Abstract
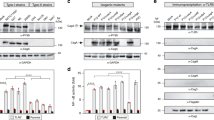
A product of the Helicobacter pylori hp0596 gene (Tip-α) is a highly immunogenic homodimeric protein, unique for this bacterium. Cell fractionation experiments indicate that Tip-α is anchored to the inner membrane. In contrast, the three-dimensional model of the protein suggests that Tip-α is soluble or, at least, largely exposed to the solvent. hp0596 gene knockout resulted in a significant decrease in the level of H. pylori colonization as measured by real-time PCR assay. In addition, the Tip-α recombinant protein was determined to stimulate macrophage to produce IL-1α and TNF-α. Both results imply that Tip-α is rather loosely connected to the inner membrane and potentially released during infection.


Similar content being viewed by others
References
Adamczak R, Porollo A, Meller J (2004) Accurate prediction of solvent accessibility using neural networks-based regression. Proteins 56:753–767
Algood HM, Cover TL (2006) Helicobacter pylori persistence: an overview of interactions between H. pylori and host immune defenses. Clin Microbiol Rev 19:597–613
Alm RA, Ling LS, Moir DT et al. (1999) Genomic-sequence comparison of two unrelated isolates of the human gastric pathogen Helicobacter pylori. Nature 397:176–180
Ames GF, Prody C, Kustu S (1984) Simple, rapid, and quantitative release of periplasmic proteins by chloroform. J Bacteriol 160:1181–1183
Baik SC, Kim KM, Song SM et al. (2004) Proteomic analysis of the sarcosine-insoluble outer membrane fraction of Helicobacter pylori strain 26695. J Bacteriol 186:949–955
Beales IL (2002) Effect of interlukin-1beta on proliferation of gastric epithelial cells in culture. BMC Gastroenterol 2:7
Blaser MJ, Hopkins JA, Berka RM et al. (1983) Identification and characterization of Campylobacter jejuni outer membrane proteins. Infect Immun 42:276–284
Bumann D, Aksu S, Wendland M et al. (2002) Proteome analysis of secreted proteins of the gastric pathogen Helicobacter pylori. Infect Immun 70:3396–3403
Cover TL, Blanke SR (2005) Helicobacter pylori VacA, a paradigm for toxin multifunctionality. Nat Rev Microbiol 3:320–332
Cuff JA, Barton GJ (2000) Application of multiple sequence alignment profiles to improve protein secondary structure prediction. Proteins 40:502–511
Dzwonek A, Mikula M, Woszczynski M et al. (2004) Protective effect of vaccination with DNA of the H. pylori genomic library in experimentally infected mice. Cell Mol Biol Lett 9:483–495
Filip C, Fletcher G, L. Wulff JL, et al. (1973) Solubilization of the cytoplasmic membrane of Escherichia coli by the ionic detergent sodium-lauryl sarcosinate. J Bacteriol 175:966–972
Fischer D (2000) Hybrid fold recognition:combining sequence derived properties with evolutionary information. In: Altman RB (ed) Pacific Symposium on Biocomputing. World Scientific, Singapore, pp 119–130
Godlewska R, Dzwonek A, Mikula M et al. (2006) Helicobacter pylori protein oxidation influences the colonization process. Int J Med Microbiol 296:321–324
Gorrell RJ, Yang J, Kusters JG et al. (2005) Restriction of DNA encoding selectable markers decreases the transformation efficiency of Helicobacter pylori. FEMS Immunol Med Microbiol 44:213–219
Haas G, Karaali G, Ebermayer K et al. (2002) Immunoproteomics of Helicobacter pylori infection and relation to gastric disease. Proteomics 2:313–324
Jones AC, Logan RP, Foynes S et al. (1997) A flagellar sheath protein of Helicobacter pylori is identical to HpaA, a putative N-acetylneuraminyllactose-binding hemagglutinin, but is not an adhesin for AGS cells. J Bacteriol 179:5643–5647
