. Aust J Chem 33:333–341
McAllister KA, Lee H, Trevors JT (1996) Microbial degradation of pentachlorophenol. Biodegradation 7:1–40
McCarthy DL, Claude AA, Copley SD (1997) In vivo levels of chlorinated hydroquinones in a pentachlorophenol-degrading bacterium. Appl Environ Microbiol 63:1883–1888
Miyauchi K, Adachi Y, Nagata Y, Takagi M (1999) Cloning and sequencing of a novel meta-cleavage dioxygenase gene whose product is involved in degradation of γ-hexachlorocyclohexane in Sphingomonas paucimobilis. J Bacteriol 181:6712–6719
Peper M, Ertl M, Gerhard I (1999) Long-term exposure to wood-preserving chemicals containing pentachlorophenol and lindane is related to neurobehavioral performance in women. Am J Ind Med 35:632–641
Sai K, Kang KS, Hirose A, Hasegawa R, Trosko JE, Inoue T (2001) Inhibition of apoptosis by pentachlorophenol in v-myc-transfected rat liver epithelial cells: relation to down-regulation of gap junctional intercellular communication. Cancer Lett 173:163–174
Su Y, Chen L, Bandy B, Yang J (2008) The catalytic product of pentachlorophenol 4-monooxygenase is tetrachlorohydroquinone rather than tetrachlorobenzoquinone. Open Microbiol J 2:100–106
Thompson TS, Treble RG, Mariasegaram M (1997) Determination of atmospheric contamination by pentachlorophenol using pine needles located near treated utility pole storage sites. Bull Environ Contam Toxicol 59:548–555
Xun L, Orser CS (1991) Purification and properties of pentachlorophenol hydroxylase, a flavoprotein from Flavobacterium sp. strain ATCC 39723. J Bacteriol 173:4447–4453
Xun L, Topp E, Orser CS (1992) Diverse substrate range of a Flavobacterium pentachlorophenol hydroxylase and reaction stoichiometries. J Bacteriol 174:2898–2902
Xun L, Topp E, Orser CS (1992) Purification and characterization of a tetrachloro-p-hydroquinone reductive dehalogenase from a Flavobacterium sp. J Bacteriol 174:8003–8007
Acknowledgments
This work was supported by a Discovery Grant from the Natural Sciences and Engineering Research Council of Canada, a New Investigator Establishment Grant from the Saskatchewan Health Research Foundation, and a New Opportunities Fund from the Canada Foundation for Innovation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, L., Maloney, K., Krol, E. et al. Cloning, Overexpression, Purification, and Characterization of the Maleylacetate Reductase from Sphingobium chlorophenolicum Strain ATCC 53874. Curr Microbiol 58, 599–603 (2009). https://doi.org/10.1007/s00284-009-9377-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-009-9377-z