Abstract
Purpose
Oxaliplatin is a crucial component of the combinatorial chemotherapeutic standard of care for advanced colorectal cancer (CRC). Unfortunately, a serious barrier to effective oxaliplatin treatment is drug resistance due to epithelial-mesenchymal transitioning (EMT). Interestingly, stable oxaliplatin-resistant CRC cell lines show differential expression of miR-1915-3p; thus, this microRNA may represent a potential modifier of oxaliplatin resistance in CRC cells.
Methods
miR-1915-3p was over-expressed in oxaliplatin-resistant CRC cells and a non-tumorigenic intestinal cell line (FHC) via lentiviral transduction. Extracellular vesicles (EVs) were purified from transduced FHC cells and co-incubated with CRC cells. Expression levels of miR-1915-3p and other RNA species were assessed by RT-qPCR, while protein expression levels were assessed by Western blotting. The effects of miR-1915-3p on CRC viability were evaluated by proliferation, apoptosis assays, and Transwell assays. Effects of miR-1915-3p over-expression on in vivo oxaliplatin sensitivity was tested via murine xenograft models.
Results
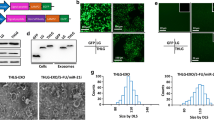
miRNA-1915-3p decreased EMT marker expression in oxaliplatin-resistant CRC cell lines and in vivo. FHC cells were able to produce and secrete miR-1915-3p-containing EVs, which we employed to mediate miR-1915-3p delivery to oxaliplatin-resistant CRC cells and increase their oxaliplatin sensitivity in vivo and in vitro. Mechanistically, miR-1915-3p overexpression downregulated the EMT-promoting oncogenes 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3 (PFKFB3) and ubiquitin carboxyl-terminal hydrolase 2 (USP2) as well as upregulated E-cadherin (a cell adhesion mediator). miR-1915-3p’s effects on chemosensitivity and EMT were mediated by its regulation of PFKFB3 and USP2.
Conclusion
Exosomal delivery of miR-1915-3p can improve the chemotherapeutic efficacy of oxaliplatin in CRC cells by suppressing the EMT-promoting oncogenes PFKFB3 and USP2.




Similar content being viewed by others
Availability of data and material
Please contact author for data requests.
References
Allen KE, Weiss GJ (2010) Resistance may not be futile: microRNA biomarkers for chemoresistance and potential therapeutics. Mol Cancer Ther 9:3126–3136
Altanerova U, Jakubechova J, Benejova K, Priscakova P, Pesta M, Pitule P, Topolcan O, Kausitz J, Zduriencikova M, Repiska V (2019) Prodrug suicide gene therapy for cancer targeted intracellular by mesenchymal stem cell exosomes. Int J Cancer 144:897–908
Arango D, Wilson A, Shi Q, Corner G, Aranes M, Nicholas C, Lesser M, Mariadason J, Augenlicht LH (2004) Molecular mechanisms of action and prediction of response to oxaliplatin in colorectal cancer cells. Br J Cancer 91:1931
Bartel DP (2009) MicroRNAs: target recognition and regulatory functions. Cell 136:215–233
Benassi B, Flavin R, Marchionni L, Zanata S, Pan Y, Chowdhury D, Marani M, Strano S, Muti P, Blandino G (2012) MYC is activated by USP2a-mediated modulation of microRNAs in prostate cancer. Cancer Discov 2:236–247
Berezikov E (2011) Evolution of microRNA diversity and regulation in animals. Nat Rev Genet 12:846
Boni V, Bitarte N, Cristobal I, Zarate R, Rodriguez J, Maiello E, Garcia-Foncillas J, Bandres E (2010) miR-192/miR-215 influence 5-fluorouracil resistance through cell cycle-mediated mechanisms complementary to its post-transcriptional thymidilate synthase regulation. Mol Cancer Ther 9:2265–2275
Brown EJ, Frazier WA (2001) Integrin-associated protein (CD47) and its ligands. Trends Cell Biol 11:130–135
Calvo M, Bartrons R, Castano E, Perales J, Navarro-Sabate A, Manzano A (2006) PFKFB3 gene silencing decreases glycolysis, induces cell-cycle delay and inhibits anchorage-independent growth in HeLa cells. FEBS Lett 580:3308–3314
Chai H, Liu M, Tian R, Li X, Tang H (2011) miR-20a targets BNIP2 and contributes chemotherapeutic resistance in colorectal adenocarcinoma SW480 and SW620 cell lines. Acta Biochim Biophys Sin 43:217–225