Jones DT (1999) GenTHREADER: an efficient and reliable protein fold recognition method for genomic sequences. J Mol Biol 287:797–815
Jungblut PR, Bumann D, Haas G et al. (2000) Comparative proteome analysis of Helicobacter pylori. Mol Microbiol 36:710–725
Karplus K, Karchin R, Draper J et al. (2003) Combining local-structure, fold-recognition, and new fold methods for protein structure prediction. Proteins 53:491–496
Keenan J, Oliaro J, Domigan N et al. (2000) Immune response to an 18-kilodalton outer membrane antigen identifies lipoprotein 20 as a Helicobacter pylori vaccine candidate. Infect Immun 68:3337–3343
Kelley LA, MacCallum RM, Sternberg MJ (2000) Enhanced genome annotation using structural profiles in the program 3D-PSSM. J Mol Biol 299:499–520
Kimmel B, Bosserhoff A, Frank R et al. (2000) Identification of immunodominant antigens from Helicobacter pylori and evaluation of their reactivities with sera from patients with different gastroduodenal pathologies. Infect Immun 68:915–920
Kosinski J, Cymerman IA, Feder M et al. (2003) A “FRankenstein’s monster” approach to comparative modeling: merging the finest fragments of fold-recognition models and iterative model refinement aided by 3D structure evaluation. Proteins 53(Suppl 6):369–379
Kostrzynska M, PW OT, Taylor DE et al. (1994) Molecular characterization of a conserved 20-kilodalton membrane-associated lipoprotein antigen of Helicobacter pylori. J Bacteriol 176:5938–5948
Kurowski MA, Bujnicki JM (2003) GeneSilico protein structure prediction meta-server. Nucleic Acids Res 31:3305–3307
Kusters JG, van Vliet AH, Kuipers EJ (2006) Pathogenesis of Helicobacter pylori infection. Clin Microbiol Rev 19:449–490
Kuzuhara T, Suganuma M, Kurusu M et al. (2007) Helicobacter pylori-secreting protein Tipalpha is a potent inducer of chemokine gene expressions in stomach cancer cells. J Cancer Res Clin Oncol 133:287–296
Kuzuhara T, Suganuma M, Tsuge H et al. (2005) Presence of a motif conserved between Helicobacter pylori TNF-alpha inducing protein (Tipalpha) and penicillin-binding proteins. Biol Pharm Bull 28:2133–2137
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–695
Lazowska I, Trzeciak L, Godlewska R et al. (2000) In search of immunogenic Helicobacter pylori proteins by screening of expression library. Digestion 61:14–21
Lee A, O’Rourke J, De Uungria MC et al. (1997) A standardized mouse model of Helicobacter pylori infection: introducing the Sydney strain. Gastroenterology 112:1386–1397
Lundstrom J, Rychlewski L, Bujnicki JM et al. (2001) Pcons: a neural-network-based consensus predictor that improves fold recognition. Protein Sci 10:2354–2362
McAtee CP, Lim MY, Fung K et al. (1998) Identification of potential diagnostic and vaccine candidates of Helicobacter pylori by two-dimensional gel electrophoresis, sequence analysis, and serum profiling. Clin Diagn Lab Immunol 5:537–542
McGuffin LJ, Bryson K, Jones DT (2000) The PSIPRED protein structure prediction server. Bioinformatics 16:404–405
Meiler J, Baker D (2003) Coupled prediction of protein secondary and tertiary structure. Proc Natl Acad Sci USA 100:12105–12110
Mikula M, Dzwonek A, Jagusztyn-Krynicka EK et al. (2003) Quantitative detection for low levels of Helicobacter pylori infection in experimentally infected mice by real-time PCR. J Microbiol Methods 55:351–359
