Abstract
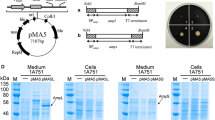
It was reported that PL promoter and alkaline phosphatase (phoA) signal peptide were used to construct secretory expression plasmid suitable to express glucagon and [Des-His1] glucagon in E. coli BL21 herein. Expression studies showed these two peptides could be expressed and secreted into the culture medium. The expression yield of recombinant glucagon reached 3.46 mg/L/OD600 unit of cells in shake flask. The yield of [Des-His1] glucagon was found to be higher than that of glucagon. In addition, some factors involved in secretion were studied too.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 26 September 2002 / Accepted: 24 October 2002
Rights and permissions
About this article
Cite this article
Wen, C., Gan, R. & Zhu, S. Construction of Secretory Expression System Suitable to Express Glucagon Under the Control of PL Promoter. Curr Microbiol 47, 0180–0185 (2003). https://doi.org/10.1007/s00284-002-3988-y
Issue Date:
DOI: https://doi.org/10.1007/s00284-002-3988-y