Cheshomi H, Matin MM (2019) Exosomes and their importance in metastasis, diagnosis, and therapy of colorectal cancer. J Cell Biochem 120:2671–2686
Clague MJ, Urbé S, Komander D (2019) Breaking the chains: deubiquitylating enzyme specificity begets function. Nat Rev Mol Cell Biol. https://doi.org/10.1038/s41580-019-0112-8
Fang T, Lv H, Lv G, Li T, Wang C, Han Q, Yu L, Su B, Guo L, Huang S (2018) Tumor-derived exosomal miR-1247-3p induces cancer-associated fibroblast activation to foster lung metastasis of liver cancer. Nat Commun 9:191
Gonzalez-Begne M, Lu B, Han X, Hagen FK, Hand AR, Melvin JE, Yates JR III (2009) Proteomic analysis of human parotid gland exosomes by multidimensional protein identification technology (MudPIT). J Proteome Res 8:1304–1314
Gustafsson NM, Färnegårdh K, Bonagas N, Ninou AH, Groth P, Wiita E, Jönsson M, Hallberg K, Lehto J, Pennisi R (2018) Targeting PFKFB3 radiosensitizes cancer cells and suppresses homologous recombination. Nat Commun 9:3872
Han J, Meng Q, Xi Q, Zhang Y, Zhuang Q, Han Y, Jiang Y, Ding Q, Wu G (2016) Interleukin-6 stimulates aerobic glycolysis by regulating PFKFB3 at early stage of colorectal cancer. Int J Oncol 48:215–224
He J, Lee H-J, Saha S, Ruan D, Guo H, Chan C-H (2019) Inhibition of USP2 eliminates cancer stem cells and enhances TNBC responsiveness to chemotherapy. Cell Death Dis 10:285
Hölzel M, Bovier A, Tüting T (2013) Plasticity of tumour and immune cells: a source of heterogeneity and a cause for therapy resistance? Nat Rev Cancer 13:365
Hu G, Drescher KM, Chen X (2012) Exosomal miRNAs: biological properties and therapeutic potential. Front Genet 3:56
Hu J, Cai G, Xu Y, Cai S (2016) The Plasma microRNA miR-1914* and-1915 suppresses chemoresistant in colorectal cancer patients by down-regulating NFIX. Curr Mol Med 16:70–82
O’Loughlin J, A, A Woffindale C, JA Wood M, (2012) Exosomes and the emerging field of exosome-based gene therapy. Curr Gene Ther 12:262–274
Jaiswal S, Jamieson CH, Pang WW, Park CY, Chao MP, Majeti R, Traver D, van Rooijen N, Weissman IL (2009) CD47 is upregulated on circulating hematopoietic stem cells and leukemia cells to avoid phagocytosis. Cell 138:271–285
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61:69–90
Johnsen KB, Gudbergsson JM, Skov MN, Pilgaard L, Moos T, Duroux M (2014) A comprehensive overview of exosomes as drug delivery vehicles—endogenous nanocarriers for targeted cancer therapy. Biochimica et Biophysica Acta (BBA) Rev Cancer 1846:75–87
Kamerkar S, LeBleu VS, Sugimoto H, Yang S, Ruivo CF, Melo SA, Lee JJ, Kalluri R (2017) Exosomes facilitate therapeutic targeting of oncogenic KRAS in pancreatic cancer. Nature 546:498
Katakowski M, Buller B, Zheng X, Lu Y, Rogers T, Osobamiro O, Shu W, Jiang F, Chopp M (2013) Exosomes from marrow stromal cells expressing miR-146b inhibit glioma growth. Cancer Lett 335:201–204
Kurokawa K, Tanahashi T, Iima T, Yamamoto Y, Akaike Y, Nishida K, Masuda K, Kuwano Y, Murakami Y, Fukushima M (2012) Role of miR-19b and its target mRNAs in 5-fluorouracil resistance in colon cancer cells. J Gastroenterol 47:883–895
Li H, Yang BB (2013) Friend or foe: the role of microRNA in chemotherapy resistance. Acta Pharmacol Sin 34:870
Li Y, Zheng Q, Bao C, Li S, Guo W, Zhao J, Chen D, Gu J, He X, Huang S (2015) Circular RNA is enriched and stable in exosomes: a promising biomarker for cancer diagnosis. Cell Res 25:981
Liu T, Zhang X, Du L, Wang Y, Liu X, Tian H, Wang L, Li P, Zhao Y, Duan W (2019) Exosome-transmitted miR-128-3p increase chemosensitivity of oxaliplatin-resistant colorectal cancer. Mol Cancer 18:43
McMillin DW, Negri JM, Mitsiades CS (2013) The role of tumour–stromal interactions in modifying drug response: challenges and opportunities. Nat Rev Drug Discov 12:217
Meads MB, Gatenby RA, Dalton WS (2009) Environment-mediated drug resistance: a major contributor to minimal residual disease. Nat Rev Cancer 9:665
Meyerhardt JA, Mayer RJ (2005) Systemic therapy for colorectal cancer. N Engl J Med 352:476–487
Mor I, Cheung E, Vousden K (2011) Control of glycolysis through regulation of PFK1: old friends and recent additions. Cold Spring Harbor symposia on quantitative biology. Cold Spring Harbor Laboratory Press, pp 211–216