Mini R, Bernardini G, Salzano AM et al. (2006) Comparative proteomics and immunoproteomics of Helicobacter pylori related to different gastric pathologies. J Chromatogr B Anal Technol Biomed Life Sci 833:63–79
Narita S, Tokuda H (2007) Amino acids at positions 3 and 4 determine the membrane specificity of Pseudomonas aeruginosa lipoproteins. J Biol Chem 282:13372–13378
O’Toole P, Janzon L, Doig P et al. (1995) The putative neuraminyllactose-binding hemagglutinin HpaA of Helicobacter pylori CCUG 17874 is a lipoprotein. J Bacteriol 177:6049–6057
Ouali M, King RD (2000) Cascaded multiple classifiers for secondary structure prediction. Protein Sci 9:1162–1176
Radosz-Komoniewska H, Bek T, Jozwiak J et al. (2005) Pathogenicity of Helicobacter pylori infection. Clin Microbiol Infect 11:602–610
Rost B, Yachdav G, Liu J (2004) The PredictProtein server. Nucleic Acids Res 32:W321–W326
Rychlewski L, Jaroszewski L, Li W et al. (2000) Comparison of sequence profiles. Strategies for structural predictions using sequence information. Protein Sci 9:232–241
Sabarth N, Hurwitz R, Meyer TF et al. (2002) Multiparameter selection of Helicobacter pylori antigens identifies two novel antigens with high protective efficacy. Infect Immun 70:6499–6503
Sambrook J, Russell DW (2001) Molecular cloning. A laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Seydel A, Gounon P, Pugsley AP (1999) Testing the ‘+2 rule’ for lipoprotein sorting in the Escherichia coli cell envelope with a new genetic selection. Mol Microbiol 34:810–821
Shi J, Blundell TL, Mizuguchi K (2001) FUGUE: sequence-structure homology recognition using environment-specific substitution tables and structure-dependent gap penalties. J Mol Biol 310:243–257
Simons KT, Kooperberg C, Huang E et al. (1997) Assembly of protein tertiary structures from fragments with similar local sequences using simulated annealing and Bayesian scoring functions. J Mol Biol 268:209–225
Soding J (2005) Protein homology detection by HMM-HMM comparison. Bioinformatics 21:951–960
Suganuma M, Kurusu M, Suzuki K et al. (2005) New tumor necrosis factor-alpha-inducing protein released from Helicobacter pylori for gastric cancer progression. J Cancer Res Clin Oncol 131:305–313
Suganuma M, Kuzuhara T, Yamaguchi K et al. (2006) Carcinogenic role of tumor necrosis factor-alpha inducing protein of Helicobacter pylori in human stomach. J Biochem Mol Biol 39:1–8
Tomb JF, White O, Kerlavage AR et al. (1997) The complete genome sequence of the gastric pathogen Helicobacter pylori. Nature 388:539–547
Vanet A, Marsan L, Labigne A et al. (2000) Inferring regulatory elements from a whole genome. An analysis of Helicobacter pylori sigma(80) family of promoter signals. J Mol Biol 297:335–353
Yoshida M, Wakatsuki Y, Kobayashi Y et al. (1999) Cloning and characterization of a novel membrane-associated antigenic protein of Helicobacter pylori. Infect Immun 67:286–293
Zhang Z, H., Niesel DW, Peterson JW et al. (1998) Lipoprotein release by bacteria: potential factor in bacterial pathogenesis. Infect Immun 66:5196–5201
Zhou H, Zhou Y (2004) Single-body residue-level knowledge-based energy score combined with sequence-profile and secondary structure information for fold recognition. Proteins 55:1005–1013
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Godlewska, R., Pawlowski, M., Dzwonek, A. et al. Tip-α (hp0596 Gene Product) Is a Highly Immunogenic Helicobacter pylori Protein Involved in Colonization of Mouse Gastric Mucosa. Curr Microbiol 56, 279–286 (2008). https://doi.org/10.1007/s00284-007-9083-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-007-9083-7