Abstract
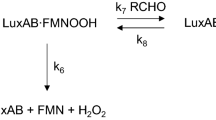
The environmental bacterium Bacillus licheniformis was transformed with two different shuttle-vectors (pCSS810 and pGFPratiometric) containing insect luciferase and green fluorescent protein genes, respectively. The cells were treated with various antimicrobial agents and the emitted bioluminescence and fluorescence were measured. Plasmid harboring the green fluorescent protein gene was totally segregated without selective pressure, and fluorescent B. licheniformis showed a slower growth rate than the wild-type strain; those cells were bright green as visualized by epifluorescent microscopy. However, fluorescence was not correlated to the growth state of cells or affected by the antibiotic treatments. To the contrary, luminescent transformant was found to be stable without antibiotic selection and showed analogous growth behavior compared to non-plasmid-bearing cells. The luminescent strain functioned as a biosensor for the antibiotics employed. Bioluminescence measurements allowed one to determine the viability of the recombinant cells and the kinetics of the antibacterial action could be followed. Thus, the light emission was found to be a reliable, sensitive, and real-time indicator of the “well-being” of cells, whereas fluorescence allowed one to visualize both metabolically active and inactive cells.



Similar content being viewed by others
References
Änkö ML, Kurittu J, Karp M (2002) An Escherichia coli biosensor strain for amplified and high throughput detection of antimicrobial agents. J Biomol Screen 7:119–125
Birrer GA, Cromwick AM, Gross RA (1994) Gamma-poly(glutamic acid) formation by Bacillus licheniformis 9945a: physiological and biochemical studies. Int J Biol Macromol 16:265–275
Chalfie M, Tu Y, Euskirchen G, Ward WW, Prasher DC (1994) Green fluorescent protein as a marker for gene expression. Science 263:802–805
Cutting SM, Vander Horn PB (1990) Genetic analysis. In: Cutting SM, Harwood CR (eds) Molecular biological methods for Bacillus. Wiley, Chichester, pp 27–74
Gray O, Chang S (1981) Molecular cloning and expression of Bacillus licheniformis beta-lactamase gene in Escherichia coli and Bacillus subtilis. J Bacteriol 145:422–428
Gwinn DD, Thorne CB (1964) Transformation of Bacillus licheniformis. J Bacteriol 87:519–526
Karp M (1989) Expression of bacterial luciferase genes from Vibrio harveyi in Bacillus subtilis and in Escherichia coli. Biochim Biophys Acta 1007:84–90
Kleinkauf H, von Dohren H (1987) Biosynthesis of peptide antibiotics. Annu Rev Microbiol 41:59–289
Lampinen J, Koivisto L, Wahlsten M, Mantsala P, Karp M (1992) Expression of luciferase genes from different origins in Bacillus subtilis. Mol Gen Genet 232:498–504
Loimaranta V, Tenovuo J, Koivisto L, Karp M (1998) Generation of bioluminescent Streptococcus mutans and its usage in rapid analysis of the efficacy of antimicrobial compounds. Antimicrob Agents Chemother 42:1906–1910
Manachini PL, Fortina MG, Levati L, Parini C (1998) Contribution to phenotypic and genotypic characterization of Bacillus licheniformis and description of new genomovars. Syst Appl Microbiol 21:520–529
March JC, Rao G, Bentley WE (2003) Biotechnological applications of green fluorescent protein. Appl Microbiol Biotechnol 62:303–315
McCuen RW, Thorne CB (1971) Genetic mapping of genes concerned with glutamyl polypeptide production by Bacillus licheniformis and a study of their relationship to the development of competence for transformation. J Bacteriol 107:636–645
Neyra C, Atkinson LA, Olubayi O, Sadasivan L, Zaurov D, Zappi E (1996) Novel microbial technologies for the enhancement of plant growth and biocontrol of fungal diseases in crops. Cahiers Opt Méd 31:447–456