Nakazawa K, Dashzeveg N, Yoshida K (2014) Tumor suppressor p53 induces miR-1915 processing to inhibit Bcl-2 in the apoptotic response to DNA damage. FEBS J 281:2937–2944
Shan J, Zhao W, Gu W (2009) Suppression of cancer cell growth by promoting cyclin D1 degradation. Mol Cell 36:469–476
Simões S, Filipe A, Faneca H, Mano M, Penacho N, Düzgünes N, Pedroso de Lima M (2005) Cationic liposomes for gene delivery. Expert Opin Drug Deliv 2:237–254
Song B, Wang Y, Xi Y, Kudo K, Bruheim S, Botchkina GI, Gavin E, Wan Y, Formentini A, Kornmann M (2009) Mechanism of chemoresistance mediated by miR-140 in human osteosarcoma and colon cancer cells. Oncogene 28:4065
Stevenson LF, Sparks A, Allende-Vega N, Xirodimas DP, Lane DP, Saville MK (2007) The deubiquitinating enzyme USP2a regulates the p53 pathway by targeting Mdm2. EMBO J 26:976–986
Sun Z, Shi K, Yang S, Liu J, Zhou Q, Wang G, Song J, Li Z, Zhang Z, Yuan W (2018) Effect of exosomal miRNA on cancer biology and clinical applications. Mol Cancer 17:147
Telang S, Yalcin A, Clem A, Bucala R, Lane A, Eaton J, Chesney J (2006) Ras transformation requires metabolic control by 6-phosphofructo-2-kinase. Oncogene 25:7225
Vader P, Mol EA, Pasterkamp G, Schiffelers RM (2016) Extracellular vesicles for drug delivery. Adv Drug Deliv Rev 106:148–156
Valeri N, Gasparini P, Braconi C, Paone A, Lovat F, Fabbri M, Sumani KM, Alder H, Amadori D, Patel T (2010) MicroRNA-21 induces resistance to 5-fluorouracil by down-regulating human DNA MutS homolog 2 (hMSH2). Proc Natl Acad Sci 107:21098–21103
van der Meel R, Fens MH, Vader P, van Solinge WW, Eniola-Adefeso O, Schiffelers RM (2014) Extracellular vesicles as drug delivery systems: lessons from the liposome field. J Control Release 195:72–85
Voinnet O (2009) Origin, biogenesis, and activity of plant microRNAs. Cell 136:669–687
Wang B-D, Kline CLB, Pastor DM, Olson TL, Frank B, Luu T, Sharma AK, Robertson G, Weirauch MT, Patierno SR (2010) Prostate apoptosis response protein 4 sensitizes human colon cancer cells to chemotherapeutic 5-FU through mediation of an NFκB and microRNA network. Mol Cancer 9:98
Wang C-L, Wang J-Y, Liu Z-Y, Ma X-M, Wang X-W, Jin H, Zhang X-P, Fu D, Hou L-J, Lu Y-C (2014) Ubiquitin-specific protease 2a stabilizes MDM4 and facilitates the p53-mediated intrinsic apoptotic pathway in glioblastoma. Carcinogenesis 35:1500–1509
Xu K, Liang X, Cui D, Wu Y, Shi W, Liu J (2013) miR-1915 inhibits Bcl-2 to modulate multidrug resistance by increasing drug-sensitivity in human colorectal carcinoma cells. Mol Carcinog 52:70–78
Yang AD, Fan F, Camp ER, van Buren G, Liu W, Somcio R, Gray MJ, Cheng H, Hoff PM, Ellis LM (2006) Chronic oxaliplatin resistance induces epithelial-to-mesenchymal transition in colorectal cancer cell lines. Clin Cancer Res 12:4147–4153
Zhang X, Yuan X, Shi H, Wu L, Qian H, Xu W (2015) Exosomes in cancer: small particle, big player. J Hematol Oncol 8:83
Zhang Y, Liu D, Chen X, Li J, Li L, Bian Z, Sun F, Lu J, Yin Y, Cai X (2010) Secreted monocytic miR-150 enhances targeted endothelial cell migration. Mol Cell 39:133–144
Funding
None.
Author information
Authors and Affiliations
Contributions
Conceived and designed the study: ZX and YL. Performed the experimental procedures: QL and YL. Analyzed the data: ZX, YL, SW, QL and YL. Drafted the manuscript: QL and YL.
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declare that they have no competing interests.
Ethics approval
All protocols and guidelines were approved in advance by the Ethics Committee of the Third Hospital of Nanchang (Nanchang, China).
Consent to publication
All authors confirm of submission and publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xiao, Z., Liu, Y., Li, Q. et al. EVs delivery of miR-1915-3p improves the chemotherapeutic efficacy of oxaliplatin in colorectal cancer. Cancer Chemother Pharmacol 88, 1021–1031 (2021). https://doi.org/10.1007/s00280-021-04348-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-021-04348-5