Olsen KN, Budde BB, Siegumfeldt H, Rechinger KB, Jakobsen M, Ingmer H (2002) Non invasive measurement of bacterial intracellular pH on a single-cell level with green fluorescent protein and fluorescence ratio imaging microscopy. Appl Environ Microbiol 68:4145–4147
Probanza A, Mateos JL, Lucas Garcia JA, Ramos B, De Felipe MR, Gutierrez Manero FJ (2001) Effects of Inoculation with PGPR Bacillus and Pisolithus tinctorius on Pinus pinea L. growth, bacterial rhizosphere colonization, and mycorrhizal infection. Microb Ecol 41:140–148
Quax W (1990) Bacillus licheniformis protoplast transformation. In: Cutting SM, Harwood CR (eds) Molecular biological methods for Bacillus. Wiley, Chichester
Rey MW, Ramaiya P, Nelson BA et al (2004) Complete genome sequence of the industrial bacterium Bacillus licheniformis and comparisons with closely related Bacillus species. Genome Biol 5:R77
Rojas A, Holguin G, Glick BR, Bashan Y (2001) Synergism between Phyllobacterium sp. (N(2)-fixer) and Bacillus licheniformis (P-solubilizer), both from a semiarid mangrove rhizosphere. FEMS Microbiol Ecol 35:181–187
Romero D, Perez-Garcia A, Veening JW, de Vicente A, Kuipers OP (2006) Transformation of undomesticated strains of Bacillus subtilis by protoplast electroporation. J Microbiol Methods 66:556–559
Schallmey M, Singh A, Ward OP (2004) Developments in the use of Bacillus species for industrial production. Can J Microbiol 50:1–17
Sneath PHA, Mair NS, Sharpe ME, Holt JG (1986) Bergey’s manual of systematic bacteriology, Vol 2. Williams and Wilkins, Baltimore
Stanley PE (1989) A review of bioluminescent ATP techniques in rapid microbiology. J Biolumin Chemilumin 4:375–380
Tenhami M, Hakkila K, Karp M (2001) Measurement of effects of antibiotics in bioluminescent Staphylococcus aureus RN4220. Antimicrob Agents Chemother 45:3456–3461
Thorne CB, Stull HB (1966) Factors affecting transformation of Bacillus licheniformis. J Bacteriol 91:1012–1020
Virta M, Lineri S, Kankaanpää P et al (1998) Determination of complement-mediated killing of bacteria by viability staining and bioluminescence. Appl Environ Microbiol 64:515–519
Vivas A, Marulanda A, Ruiz-Lozano JM, Barea JM, Azcon R (2003) Influence of a Bacillus sp. on physiological activities of two arbuscular mycorrhizal fungi and on plant responses to PEG-induced drought stress. Mycorrhiza 13:249–256
Webb CD, Decatur A, Teleman A, Losick R (1995) Use of green fluorescent protein for visualization of cell-specific gene expression and subcellular protein localization during sporulation in Bacillus subtilis. J Bacteriol 177:5906–5911
Xue GP, Johnson JS, Dalrymple BP (1999) High osmolarity improves the electro-transformation efficiency of the gram-positive bacteria Bacillus subtilis and Bacillus licheniformis. J Microbiol Methods 34:183–191
Xue M, Ma J, Wang ZX (2004) Effect of high osmolarity on electrotransformation efficiency of bacteria. J Wuxi Uni Light Ind 23:98–100
Acknowledgments
We wish to thank Ville Santala for his helpful suggestions and for the laboratory assistance. We are also grateful to Katariina Tolvanen, Nina Virolainen, and Laura Nevatalo for technical support. Special thanks to Anna-Liisa Välimaa at Tampere University of Technology and Stefania Arioli at University of Milan for their kind help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tamagnini, I., Guglielmetti, S., Mora, D. et al. Generation and Comparison of Bioluminescent and Fluorescent Bacillus licheniformis . Curr Microbiol 57, 245–250 (2008). https://doi.org/10.1007/s00284-008-9184-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-008-9184-y